Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
ChapterA1: Evaluation Of Analytical Data
Section: Chapter Questions
Problem A1.9QAP
Related questions
Question
Asap please
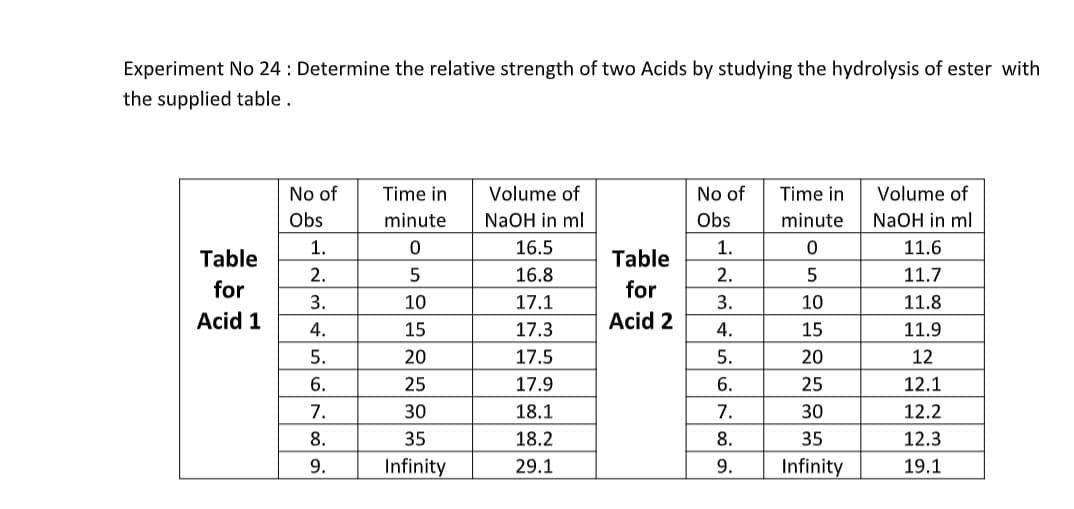
Transcribed Image Text:Experiment No 24: Determine the relative strength of two Acids by studying the hydrolysis of ester with
the supplied table.
No of
Time in
minute
Volume of
NaOH in ml
16.5
No of
Obs
Time in
minute
Volume of
NaOH in ml
Obs
1.
0
1.
0
11.6
Table
for
2.
5
16.8
2.
5
11.7
3.
17.1
3.
10
11.8
Acid 1
4.
17.3
4.
15
11.9
5.
17.5
5.
20
12
6.
17.9
6.
25
12.1
7.
18.1
7.
30
12.2
8.
18.2
8.
35
12.3
9.
29.1
9.
Infinity
19.1
95
10
15
20
25
30
35
Infinity
Table
for
Acid 2

Transcribed Image Text:2. With supplied data, perform the following analyses:
I.
Show relevant calculations
II.
Draw the graph
III.
Find the result and interpret it.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 6 steps with 9 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

