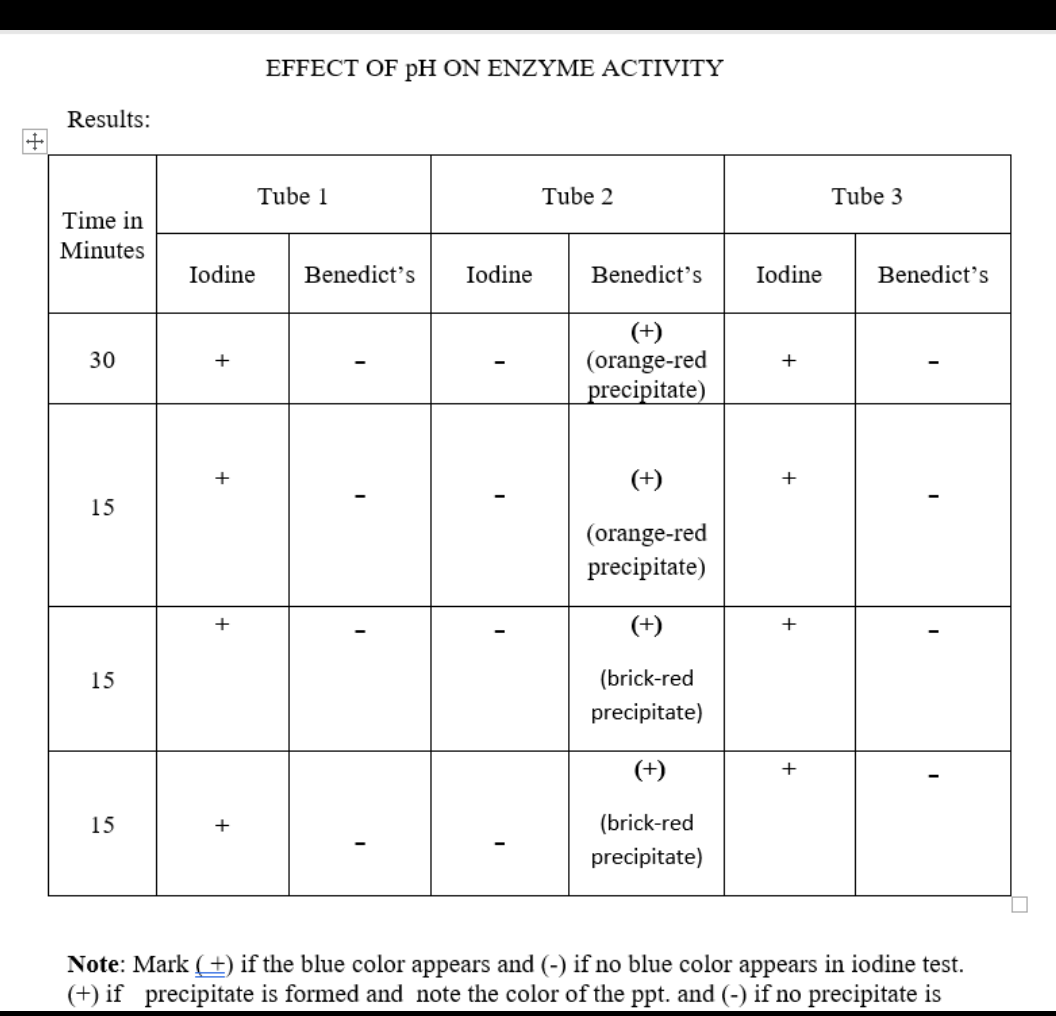
EFFECT OF pH ON ENZYME ACTIVITY Results: Tube 1 Tube 2 Tube 3 Time in Minutes Iodine Benedict's Iodine Benedict's Iodine Benedict's (+) (orange-red precipitate) 30 + (+) + 15 (orange-red precipitate) + (+) + 15 (brick-red precipitate) (+) + 15 + (brick-red precipitate) Note: Mark (+) if the blue color appears and (-) if no blue color appears in iodine test. (+) if precipitate is formed and note the color of the ppt. and (-) if no precipitate is
Q: running the reaction at 83 °C cooling the reaction to 11 °C changing the pH to 5.4 Increase reaction…
A: The rate of an enzyme catalyzed reaction depends on various factors like temperature and pH of the…
Q: You have obtained experimental kinetic data for two versions of the same enzyme, a wild-type and a…
A: The Km Michaelis constant (or equilibrium constant of the dissociation of the E-S complex) is…
Q: Which of the following is TRUE concerning the induced fit model of enzyme catalysis? * (One correct…
A: Induced fit model: this theory explains that the active site of an enzyme is so flexible that…
Q: The rate of an enzyme- catalyzed reaction increases as the temperature is raised beyond optimum…
A:
Q: Which enzyme would have the greatest catalytic efficiency? a) Km 20 nM; kcat 50 s-1 b) Km 0.04 uM;…
A: [S] is Substrate concentration V is velocity of reaction Kcat - Catalytic constant(Turnover…
Q: Why must you only add the enzyme after all the other reagents have been added? Because until all…
A: Enzyme - Have optimal pH and temperature at which they function best. Reaction conditions are…
Q: When enzyme solutions are heated, there is a progressive loss of catalytic activity over time due to…
A: Enzyme is a protein and its denatured at high temperature. High temperature broke its peptide bond.…
Q: Consider the following data set of enzymes A, BC and D which catalyze the same reaction. Enzyme A km…
A: Introduction: Catalysts are those substances that increase the rate of the reaction without…
Q: [S] mM 20 50 100 200 300 500 700 Initial Velocity (mM/min) 1.012 1.950 2.823 3.637 4.024 4.398 4.580
A: Enzyme kinetics can be determined by constructing mm plot or lb plot. With them km and Vmax can be…
Q: What general effects would you expect the following changes to have on the rate of an…
A: Enzyme catalysis is the increase in the rate of reaction by a biological molecule known as enzyme.…
Q: The initial rate for an enzyme-catalyzed reaction has been determined at a number of substrate…
A: Enzyme catalyzed reaction is any chemical or biochemical reaction which involves biological molecule…
Q: Which of the following statements regarding enzyme catalysis is false? All options are false. Once…
A: Biochemical reaction tend to be slow, unlikely or unfavorable in the chemical environment is the…
Q: Students conducting research observe the rate of an enzyme-catalyzed reaction under various…
A: Enzymes are the biological catalysts that affect the rate of reaction. In most cases, enzymes are…
Q: Table 2: Effect of pH on Enzyme Activity pH Absorbance 2 0.05 4 0.35 6 0.8…
A: The point where the enzyme is most active is called the optimum pH. At higher or lower pH, enzyme…
Q: The following experimental data were obtained for the kinetics of the enzyme reaction Glucose + ATP…
A: Enzyme kinetics are explained by Michaelis Menten equation shown below: V=VmaxSKm+S Here, V is…
Q: As an intermediate step in making the enzyme solution, you need to prepare Buffer X using the stock…
A: Calculations play an important role in taking the right concentration of solutes. They help to…
Q: Use the data below to determine the maximum velocity [in mM/s] of a certain enzyme-catalyzed…
A: We must know the Michaelis Menten equation: V=Vmax [S]Km+ [S]
Q: Two substrates (A and B) are used to produce a certain biological product in an enzyme- catalyzed…
A: The substance which helps in speeding up a chemical reaction without being involved in a reaction is…
Q: Draw the Michaelis-Menten Plot and Lineweaver-Burke Plot of an enzyme in the following situation.…
A: Enzymes are biocatalysts which increase the rate of biochemical reactions. Enzymes will not…
Q: What is the biological purpose of enzyme regulation, i.e., why is it necessary to regulate enzyme…
A: Introduction: Enzymes are biological catalysts that are synthesized by the living cells of the body.…
Q: Enzyme Activity vs. Drops of Enzyme 0.030 0 025 0.020 0 015 0.010 0.005 0.000 10 Enzyme (drops) 15…
A: The rate of a chemical reaction increases when the substrate concentration increases. Enzymes speed…
Q: iven the following information, calculate the catalytic efficiency of the enzyme. Step by step…
A: The substrate binds to the enzyme's active site and is converted to the product. Multiple…
Q: Consider an enzyme that uses general acid-base catalysis such that the reverse protonated form (call…
A: pKa values denote the deprotonation of a ionizable group. When pH value is below the pKa of…
Q: In the time course experiment, you will perform the incubations of 5 mL of the reaction mixtures of…
A: Add the 5ml of reaction mixture of 3 different enzymes in tripilicate in 10 ml test tubes. Add 5 ml…
Q: Which of the following statements regarding size exclusion chromatography is false? During…
A: Hi! Thank you for the question, as per the honor code, we are allowed to answer the first…
Q: kąk4 k2k1 E +S → ES → E + P (Vmax,f\ rax.f (Vmax,b [P] KM.f KM.b [S] [P] Vo 1+ KM.f Кмь KM,b ||
A: In this enzyme kinetics, I am going assume a general mechanism and then derive the rate equation. As…
Q: If you multiplied enzyme concentration by kcat, think about what you would get. Which one of the…
A: The study of the in-vitro and in-vivo chemical reactions which occur in the biological system and…
Q: Draw a hypothetical Michaelis-Menten plot for an enzyme reaction (i) without and (ii) with a pure…
A: Enzymes are the class of proteins that elevates the rate of the chemical reaction within the living…
Q: Vo with Vo without [Substrate, µM] DEDS DEDS (µM/min) (µM/min) 3.333 0.774 1.196
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation energy…
Q: The effect of temperature on enzyme activity. Temperature °C 10 Reaction rate 1.0 15 1.5 20 2.5 25…
A: Enzymes are biomolecules that act as a catalyst to speed up and facilitate many biochemical…
Q: Several factors contribute to enzyme catalysis. What arethey? Briefly explain the effect of each.
A: Enzymes are proteins that act as biocatalysts in speeding up biochemical reactions without actually…
Q: For some Enzyme, the Vmax is 18 micromols/min, Km is 400 microM. If the substrate concentration is…
A: Velocity of the reaction defines the rate of reaction which is catalyzed by the enzyme. It depends…
Q: Given the Km and Vmax values of three enzymes, A, B and C: Enzyme Km Vmax A 2.0 x 103 M 4.0 B 4.2 x…
A: Enzymes are proteins that assist the biochemical reactions. Enzymes are sensitive to temperature,…
Q: When enzyme solutions are heated, there is a progessive loss of catalytic activty over time due to…
A: Enzyme is defined as a biological catalyst that speeds up the rate of chemical reaction. All…
Q: By using Excel or GoogleSheets, graph the Lineweaver-Burk plots for the behavior of an enzyme for…
A: An enzyme is a catalytic molecule that increases the rate of any chemical reaction without being…
Q: Which of the following statements regarding enzyme catalysis is false? All options are false. Once…
A: Normally an energy barrier exists between substrates (S) and products (P) i.e., S -> P…
Q: The kinetic data for the activity of 0.01mM enzyme is shown below. The concentration of {S} is…
A:
Q: Why is the enzyme added last? Why can’t it be added at the start and the rates measured sometime…
A: For an enzyme catalysed reaction, the graph of the initial rate of reaction vs substrate…
Q: An enzyme following Michaelis-Menten kinetics was found to have highest activity at 37° C and pH 7.…
A: The Michaelis-Menten equation is model of enzyme kinetics. It is mainly used to characterize the…
Q: 1/V, min/umol 5.50 5.00 4.50 4.00 3.50 answer. 3.00 2.50 2.00 0.00 y = 0.9474x + 2.6649 0.50 1.00…
A: Double reciprocal plot is also called as lb plot that is drawn by taking inverse of Substrate…
Q: Use the plot (below) to estimate values of KM and Vmax for an enzyme-catalyzed reaction. Use 1…
A: Michaelis Menten (MM) kinetics was developed by Canadian physician Maud Menten and German biochemist…
Q: Which of the listed effects would be brought about by any enzyme catalyzing the following simple…
A: Introduction: Those compound that increases the rate of the reaction without undergoing any change…
Q: When enzyme solutions are heated, there is a progressive loss of catalytic activity over time due to…
A: Whenever any specific substrate comes in contact with its enzyme, it will go and bind to the active…
Q: From the Lineweaver-Burke plot of an enzyme-catalyzed reaction containing 4 μM total enzyme, you…
A: Hi. Thank you for the question. As per the honor code, We'll answer the first question since the…
Q: What is the percent change for the enzyme active at 40 C compared to 25 C* 220 200 180 160 e…
A: Enzymes are proteins that act as a catalyst to speed up chemical reactions. A catalyst is a…
Q: A plot of the reaction rate, V, vs. the substrate concentration for an enzyme, alone and in the…
A: Hi! Thanks for your question. As you have posted multiple questions and have not mentioned which one…
Q: Consider the following free energy diagram for an uncatalyzed and enzyme-catalyzed reaction. Select…
A: The process associated with the formation of bonds as well as conversion of a reactant into another…
Q: n enzyme-catalyzed reaction has a change in free energy equal to -20 kcal/mol. If ou double the…
A: Free energy determines whether a conversion of reactants to products will occur spontaneously or…
Q: Use the Michaelis-Menten equation to complete the enzyme kinetic data set, when Km is known to have…
A: Introduction: The theory of enzyme kinetics was proposed by two scientists Leonor Michaelis and…
Q: Allosteric activators block the active site, so the enzyme cannot bind True Or False
A: Enzymes are proteins in nature, that are capable of altering rate of chemical reactions taking…
Why is it the answer in number 1 are test tubes 1 and 3

Step by step
Solved in 2 steps

- Studies at diff erent pH’s show that an enzyme has two catalytically important residues whose pKs are ∼4 and ∼10. Chemical modifi cation experiments indicate that a Glu and a Lys residue are essential for activity. Match the residues to their pKs and explain whether they are likely to act as acid or base catalysts.A histidine side chain is known to play an import-ant role in the catalytic mechanism of an enzyme; how-ever, it is not clear whether histidine is required in its pro-tonated (charged) or unprotonated (uncharged) state. Toanswer this question you measure enzyme activity over arange of pH, with the results shown in Figure Q2–1. Whichform of histidine is required for enzyme activity?Utilising the provided class data generate the following graphs: I) Michaelis Menten; II) Lineweaver-Burk; and III) Hanes-Woolf. Ensure that you clearly label each graph,and add the relevant trendlines with equations. Table 1: Class data demonstrating the Absorbance at 700nm obtained for the alkaline phosphatase enzyme reaction Table 1 tube Abs700mm 1 0.000 2 0.060 2 0.090 4 0.140 5 0.190 6 0.250 7 0.290 The equipment we used are • 20mM Tris Buffer pH 8.5 • 33mM MgCl2 • Alkaline Phosphatase (2mg/ml) in 20mM Tris Buffer pH 8.5 • 4mM Glucose-1-phosphate • Acid Molybdate pH 5.0 • Reducing Agent • Distilled Water • Glass Test tubes • Tube Rack • Cuvette • Pipettes and Tips • Water bath set to 37oC The method we used is Method/Protocol: 1. Read the protocol in its entirety before starting. Take note of any additional information that appears in subsequent steps that may influence how previous steps are performed. 2. Using glass tubes, generate the reactions mixtures…
- Sodium fluoroacetate (FH2CCOO- Na+) is highly toxic. Patients with fluoroacetate poisoning accumulate citrate and fluorocitrate in their cells. Which enzyme is inhibited by fluoroacetate for this to occur? Explain.The Michaelis‑Menten equation models the hyperbolic relationship between [S] and the initial reaction rate ?0V0 for an enzyme‑catalyzed, single‑substrate reaction E+S↽−−⇀ES⟶E+PE+S↽−−⇀ES⟶E+P. The model can be more readily understood when comparing three conditions: [S]<<?m[S]<<Km, [S]=?m[S]=Km, and [S]>>?m[S]>>Km. Match each statement with the condition that it describes. Note that "rate" refers to initial velocity ?0V0 where steady state conditions are assumed. [Etotal][Etotal] refers to the total enzyme concentration and [Efree][Efree] refers to the concentration of free enzyme.Protein: QQICIMFELTQISS Predict the products of the following reactions with the protein given, if there is none, write NO RXN. Also indicate, if the reaction is fast or slow.
- Many enzymes obey simple Michaelis–Mentenkinetics, which are summarized by the equationrate = Vmax [S]/([S] + Km)where Vmax = maximum velocity, [S] = concentration ofsubstrate, and Km = the Michaelis constant.It is instructive to plug a few values of [S] into theequation to see how rate is affected. What are the rates for[S] equal to zero, equal to Km, and equal to infinite concen-tration?The activity of an enzyme requires a glutamic acid to display its -COOHfunctional group in the protonated state. Suppose the pKa of the -COOHgroup is 4.07.(a) Will the enzyme be more active at pH 3.5 or 4.5? Explain.(b) What fraction of the enzymes will be active at pH = 4.07? Explain.(c) At what pH will the enzyme show 78% of maximal activity?Initial rate data for an enzyme that obeys Michaelis–Menten kinetics areshown in the following table. When the enzyme concentration is 3 nmolml-1, a Lineweaver–Burk plot of this data gives a line with a y-intercept of0.00426 (μmol-1 ml s). (a) Calculate kcat for the reaction.(b) Calculate KM for the enzyme.(c) When the reactions in part (b) are repeated in the presence of 12 μM ofan uncompetitive inhibitor, the y-intercept of the Lineweaver–Burk plotis 0.352 (μmol-1 ml s). Calculate K′I for this inhibitor.
- a) Determine kcat (in units of sec-1) for a particular enzyme, given the following information: Vo = 144 mmol/min; [S] = 2 mM; Km = 0.5 mM; Enzyme Molecular weight = 40,000 mg/mmole; 8 mg of enzyme used in assay generating this data. b) In general, explain how the total enzyme concentration affects turnover number and Vmax?Why is the reaction rate low at pH7? Be specific and say something about the enzyme structure at the molecular level!Trend observed in graph and conclusion about the effect of temperature on enzyme activity. i) include a concise description of the trend observed in the graph shown in question 3 above, and explain this trend using the language presented in this unit and your biochemical knowledge of enzymes and reactions. In your conclusion, provide a logical argument supported by molecular theory that would explain any change observed in enzyme activity.

