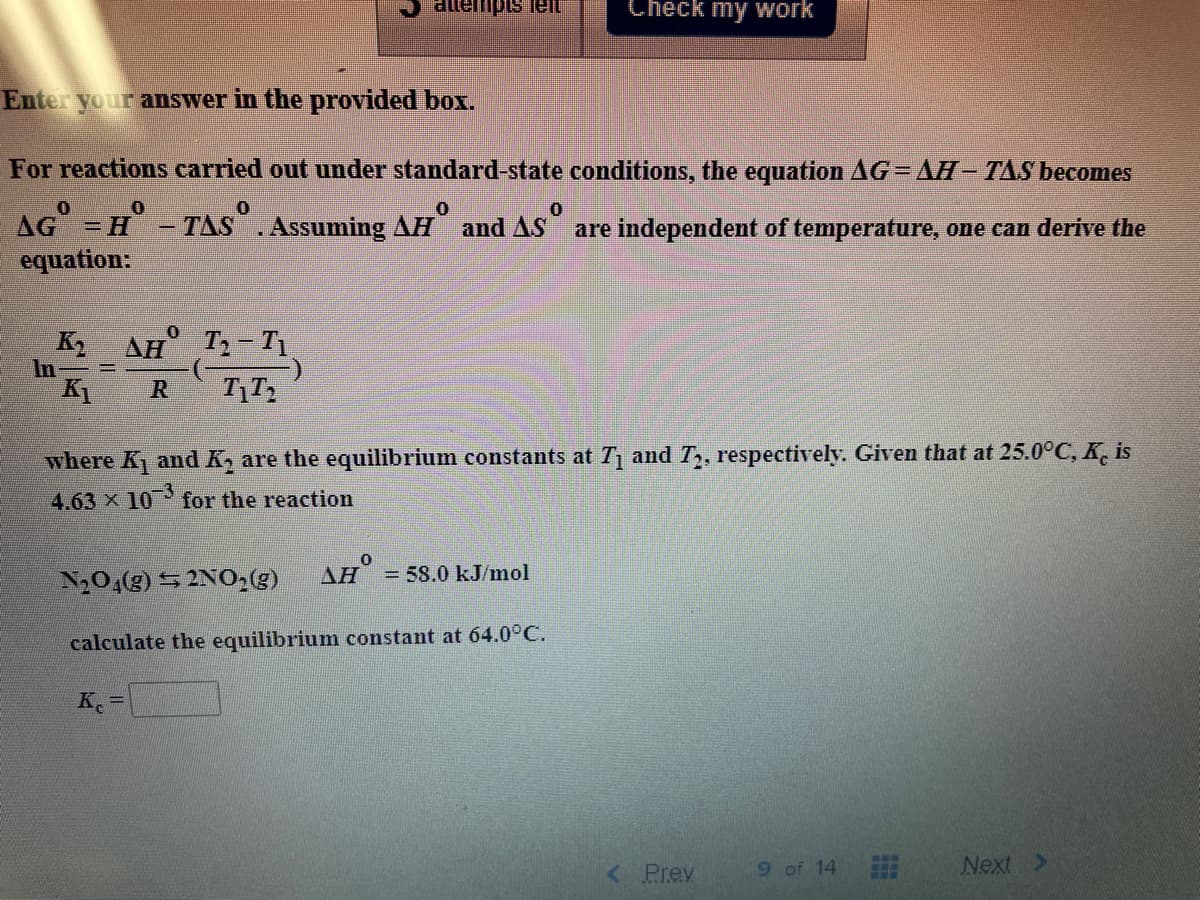
er your answer in the provided box. r reactions carried out under standard-state conditions, the equation AG = AH - TAS b S = H - TAS . Assuming AH and AS are independent of temperature, one can d quation: K AH T- T1. K R where K and K, are the equilibrium constants at T and T, respectively. Given that at 25.00 4.63 x 10 for the reaction N204(g) 5 2NO2) AH = 58.0 kJ/mol calculate the equilibrium constant at 64.0°C. Ke < Prev 9 of 14 Next
er your answer in the provided box. r reactions carried out under standard-state conditions, the equation AG = AH - TAS b S = H - TAS . Assuming AH and AS are independent of temperature, one can d quation: K AH T- T1. K R where K and K, are the equilibrium constants at T and T, respectively. Given that at 25.00 4.63 x 10 for the reaction N204(g) 5 2NO2) AH = 58.0 kJ/mol calculate the equilibrium constant at 64.0°C. Ke < Prev 9 of 14 Next
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 6QRT: Indicate whether each statement below is true or false. If a statement is false, rewrite it to...
Related questions
Question
Transcribed Image Text:tempis left
Check my wwork
Enter your answer in the provided box.
For reactions carried out under standard-state conditions, the equation AG= AH– TAS becomes
AG =H -TAS. Assuming AH and AS are independet of temperature, one can derive the
equation:
AH T- T1
K2
In-
KI
R
where K and K, are the equilibrium constants at T and T, respectively. Given that at 25.0°C, K. is
4.63 x 10 for the reaction
AH = 58.0 kJ/mol
N20,(g) 52NO2(3)
calculate the equilibrium constant at 64.0°C.
< Prev
9 of 14
Next >
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning