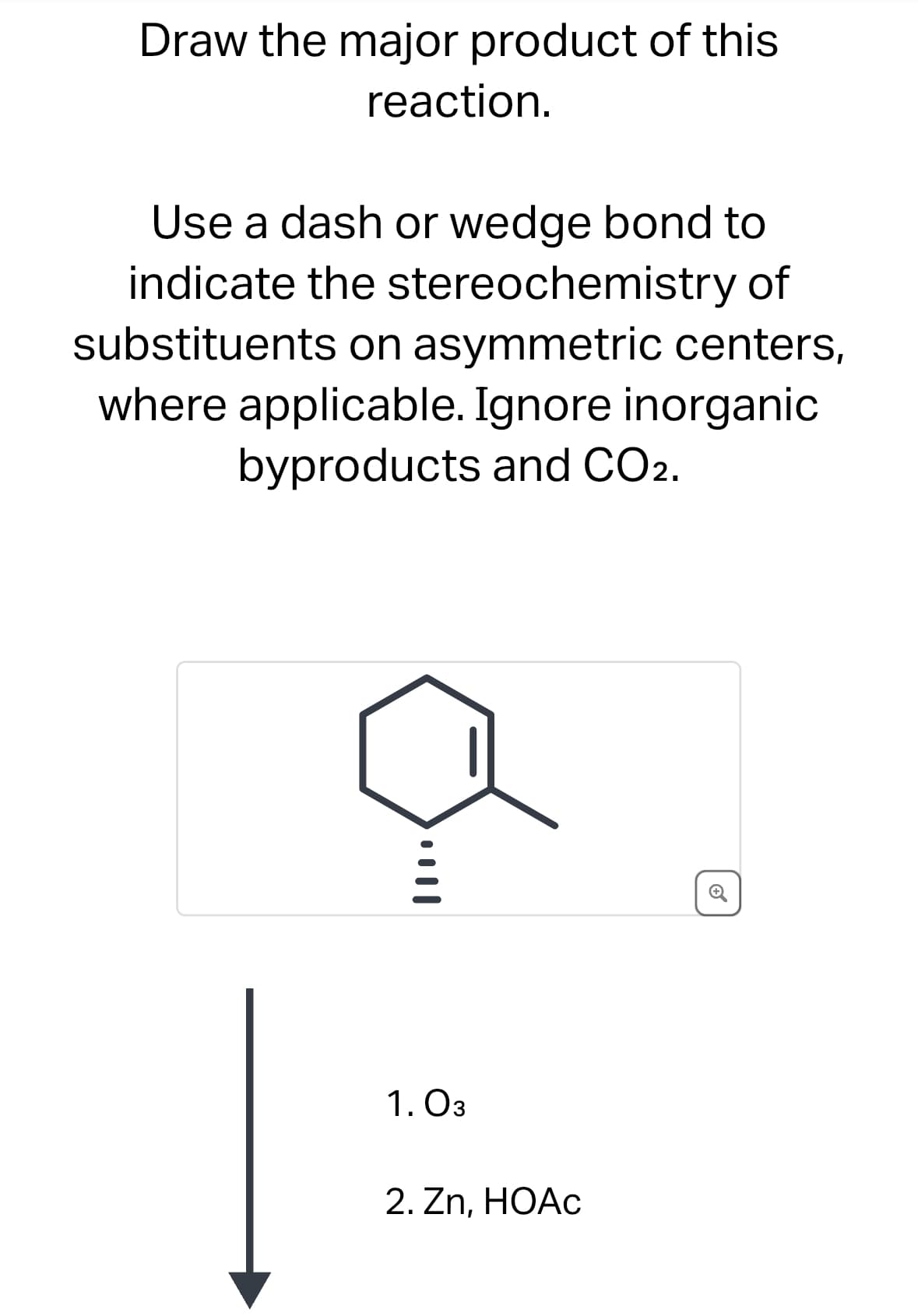
Draw the major product of this reaction. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, where applicable. Ignore inorganic byproducts and CO2. 1. 03 2. Zn, HOAC +
Q: Draw the major product for this reaction. Ignore inorganic byproducts. 1. disiamylborane, THF 2.…
A:
Q: 0. CH3-CH2- .CH. O CH3- -CH21 NH3 glutamate mutate coenzyme B12 HN CH3 CH3 CH2-CH2-CH- HN CH3…
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw the structure(s) of the major product(s) of the following reaction after workup to neutralize…
A:
Q: Protons H, and Hb in the compound given are Br H Hb Ha homotopic enantiotopic diaster@gtopic ○…
A: The structures possesses diastereoisomer relation, obtained after replacing protons individually.…
Q: Chemistry
A: The freezing points of the given substances are as follows: H₂O (Water): The freezing point is…
Q: Can you provide a mechanism for this reaction?
A: I have assumed the bigger group as (S) so that I could explain mechanism easily but still you have…
Q: please predict each of the following with the product
A: Step 1: Lithium Aluminium hydride is a strong reducing agent, it reduces aldehydes, ketone,…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. 1. acetonitrile /…
A:
Q: Write the equation for the other half-reaction, called the dark reaction, for photosynthesis .…
A: The objective of the question is to write the chemical equation for the dark reaction, also known as…
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: Step 1: Reaction of 2-iodopent-1-ene on heating with methyl amide ion gives E2 elimination…
Q: synthetic polymers: Draw AIBN + heat or light.
A: Step 1: Step 2: Step 3: Step 4:
Q: Identify the following amino acids and their 3-letter and 1-letter abbreviations. (Use capital…
A: Aspartic acid and glutamine are two amino acids that are going to be discussed in further detail in…
Q: Help with question
A: a.) Based on the structure, benzophenone is relatively more polar than biphenyl. Since the TLC plate…
Q: I understand how to solve the questions, but in the overall question how do we know that the unknown…
A:
Q: Identify the structural isomer of the following molecule: OH
A: The molecule you presented is ethanol, with the chemical formula C2H5OH. It features a two-carbon…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. ⑤ 1. Hg(OAc)2, H2O 2. NaBH4,…
A:
Q: Enough of a monoprotic weak acid is dissolved in water to produve a 0.0111 M solution. The pH of the…
A:
Q: A 0.892-g sample of an unknown gas has a volume of 580 mL and a pressure of 421 mm Hg at 31.1 °C.…
A:
Q: Each row of the table below describes an aqueous solution at about 25 °C. Complete the table. That…
A: Step 1: Solution of part(A) Given,[H3O+] = 3.7 × 10-8 mol/L we know, => pH = -log[H3O+] on…
Q: What masses of dimethylamine and dimethylammonium chloride do you need to prepare 5.00 L of pH =…
A: Step-1: pH = 12Volume= 5LConcentration of both = 0.5M.pKa of dimethylamine= 10.73. Step 2: The…
Q: Draw structural formulas for organic products A and B in the window below. -CI Li pentane H₂O A B •…
A: Step 1:Alkyl chloride reacts with Lithium to form alkyl lithium . A is cyclopentyl lithium. Step…
Q: What is the expected major product for the following reaction? 1. Hg(OAc)2, H₂O 2. NaBH4, NaOH OH HO…
A: Step 1: Step 2: Step 3: Step 4:
Q: Predict the potential during a potentiometric redox titration of Fe2+ (analyte) with Ce4+ (titrant)…
A: Step 1: Step 2: Step 3: Step 4:
Q: what starting material would be required to obtain these products
A:
Q: For the provided reaction, which option among those listed both shows the allowed product of an…
A: The question is asking to identify the correct product of an electrocyclization reaction and the…
Q: Please help
A: 2. Tollens' reagent is commonly used to test for the presence of aldehydes but does not typically…
Q: CI + ΑΙ CH2C12 4 + HCI
A: Step 1: Step 2: Step 3: Step 4:
Q: + Draw the missing organic structures in the following multistep synthesis. Show the final product…
A: Step 1: Step 2: Step 3: Step 4:
Q: QUESTION 9 How many grams of solute are needed to make 1300 mL of a 4.5% (m/v) NH4Cl solution? Do…
A: 9). - Calculate the mass of solute using the formula for mass/volume percent; % (m/v) = [mass of…
Q: Calculate the value of is for H+, He+ and Li+2 ions at r = 0, r = a, and r = 4a,, Rationalize the…
A: Step 1: Step 2: Step 3: Step 4:
Q: A voltaic cell consists of Cu^2+/Cu and Cd ^2+/Cd half-cells. Each compartment has a volume of 0.500…
A: To find the concentration of [Cu^2+] when the cell voltage is 0.728 V, we can use the Nernst…
Q: how to get 1-(1-methyl-3,4-dihydro-1H-isoquinolin-2-yl)ethanone from…
A: 1. Starting Material Analysis- **N-ethenyl-N-(2-phenylethyl)acetamide** has the structure where an…
Q: + H2 Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and charges),…
A: Detailed explanation:(H3C)2CH-CH=CH-CH3 + H2/Pt ---> (CH3)2CH-CH2-CH2-CH3…
Q: Create a titration curve when 13 mL of 0.12 M HCl is titrated with 0.09 M NaOH. Generate a titration…
A: Volume NaOH,…
Q: An electron in the n = 6 level emits a photon with a wavelength of 410.2 nm. To what energy level…
A:
Q: A buffer solution made from HNO2 and KNO2 has a pH of 3.72. If PK for pka HNO2 is 3.35, what is the…
A: Step 1: Buffer solutionsAqueous solutions containing a mixture of a weak acid and its conjugate base…
Q: Consider the following carbocation (i.e., a molecule with a positive formal charge on a carbon).…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A:
Q: Phosgene decomposes to carbon monoxide and chlorine gas. COCl2 (g) CO (g) + Cl2 (g) If the reaction…
A: The question is asking about the effect of adding more phosgene gas (COCl2) to a reaction that has…
Q: + Ph,POCH H₂O CHO
A: The image you sent appears to be a multiple choice question about organic chemistry. The question…
Q: 8
A: Step 1: Step 2: Step 3: Step 4:
Q: According to this reaction energy diagram, the value of the change in energy is. Potential energy…
A: Step 1: Identify the parts of the diagram Point A in the given diagram represents the reactants and…
Q: A reaction with an enthalpy of -16.9 kJ and and entropy of -265 J/K is being run at -115°C. What is…
A: Given: ΔH=−16.9kJ;ΔS=−265J/K;ΔG=???;T=−115°CStep 1: Write the equation we need.ΔG=ΔH−TΔS Step 2:…
Q: Draw the major product of this reaction. Ignore any inorganic byproducts. HBr (1 equiv)
A: Step 1:
Q: Analyze the H1-NMR of 3-Carboxy-Umbelliferone. Assign the protons in this structure to peaks seen in…
A: Step 1:Step 2:Step 3:Here's a simplified example of what the table might look like: Proton…
Q: Determine ∆S° for 2 O₃(g) → 3 O₂(g). Substance Sf° (J/mol・K) O₃ (g) 239 O₂ (g) 205
A:
Q: 3 a) State Zaitsev's rule and show the structures elimination product(s) of the following compounds…
A: The objective of the question is to explain Zaitsev's rule and apply it to predict the elimination…
Q: Need help with question please
A: (5) using too much solvent is the correct answer.Reason: if too much solvent is used then it will…
Q: The general solution of Schrodinger Equation is a wave, Ψ=eikx=Cos kx + isinkx, where k= (2m/ħ2)1/2.…
A: Step 1:Step 2:Step 3:
Q: 5. Calculate the pH of 1.5 M solution of hydroxylamine, NH₂OH at 25°C. K = 9.1 x 10-9 HONH2(aq) +…
A: The objective of the question is to calculate the pH of a 1.5 M solution of hydroxylamine, NH2OH at…
Step by step
Solved in 2 steps with 1 images

- In some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer is present. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1- phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Whichproduct predominates—the product of inversion or the product of retention of conguration? (c) Suggest an explanation for this phenomenon.In some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer ispresent. For example, treatment of optically pure 1-bromo-1phenylpropane with water forms 1-phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Which product predominates—the product of inversion or the product of retention of configuration? (c) Suggest an explanation for this phenomenon.Explain how substituents on thearomatic ring promote substitutionat some positions but not at others
- In some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer is present. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1-phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Which product predominates—the product of inversion or the product of retention of configuration? (c) Suggest an explanation for this phenomenon.fill in the blanks. where appropriate, write the major product with proper stereochemistryAssign every stereo center in callyspongiolide as either R or S using CIP notation
- Provide the major product of the reaction sequence. If cis/trans isomers are possible, draw only the major isomer. If enantiomers are possible, do not specify configuration.In some nucleophilic substitutions under SN1 conditions, completeracemization does not occur and a small excess of one enantiomer ispresent. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1-phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data.(b) Which product predominates—the product of inversion or the product of retention of configuration? (c) Suggest an explanation for this phenomenon.Draw the products of this reduction of a ketone with sodium borohydride. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, where applicableIgnore any inorganic byproducts.
- Give the MAJOR PRODUCT/S of the following reactions. Include stereochemistry (wedges and dashes) asappropriate. Draw all major enantiomers and diastereomers.Draw the (pericyclic) reaction (reaction 1), including all mechanistic steps, that lead from the starting materials to the intermediate product. Show all isomers that can be formed and provide an explanation as to why that is. Determine the configuration of all chiral centers. I included the molecule, the retrosynthesis of the molecule, and reaction 1 to help solve this question.Discuss comprehensively why -Cl and -NO2 aredeactivators and why -Cl is a weak and -NO2 astrong deactivator?

