Enter an orbital diagram for Ni²+ Drag the appropriate labels to their respective targets. Labels can be used once, more than once, or not at all. Targets may be left blank, such as for unused orbitals. Y esc G1 Part D G2 Submit ! 1 Mother to Son &....pdf A Enter anahital dinnom for Dat FI G1 Request Answer Q G2 A 1 G1 G1 G1 N @ G2 2 ** pods W 1 11 1s 2s 3s 4s S X option command # 3 G1 G2 80 F3 E D G1 GÌ GÌ G2 59. $ 4 C a F4 R 2p F G1 G2 dlo L % 5 3p 4p 3d GÌ GÌ GÌ GÌ GÌ D FS V T G2 MacBook Air A <(( G 6 Y B GÌ GÌ GÌ G2 & 7 H Reset F7 U * 8 N Help J DII FB 1 ( 9 DO M F9 K O O a < F10 L 9.0 36 com
Enter an orbital diagram for Ni²+ Drag the appropriate labels to their respective targets. Labels can be used once, more than once, or not at all. Targets may be left blank, such as for unused orbitals. Y esc G1 Part D G2 Submit ! 1 Mother to Son &....pdf A Enter anahital dinnom for Dat FI G1 Request Answer Q G2 A 1 G1 G1 G1 N @ G2 2 ** pods W 1 11 1s 2s 3s 4s S X option command # 3 G1 G2 80 F3 E D G1 GÌ GÌ G2 59. $ 4 C a F4 R 2p F G1 G2 dlo L % 5 3p 4p 3d GÌ GÌ GÌ GÌ GÌ D FS V T G2 MacBook Air A <(( G 6 Y B GÌ GÌ GÌ G2 & 7 H Reset F7 U * 8 N Help J DII FB 1 ( 9 DO M F9 K O O a < F10 L 9.0 36 com
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter5: Quantum Mechanics And Atomic Structure
Section: Chapter Questions
Problem 14P: Spectroscopic studies of Li also show that Zeff(2p)=1.02 . Estimate the energy of the 2p orbital of...
Related questions
Question
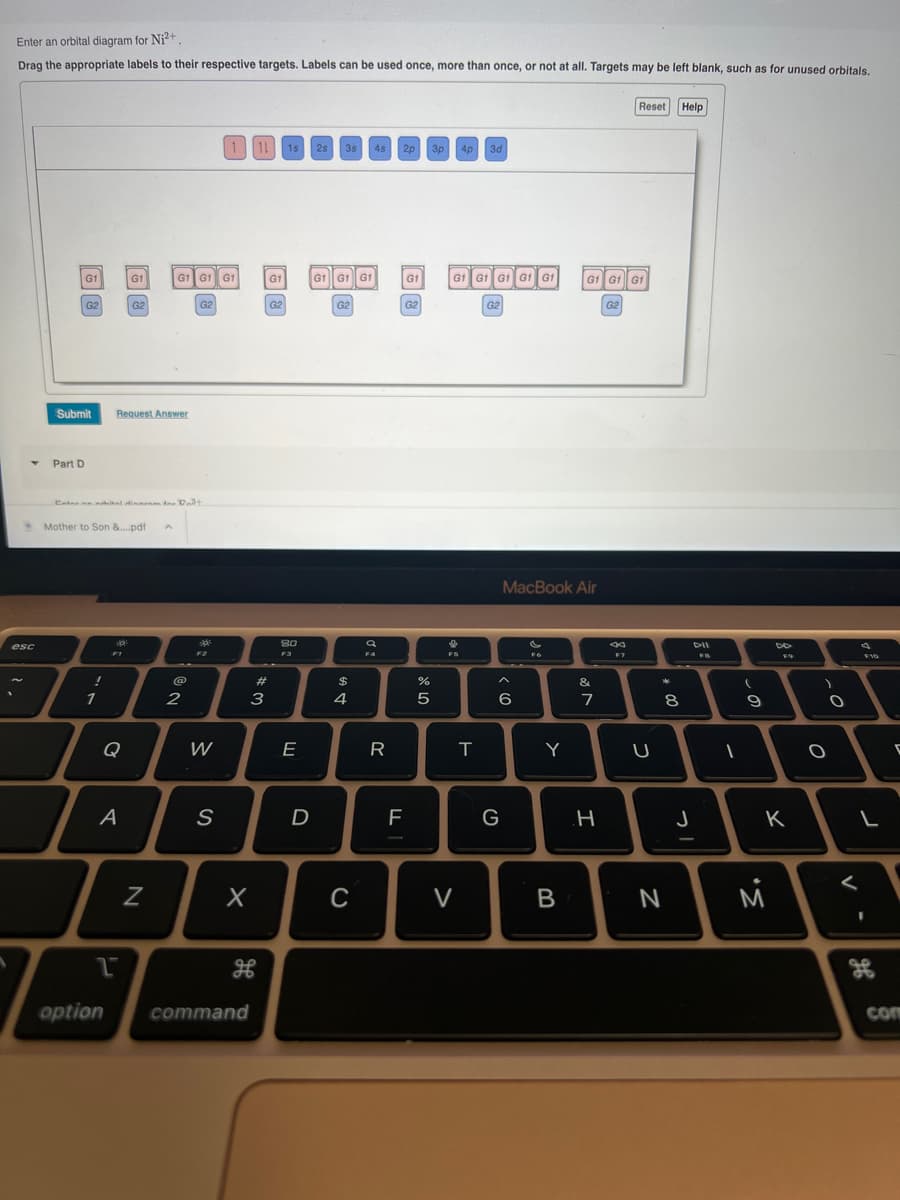
Transcribed Image Text:Enter an orbital diagram for Ni²+
Drag the appropriate labels to their respective targets. Labels can be used once, more than once, or not at all. Targets may be left blank, such as for unused orbitals.
esc
G1
Part D
G2
Submit
!
1
Mother to Son &....pdf
Enter an hital dinnom for Dat
FI
G1
Request Answer
Q
G2
A
1
G1 G161
N
@
G2
2
**
2494
W
S
X
option command
#
3
G1
G2
1s
80
F3
E
D
2s 3s 4s 2p
G1 G1 GÌ
G2
$
4
C
a
F4
R
F
G1
G2
dlo L
%
5
3p 4p 3d
GÌ GÌ GÌ GÌ GÌ
FS
V
T
G2
6
G
MacBook Air
Y
B
GÌ GÌ GÌ
G2
&
7
H
Reset
F7
U
8
N
Help
J
DII
FB
-
1
(
9
DO
K
M
)
O
O
a
F10
L
9.
com
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning