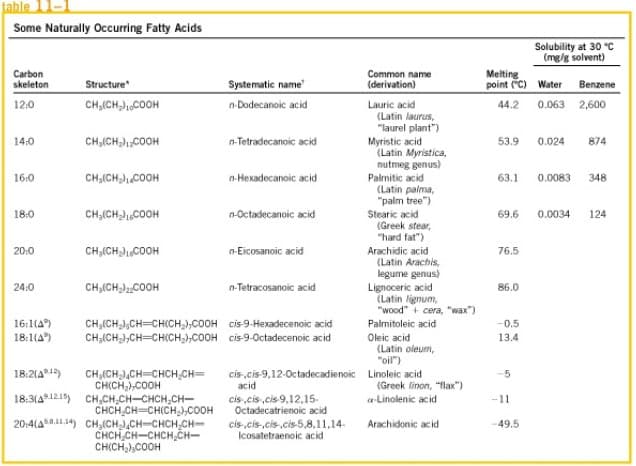
From Table 1.1 indicates, among other things, the melting points of various fatty acids. Analyze the trend that the melting points of saturated fatty acids follow, from n-dodecanoic (CH3-(CH2) 10-COOH) to n-tetracosanoic (CH3-(CH2)22-COOH), and answer both next questions. Briefly justify your answers. i) What value would you expect the melting point of triacontanoic acid (CH3-(CH2) 28-COOH) to have? Choose one of the following options: a) More than 200 ° C b) About 94 ° C c) Less than 86 ° C d) Exactly 86.1 ° C ii) What value would you expect the melting
From Table 1.1 indicates, among other things, the melting points of various fatty acids. Analyze the trend that the melting points of saturated fatty acids follow, from n-dodecanoic (CH3-(CH2) 10-COOH) to n-tetracosanoic (CH3-(CH2)22-COOH), and answer both next questions. Briefly justify your answers. i) What value would you expect the melting point of triacontanoic acid (CH3-(CH2) 28-COOH) to have? Choose one of the following options: a) More than 200 ° C b) About 94 ° C c) Less than 86 ° C d) Exactly 86.1 ° C ii) What value would you expect the melting
World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter20: Organic Chemistry
Section: Chapter Questions
Problem 46A
Related questions
Question
From Table 1.1 indicates, among other things, the melting points of various fatty acids. Analyze the trend that the melting points of saturated fatty acids follow, from n-dodecanoic (CH3-(CH2) 10-COOH) to n-tetracosanoic (CH3-(CH2)22-COOH), and answer both next questions. Briefly justify your answers.
i) What value would you expect the melting point of triacontanoic acid (CH3-(CH2) 28-COOH) to have? Choose one of the following options:
a) More than 200 ° C
b) About 94 ° C
c) Less than 86 ° C
d) Exactly 86.1 ° C
ii) What value would you expect the melting point of octanoic acid (CH3-(CH2) 6-COOH) to have? Choose one of the following options:
a) 43.5 ° C
b) -20 ° C
c) About 20 ° C
Transcribed Image Text:table
Some Naturally Occurring Fatty Acids
Solubility at 30 °C
(mg/g solvent)
Carbon
skeleton
Structure
Systematic name
Common name
(derivation)
Melting
point ("C) Water
Benzene
12:0
CH,(CH,.C0OH
n Dodecanoic acid
44.2
0.063 2,600
Lauric acid
(Latin laurus,
"laurel plant")
Myristic acid
(Latin Myristica,
nutmeg genus)
Palmitic acid
(Latin palma,
"palm tree")
14:0
CH,(CH,),COOH
n-Tetradecanoic acid
53.9
0.024
874
16:0
CH,(CH,.COOH
n-Hexadecanoic acid
63.1
0.0083
348
18:0
CH;(CHJsCOOH
n-Octadecanoic acid
Stearic acid
(Greek stear,
"hard fat")
69.6
0.0034
124
20:0
CH,(CHJuCOOH
n-Eicosanoic acid
Arachidic acid
(Latin Arachis,
legume genus)
76.5
24:0
CH,(CH)COOH
n-Tetracosanoic acid
86.0
Lignoceric acid
(Latin lignum,
"wood" + cera, "wax")
16:1(A)
CH,(CHJ;CH=CH(CH,COOH cis-9-Hexadecenoic acid
CH,(CH,),CH=CH(CH,)»COOH cis-9-Octadecenoic acid
Palmitoleic acid
--0,5
18:1(4)
Oleic acid
(Latin oleum,
"oil")
13.4
CH,(CHJ.CH CHCH CH=
CHICH,,COOH
18:3(4121) CH,CH,CH-CHCH,CH-
CHCH,CH=CH(CH,),COOH
20:4(A. CH,(CHJ,CH-CHCH,CH-
CHCH CH-CHCH,CH-
CH(CH,),COOH
18:2(A19)
cis-,cis-9,12-Octadecadienoic Linoleic acid
acid
-5
(Greek linon, "flax")
cis ,cis,cis-9,12,15-
Octadecatrienoic acid
a-Linolenic acid
-11
cis,cis,cis-.cis-5,8,11,14-
Icosatetraenoic acid
Arachidonic acid
-49.5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning