Each sketch below shows three objects with an electric charge. In each case, decide whether there is a net force acting on the object outlined in green. If there is a net force, decide whether it pushes the green-ring object to the left or right. Then select the appropriate button under the sketch. For example, if there is a net force pushing the green-ring object in the first sketch to the left, select the left button under the first sketch. If there is no net force on the green-ring object in the second sketch, select the middle button under the second sketch. And so on. -3 +2 - O O O Explanation Check Privacy Ace 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use
Electronic Effects
The effect of electrons that are located in the chemical bonds within the atoms of the molecule is termed an electronic effect. The electronic effect is also explained as the effect through which the reactivity of the compound in one portion is controlled by the electron repulsion or attraction producing in another portion of the molecule.
Drawing Resonance Forms
In organic chemistry, resonance may be a mental exercise that illustrates the delocalization of electrons inside molecules within the valence bond theory of octet bonding. It entails creating several Lewis structures that, when combined, reflect the molecule's entire electronic structure. One Lewis diagram cannot explain the bonding (lone pair, double bond, octet) elaborately. A hybrid describes a combination of possible resonance structures that represents the entire delocalization of electrons within the molecule.
Using Molecular Structure To Predict Equilibrium
Equilibrium does not always imply an equal presence of reactants and products. This signifies that the reaction reaches a point when reactant and product quantities remain constant as the rate of forward and backward reaction is the same. Molecular structures of various compounds can help in predicting equilibrium.
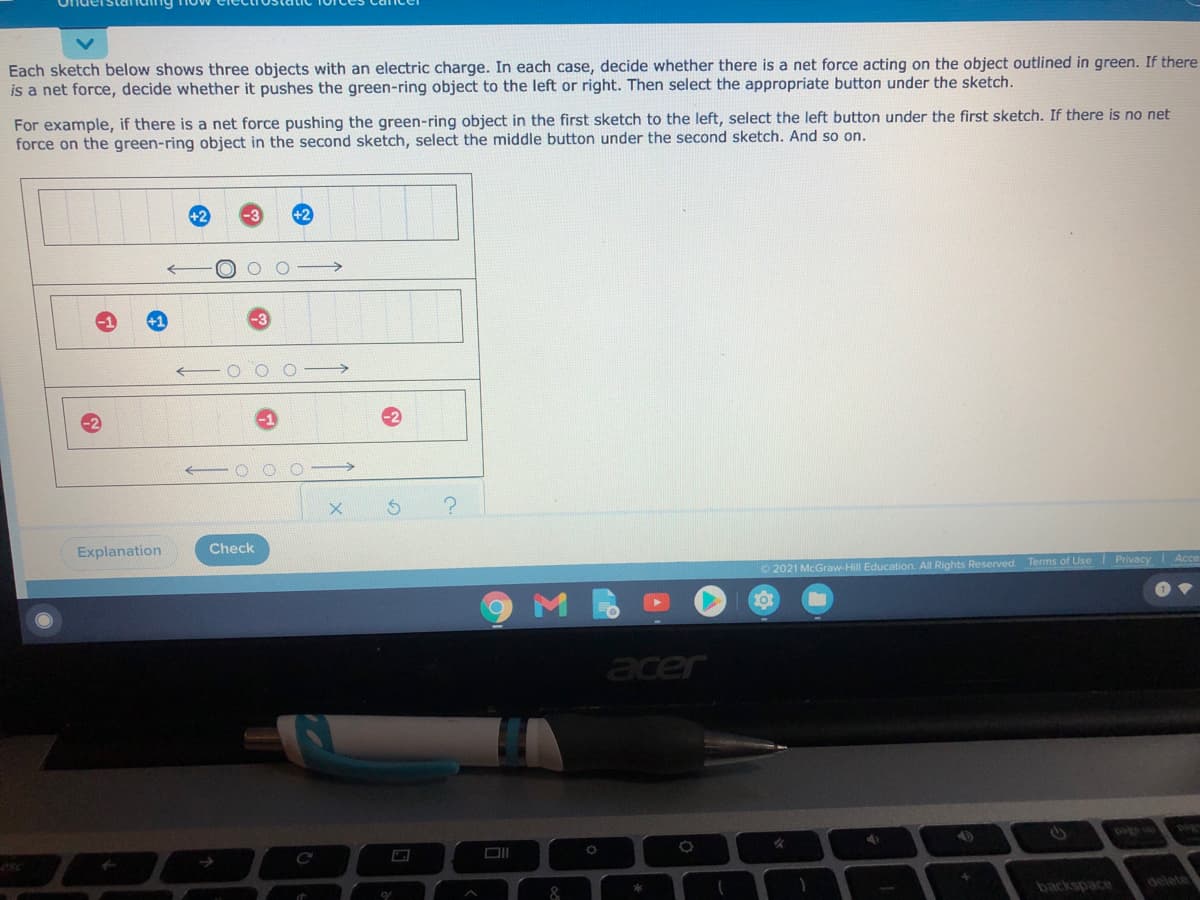
As we know, according to Coloumb's law of force of attraction, two oppositively charges attracted each other when they are near to each other while similiar charges repel each other when they are near to each other.
Similarly when charges on ion are high then force of attraction is more and vice versa.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps









