Estimate the pH of a 7.18 x 106 MHF solution. You can calculate the lowest possible pH, knowing that the actual value will be higher. Your answer should contain 3 decimal places as this corresponds to 3 significant figures when dealing with logs. pH will be above
Estimate the pH of a 7.18 x 106 MHF solution. You can calculate the lowest possible pH, knowing that the actual value will be higher. Your answer should contain 3 decimal places as this corresponds to 3 significant figures when dealing with logs. pH will be above
Biology (MindTap Course List)
11th Edition
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Chapter2: Atoms And Molecules: The Chemical Basis Of Life
Section2.6: Acids, Bases, And Salts
Problem 1C
Related questions
Question
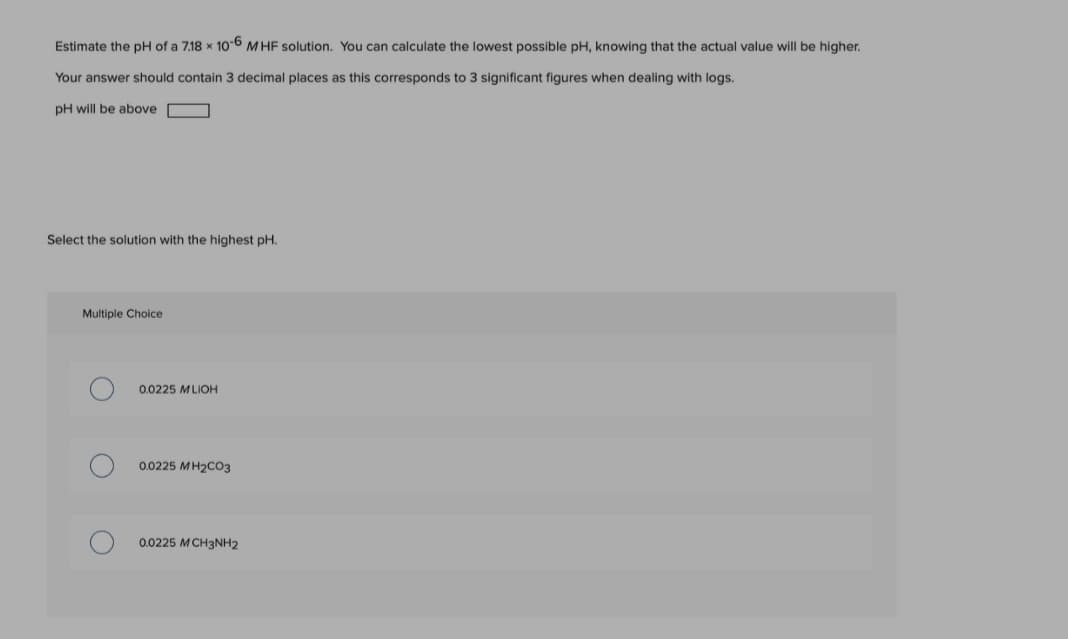
Transcribed Image Text:Estimate the pH of a 7.18 x 106 M HF solution. You can calculate the lowest possible pH, knowing that the actual value will be higher.
Your answer should contain 3 decimal places as this corresponds to 3 significant figures when dealing with logs.
pH will be above
Select the solution with the highest pH.
Multiple Choice
0.0225 MLIOH
0.0225 MH2CO3
0.0225 MCH3NH2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning



Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning




Basic Clinical Lab Competencies for Respiratory C…
Nursing
ISBN:
9781285244662
Author:
White
Publisher:
Cengage
