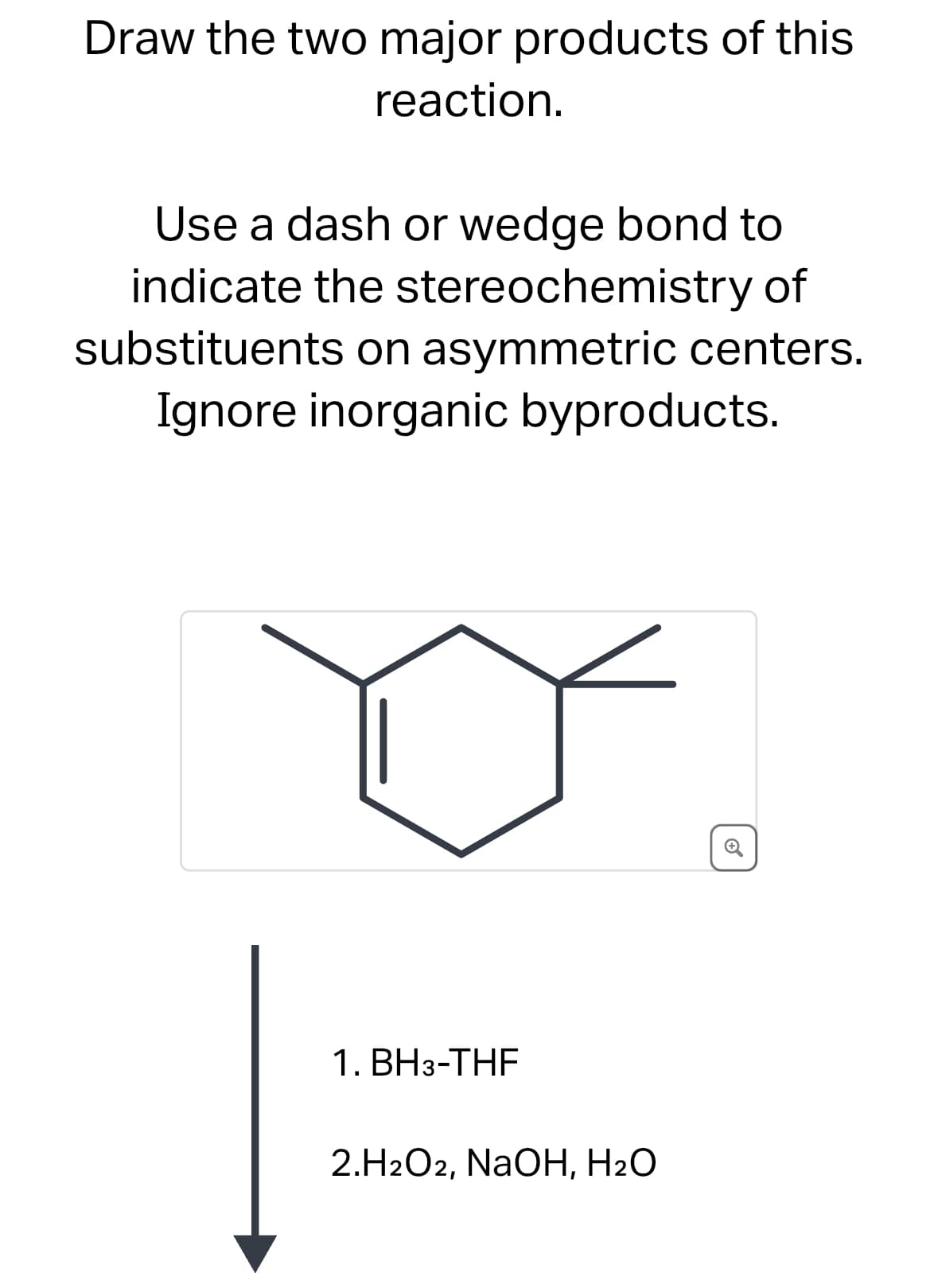
Draw the two major products of this reaction. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers. Ignore inorganic byproducts. ⑤ 1. BH 3-THF 2.H2O2, NaOH, H2O
Q: Please don't upload any image just give me the answer with proper explanation
A: Approach to solving the question:Read alot of related books or research onlineDetailed…
Q: H3C-CH-CH=CH₂ C=C-H 2eq. KMnO4 NaOH, H₂O
A:
Q: What molecules correspond to IR spectra C and D? Briefly justify (only one molecule per spectrum).
A:
Q: 5. The element Chromium has a KB X-ray wavelength of 0.209nm. The K-shell binding energy is 5989eV.…
A: Step 1: Step 2: Step 3: Step 4:
Q: Q3. The main product of the following reaction is likely to be: CI CI CI hy A) a 4-membered…
A:
Q: 5. Calculate the pH of 1.5 M solution of hydroxylamine, NH₂OH at 25°C. K = 9.1 x 10-9 HONH2(aq) +…
A: The objective of the question is to calculate the pH of a 1.5 M solution of hydroxylamine, NH2OH at…
Q: You are required to use commercially available Caffeine Tablet to obtain caffeine for your…
A: The objective of the question is to determine the number of caffeine tablets needed to compound 10…
Q: Only typed solution No AI generated solution
A: The objective of the question is to calculate the wavelength of a photon given its energy and…
Q: A sample of hydrogen gas has a density of g/L at a pressure of 0.820 atm and a temperature of 59°C.…
A:
Q: None
A: Here's a breakdown of why each option is correct or incorrect:Option (a):- The hydroxyl group on the…
Q: Choose the TRUE statement about the effect of the presence of a catalyst on reaction rate. In the…
A: The objective of the question is to identify the correct statement about the effect of a catalyst on…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. conc. HBr Drawing
A:
Q: Calculate the pH, percent dissociation, and concentrations of all species of 0.25M acetic acid,…
A: Given: [HC2H3O2]=0.25M;Ka=1.8x10−5;pH=???;Step 1: Write the dissociation of the…
Q: Draw a structural formula for the major organic product(s) of the reaction shown below. 1. CH3l…
A: Step 1: Step 2: Step 3: Step 4:
Q: If you combine 410.0 mL of water at 25.0°C and 130.0 mL of water at 95.0°C, what is the final…
A:
Q: Analyze the H1-NMR of 3-Carboxy-Umbelliferone. Assign the protons in this structure to peaks seen in…
A: Step 1:Step 2:Step 3:Here's a simplified example of what the table might look like: Proton…
Q: Draw estrogen and testosterone. Number the positions of the core steroid nucleus
A: Estrogen: The core steroid nucleus of estrogen is comprised of four fused rings labeled A, B, C, and…
Q: Phosgene decomposes to carbon monoxide and chlorine gas. COCl2 (g) CO (g) + Cl2 (g) If the reaction…
A: The question is asking about the effect of adding more phosgene gas (COCl2) to a reaction that has…
Q: A 1.44-g sample of an unknown gas has a volume of 896 mL and a pressure of 742 mm Hg at 37.9 °C.…
A: The objective of this question is to calculate the molar mass of an unknown gas given its mass,…
Q: Predict the product(s) for the following reaction. IV HI excess heat OH IV = II OH
A:
Q: Which one of the following substances would be the most soluble in CCl4? C6H14 Li2O HBr…
A: The question is asking us to determine which of the given substances would be most soluble in carbon…
Q: OH This is another synthesis problem. Show reagents and intermediates synthesized along the way that…
A: Step 1:Step 2:Step 3:Step 4: Here, due to the presence of strongly activating -OH group; Cl atom…
Q: Add the reagents and conditions above and below the arrow that complete this proposed acetoacetic…
A:
Q: A galvanic cell is powered by the following redox reaction: O2(g) + 4H(aq) + 2 Zn(s) 2+ 2H2O(l) +…
A: Step 1 : Reduction involves gain of electrons.Oxidation involves loss of electrons. Step 2:In…
Q: 16
A: I tried my best to make you understand but still if any further doubt regarding the mechanism of the…
Q: 1. What compounds are in the following? Sodium bicarbonate Baking powder Baker's ammonia 2. What is…
A: What compounds are in the following?Sodium bicarbonate: It is composed of sodium ions (Na+) and…
Q: The standard electrode potential (E) for the half-reaction Ni2+(aq) + 2e → Ni(s) is -0.257 V. What…
A: Step 1:In a galvanic cell, oxidation always takes place at anode and reduction always takes place at…
Q: ANSWER IN kJ!!!!!! Careful with sig figs
A: Step 1:Step 2:
Q: Chemistry
A: The freezing points of the given substances are as follows: H₂O (Water): The freezing point is…
Q: The solubility of CO2 in h2o is very low in air (1.05e-5 M at 25 C) because the partial pressure of…
A: The objective of this question is to find the partial pressure of CO2 required to dissolve a given…
Q: Draw a structural formula for the major organic product(s) of the reaction shown below. 1. CH3l…
A: Step 1: Step 2: Step 3: Step 4:
Q: dont provide handwriting solutio......
A: The objective of the question is to draw the structure of two repeating monomer units of poly(methyl…
Q: None
A: Step 1:R and S configurations:It is a nomenclature system in organic chemistry that helps in…
Q: An automobile tire is inflated to a pressure of 29.0 psi. Express this pressure in atmospheres,…
A: The objective of the question is to convert the pressure of an automobile tire from psi (pounds per…
Q: Unit 4: Chemical Manson Precipitation Stoichiometry PL 1. When solutions of lead(II) nitrate and…
A:
Q: Can you provide a mechanism for this reaction?
A: I have assumed the bigger group as (S) so that I could explain mechanism easily but still you have…
Q: 2) Predict the product of the following reaction and provide a step-by-step mechanism for this…
A: The given reaction involves the substitution of a bromine atom with a carboxylate anion. This…
Q: The air pressure is approximately 0.35 atm at an altitude of 3.0 km. Most airplane cabins are…
A: The objective of this question is to understand the relationship between pressure and volume of a…
Q: Give Product Na OE t E+ OH
A: In organic chemistry, a carbonyl-containing compound undergoes condensation reaction in which one…
Q: A triprotic acid (H3A) has pKA values of 3.08 (pKa1), 5.39 (pKa2), and 7.70 (pKa3). Which of the…
A: Therefore when the pH of a solution containing the conjugate pair is equal to the "pKa" of the weak…
Q: Don't provide hand writing solution
A: The objective of the question is to find the light intensity at a certain point on the screen when…
Q: Draw two major enantiomers formed in this reaction. Use a dash or wedge bond to indicate the…
A: Step 1:
Q: How much 0.2 M KCl solution can be made by diluting 1 L of 5 M KCl?
A: The objective of this question is to find out the volume of 0.2 M KCl solution that can be made by…
Q: 1. How many milliliters of solute are needed to make 840. mL of a 16.5% (v/v) acetic acid solution?…
A: 1. To make a 16.5% (v/v) acetic acid solution, the volume of solute (acetic acid) needed can be…
Q: Please don't provide handwritten solution ...
A: If you can describe the image or provide more specific details, I can offer further assistance in…
Q: None
A: Polarity:There are two types of compounds and these compounds are ionic compounds and covalent…
Q: None
A: Approach to solving the question:Please see attached photos for detailed solutions. Thank you.…
Q: Predict the potential during a potentiometric redox titration of Fe2+ (analyte) with Ce4+ (titrant)…
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw the structure(s) of the major organic product(s) of the following reaction. + 1. in benzene 2.…
A: The product can undergor further hydrolysis to give the acid.
Q: 1.46 mol sample of nitrogen gas at a temperature of 13.0 °C is found to occupy a volume of 30.0…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Step by step
Solved in 2 steps

- Draw two major products of this reaction. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers. Ignore inorganic byproducts.Name the alkene below.Use only E/Z designators to indicate stereochemistry.Can someone please explain the logic of 2 vs 3 for stereochemistry? They are both C-C so I don't understand how to rank 2 vs 3. Thank you!
- The 1,2‑dibromide is synthesized from an alkene starting material. Draw the alkene starting material. Clearly, show stereochemistry of the alkene.a.Draw three-dimensional representations for all stereoisomers of 2chloro-3-methylpentane, and label pairs of enantiomers. b. Considering dehydrohalogenation across only C2 and C3, draw the E2 product that results from each of these alkyl halides. How many different products have you drawn? c. How are these products related to each other?Consider a reaction where cis-but-2-ene is treated with sO followed by NaHSO/H, O. Draw the structure of one product that is formed in the reaction, including correct stereochemistry.
- A hydrocarbon of unknown structure has the formula C8H10. On catalytichydrogenation over the Lindlar catalyst, 1 equivalent of H2 is absorbed. Onhydrogenation over a palladium catalyst, 3 equivalents of H2 are absorbed.(a) How rnany degrees of unsaturation are present in the unknown?(b) How many triple bonds are present?(c) How many double bonds are present?(d) How many rings ar e present?(e) Draw a structure that fits the data.Which carbocation is more stable? CH3CH2+ or H2C=CH+||| IV Identify the least stable carbocation. A) I B) II C) III D) IV
- Draw the products of this reaction. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers. Ignore inorganic byproducts.i. Fill in the missing starting materials, products, or reagents as necessary. ii. Label each transformation as SN1, SN2, or acid/base. iii. Indicate if the product is racemic or a single enantiomerDraw the product of an SN2 reaction shown below. Use the dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers. Ignore inorganic byproducts.

