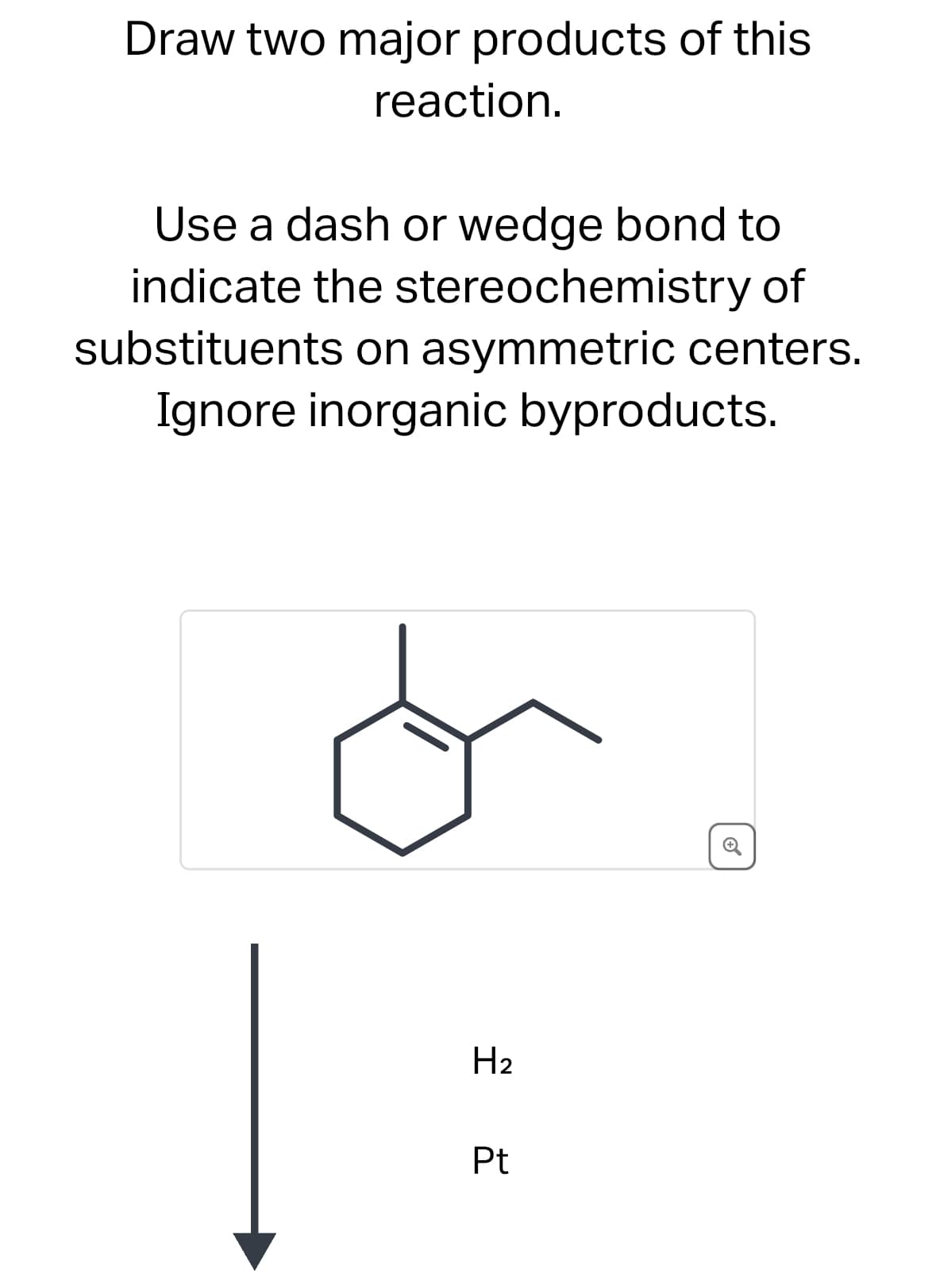
Draw two major products of this reaction. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers. Ignore inorganic byproducts. H2 Pt ⑤
Q: A patient ingests 1.42 μg�g of 131I (iodine-131), a beta emitter with a half-life of 8.0 days.…
A: Convert the initial activity from micrograms (μg) to disintegrations per second (dps):We know that 1…
Q: Draw the product of the reaction shown below. Use a dash or wedge bond to indicate the…
A:
Q: 5. What is produced at the cathode when 1.0 M NaCl is electrolyzed (TChloride Anomaly)? (A) Cl2(g)…
A: Step 1: In the electrolysis of 1.0 M NaCl solution, sodium metal (Na) can be produced at cathode.…
Q: None
A: Generally P exhibits valence 3, sulfur and oxygen exhibit valence 2. Among the given structures…
Q: None
A: Here's a breakdown of why each option is correct or incorrect:Option (a):- The hydroxyl group on the…
Q: Chemistry
A: The freezing points of the given substances are as follows: H₂O (Water): The freezing point is…
Q: A concentration cell was made using a Ba metal electrode in a saturated solution of BaCO3 for the…
A: Step 1The cell reaction is as follows. Anode: Ba(s)→Baaq2++2e−Cathode: Baaq2++2e−→BasApplying…
Q: Experiment shows that the rate formation of carbon tetrachloride from chloroform, CH3 +Cl2 → CCl4 +…
A: Step 1:To determine if the proposed mechanism is consistent with the observed rate law, let's…
Q: Please please please answer everything super super fast
A: Step 1:.
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. conc. HBr Drawing
A:
Q: Calculate the density of oxygen gas (in g/L) at 477 mm Hg and 45.8 °C. g/L
A: The objective of this question is to calculate the density of oxygen gas under given conditions of…
Q: 1. CN Br 2. LiAlH4 3. H₂O NH2 2a Primary amines can be prepared from nitriles by reduction with…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Don't use extra explanation solve correct
A:
Q: The reagents used for this reaction are H2SO4, HNO3, but what is the mechanism?
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw one of the two stereoisomers of the major product from this reaction. Use a dash or wedge bond…
A:
Q: A sample of argon gas occupies a volume of 6.57 L at 53.0°C and 0.880 atm. If it is desired to…
A: The objective of this question is to find the temperature of a sample of argon gas when its volume…
Q: What is the mechanism and reagents for this reaction? with the circled molecule being attacked first…
A: Step 2: Step 3: Step 4:
Q: A 0.175 mol sample of N2 gas is contained in a 4.00 L flask at room temperature and pressure. What…
A: The objective of this question is to calculate the density of nitrogen gas under given conditions.…
Q: Determine K for a reaction at 200 K if ∆G° =20.4 kJ/mol. (R = 8.314 J/mol ・ K)
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: The standard electrode potential (E) for the half-reaction Ni2+(aq) + 2e → Ni(s) is -0.257 V. What…
A: Step 1:In a galvanic cell, oxidation always takes place at anode and reduction always takes place at…
Q: dont provide hnadwriting solutio .....
A: The formation of 1-(cyclopent-1-en-1-yl)cyclopent-1-ene is favored over the reformation of…
Q: Sagar
A: 1. **Arrhenius Equation**: The Arrhenius equation relates the rate constant \( k \) of a reaction…
Q: Br 1. N3 2. LiAlH4 3. H₂O NH₂ 26 Preparation of primary amines using azide ion avoids the problem of…
A: Step 1:Step 2:Step 3:Step 4:
Q: Which reaction will occur if each substance is in its standard state? Assume potentials are given in…
A: Approach to solving the question: I believe that this is the complete question together with its…
Q: Calculate and balance of equations that show how the substance dissolves in water.
A: The objective of the question is to balance the equations that show how the given substances…
Q: Draw the starting structure that would lead to the major product shown under the provided…
A:
Q: Please help me solve them, thumbs up will be given. thank u
A: Q4. Cross Claisen condensation reaction Q5. The mechanism followed is Claisen and Aldol condensation…
Q: Draw estrogen and testosterone. Number the positions of the core steroid nucleus
A: Estrogen: The core steroid nucleus of estrogen is comprised of four fused rings labeled A, B, C, and…
Q: A mixture of 2 mL of 3 M NaOH solution, 2.5 mL 95% ethanol, 0.212 g benzaldehyde, and 0.058 g…
A: The reaction is the Aldol condensation, 2C6H5CHO+CH3COCH3→C6H5CH=CHC(O)CH=C6H5+2H2Owhere…
Q: 3+ 2+ For the reaction 2Ga (aq) + 3Zn(s) → 2Ga(s) + 3Zn (aq)E° = 0.21V Determine the 3+ value of E °…
A: To determine the reduction potential (E°red) for the Ga3+/Ga half-cell from the given overall…
Q: Draw the product of this reaction. Ignore inorganic byproducts. Br2 (1 equiv) Drawing Q
A:
Q: 5 ml of a 0.2 M NH3 solution are added to 5 ml of a 0.1 M NH4Cl solution (Kb for NH3 is = 1.8 ×…
A: Step 1:Solution of weak base (NH3) and its conjugate acid (NH4Cl) is a basic buffer.Step…
Q: The wavelength is w = c,V (w) = 3 1 C2 C W C The wavelength is w=c W kw C -4/2 C v') = {(+9)*(-S)…
A: Given w=cWe have to solve V!(w) for w=c,Given,V∣(w)=(k/2)∗((w/c)+(c/w))−1/2∗(1/c−c/w2)putting w=c…
Q: Draw a detailed mechanism, using curved arrow notation,for the synthesis of 3-carboxy-umbeliferone…
A: Step 1: Step 2: Step 3: Step 4:
Q: Explain why a solution of ammonium nitrate is a weak acid. Give a balanced rection equation…
A: The objective of the question is to explain why a solution of ammonium nitrate behaves as a weak…
Q: 1) following reactions. Give the major organic product or missing starting material for the a) 1.…
A: Step 1: Step 2: Step 3: Step 4:
Q: According to this reaction energy diagram, the value of the change in energy is. Potential energy…
A: Step 1: Identify the parts of the diagram Point A in the given diagram represents the reactants and…
Q: please help
A: The objective of this question is to write a balanced chemical equation for the reaction between…
Q: Propane reacts with oxygen to produce carbon dioxide and water according to the following equation.…
A: Step 1:
Q: Chemistry
A: Step 1: Step 2: Step 3:
Q: Calculate the mass (in g) of SiO2 that reacts completely with 15.0 grams of carbon according to the…
A: Given mass of carbon= 15.0 gMolecular mass of carbon = 12.0 g/molNumber of moles of carbon= given…
Q: The following mechanism was proposed for the reaction with overall stoichiometry A + B → R A → X X+…
A: Step 1: The mechanism given for the reaction is A↔X X+B→R The intermediate X can be…
Q: A sample of oxygen gas occupies a volume of 8.38 L at 56.0°C and 344 torr. If the volume of the gas…
A: The objective of the question is to find the resulting gas pressure when the volume and temperature…
Q: 5. Calculate the pH of 1.5 M solution of hydroxylamine, NH₂OH at 25°C. K = 9.1 x 10-9 HONH2(aq) +…
A: The objective of the question is to calculate the pH of a 1.5 M solution of hydroxylamine, NH2OH at…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. • • 1. in…
A: First one is the product. But due to the presence of acid, the ester undergoes hydrolysis to give…
Q: Calculate AGO for each of the following reactions from the equilibrium constant at the temperature…
A: (a)(b)(c)(d)(e)(f)ReactionsN2(g)+O2(g)→2NO(g)H2(g)+I2(g)→HI(g)CO2(g)+H2(g)→CO(g)+H2O(g)C…
Q: Explain the difference in pH between diH2O and tap water
A: The objective of this question is to understand the difference in pH between distilled water and tap…
Q: Determine the person by mass of a solution prepared by dissolving 45.8 g NaCIO4 in 275 g of water.
A: The objective of this question is to calculate the percent by mass of a solution prepared by…
Q: Choose the TRUE statement about the effect of the presence of a catalyst on reaction rate. In the…
A: The objective of the question is to identify the correct statement about the effect of a catalyst on…
Q: A gas had a solubility of 0.2 g/L at 2.5 atm. What is the pressure of a 1.0 L sample that contains…
A: The objective of the question is to find the pressure of a gas sample given its solubility at a…
Step by step
Solved in 2 steps with 1 images

- Draw the products of this reaction. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers. Ignore inorganic byproducts.Draw the possible products of this epoxied ring opening reaction. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, Ignore any inorganic byproducts.a. With attention to stereochemistry, indicate the product of the SN2 reaction of cis-1-bromo-2methylcyclohexane. b. Draw and identify (R) and (S) enantiomers of lactic acid, CH3CH(OH)COOH. Use the proper conventions to draw Fischer projections.
- draw one of the two enantiomers of the major product from this reaction. use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers. ignore inorganic byproductsDraw the products of this halogenation reaction. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers. Ignore inorganic byproducts.Can someone please explain the logic of 2 vs 3 for stereochemistry? They are both C-C so I don't understand how to rank 2 vs 3. Thank you!
- 4-Chloro-2-pentene has a double bond that can have either the E or the Z configuration and a stereogenic center that can have either the R or the S configuration. How many stereoisomers arepossible altogether? Draw the structure of each, and group the pairs of enantiomers.The substitution product of (S)-3-chloro-3-phenyl-heptane in methanol will be _______________. a racemix mix the (S)-enantiomer the (R)-enantiomer not chiralDraw the product of an SN2 reaction shown below. Use the dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers. Ignore inorganic byproducts.
- Consider the E2 elimination of Compound A, and answer the following questions: (a) Label each stereocenter in Compound A with the correct R or S configuration. (b) Draw the structures of the major product with correct stereochemistry. Assume D and H have the same reactivity.Consider a reaction where cis-but-2-ene is treated with OsO4 followed by NaHSO3/H2O. Draw the structure of one product that is formed in the reaction, including correct stereochemistry.Draw the structures for (Z)-3-methylhex-3-ene and the two major organic products for its reaction with HBr. Be sure to show all stereoisomers using wedge and dash stereochemistry at any chirality center.

