Q: What Are the Anaplerotic, or “Filling Up,”Reactions?
A: Metabolic pathways can be defined as the set of chemical reactions that are occurring in a living…
Q: What product is formed when the compound is treated with Benedict's reagent?
A:
Q: In the following reaction, what type of reaction is occurring? H2CO3 H* + HCO3
A: Answer 1: Option NEUTRALIZATION is correct. Because : In Biochemistry, the term "carbonic acid" is…
Q: Why is the term dark reactions misleading?
A: Photoautotrophs are a type of higher organisms that can make their source of energy by using light…
Q: Identify the coefficients of the correct balanced equation for the reaction, CaCl2 + AGNO3 D AgCl +…
A: The given reaction is, CaCl2(aq)+AgNO3(aq)→ AgCl(s) +Ca(NO3)2(aq).
Q: What reaction is taking place in the above activity?
A: Enzymes are the biocatalysts that speed up biochemical reactions at optimum pH and temperature. The…
Q: Which of the following statements is CORRECT regarding the following intermediate? H2N H NH2
A: "Since you have asked multiple questions, we will solve the first question for you. If you want a…
Q: Write an unbalanced equation, using the correct formulas for all reactants and products.
A: An unbalanced equation is a reaction in which the number of atoms in the reactant is not equaled to…
Q: Cao (s) (calcium oxide) is the main ingredient of concrete. While mixing Cao with water, the mixture…
A: Given that the CaO reaction with H2O gives out heat and forms Ca(OH)2 in the product. So the…
Q: What are the possible sources of error when experimenting with sodium bicarbonate and hydrochloric…
A: Sodium bicarbonate : Sodium bicarbonate is a salt that breaks down to form sodium and bicarbonate in…
Q: H2N H -N Orcinol но A но он
A: The shown biomolecule is Guanosine(nucleoside) that is heated with orcinol for its quantitative and…
Q: Which set of conditions will give the product shown in this reaction? Br
A: Given Reaction:
Q: Calculate ΔG for the reaction H2O(l) ⇆ H+(aq) + OH−(aq) at 25°C for the following conditions.…
A: H2O(l) ⇆ H+(aq) + OH−(aq) ΔG=ΔG0+RTlnK Here, K= ratio of concentrations of the products (K2)divided…
Q: On the basis of the appearance of this plate, what are you testing for? Describe the reactions at A,…
A: The disc diffusion methodology (DDM) is an agar diffusion method (ADM) because the plant extract to…
Q: Write the reaction, occurring in the liver, that causes theoxidation of ethanol. What is the product…
A: The liver is an organ involved in detoxification reactions in the human body.
Q: Give a reaction equation for the qualitative and quantitative determination of I2.
A: Iodine: Iodine (I) is a lustrous element; atomic number is 53. It is the heaviest of the stable…
Q: Where in the periodic table are the best reducing agents found? The best oxidizing agents?
A: The reducing agents are the elements that reduces other elements and in the process, gets oxidized.…
Q: Which of the following correctly identifies the products and reactants labeled in the image above?
A: Photosynthesis is the process found in the chloroplast. Citric acid cycle and electron transport…
Q: Identify the oxidized reactant, the reduced reactant, the oxidizing agent, and the reducing agent in…
A: The electron accepting molecule or ion is known as oxidizing agents. As the electrons are accepted…
Q: Draw the alcohol used to produce each of the following ketones. Include all hydrogen atoms.
A: 3- Pentanone is also known as diethyl ketone. It can be prepared by ketonic decarboxylation of…
Q: What is the definition of hydrolosis reaction?
A: In human body, digestion and glucose metabolism are examples of hydrolysis reaction. Solubilization…
Q: In the reaction shown above, Acetaldehyde is being. to form Ethanol. (note the direction of the…
A: The fermentation process is an anaerobic process of generating ATP(adenosine triphosphate); a unit…
Q: hat is the condensed formula of the products obtained from complete hydrolysis with the picture?
A: Background information for lipids Lipids are a group of compounds that are oily or greasy in…
Q: In the electrolysis of aqueous sodium bromide, there are two possible anodic reactions: *2H2O(l)…
A: Water is reduced at the cathode in the electrolysis of NaBr. This occurs because water is more…
Q: Draw the experimental setups for the preparation of benzoin and write the reaction mechanism.
A: Introduction Benzoin condensation involves the formation of benzoin from two benzaldehyde molecules…
Q: Is the total enthalpy (H) of the reactants for an endothermic reaction greater than or less than the…
A: The Heat of reaction or enthalpy of reaction is the change in enthalpy of a chemical reaction that…
Q: Potassium, a silvery metal, reacts with bromine, a corrosive, reddish liquid, to yield potassium…
A: A chemical equation in which the number of each type of atoms is equal on the two sides of the…
Q: How many mL of 0.254 M NaOH are needed to fully react with 37.50 mL of 0.358 M H2SO4?
A: Concentration of the NaOH = 0.254 M Conc. of H2SO4 solution = 0.358 M or conc. of [H+]=2×0.358 M…
Q: Give full reaction for the synthesis of Diclofenac sodium
A: Diclofenac Sodium is the sodium salt of diclofenac, a benzene acetic acid derivate and NSAID with…
Q: Complete the following precipitation reactions using balanced chemical equations:
A: The reaction in which the formation of insoluble salt when two soluble salts mix together in the…
Q: Bacteria that have a composition of CSH702N. Determine the BOD for 30mg/L, following the following…
A: Microbes, which are tiny and nearly invisible, have had a huge influence on society since the…
Q: How many times does a secondary alcohol oxidized to a ketone group?
A: In organic chemistry, alcohols are those substances that have a hydroxyl (-OH) functional group…
Q: +2 H3N O:
A: The compounds that contain COOH as functional groups are called acids. The compounds that contain…
Q: The reactants in the dark reaction are: and
A: There are two phases to photosynthesis. The first one needs sunshine, but the second one does not.…
Q: discuss the following statement: “Whether the ΔG for a reaction is larger, smaller, or the same as…
A: Gibbs free energy is defined as the energy that is available to do work. This is the work done in a…
Q: What structural feature is necessary for an alcohol to undergo oxidation reactions?
A: Oxidation of alcohols is also known as Dehydrogenation reaction. In human body, many reactions take…
Q: A +B C+D 1) Calculate AG for the above reac un and indicáte whether the reaction is favorable or…
A: Gibb's free energy∆G: It determines whether the given reaction will be Spontaneous or…
Q: Give the reagent or chemical test that would differentiate the following pairs of compounds. Write…
A: Multiple questions asked. I will answer 1st question which has subparts in accordance with…
Q: Describe the Bohr effect.
A: According to Bohr Effect the affinity of hemoglobin for dioxygen decreases with decrease in pH
Q: What are the products when the following molecule is hydrolyzed using NaOH (soaponification)?
A: Saponification is a process, in which lipids are converted into soap.
Q: Identify the Lewis acid in the following reaction: A. BF3 B. F C. BF4 D. None of these is an acid.…
A: Boron trifluoride (BF3) is a Lewis acid because it is an electron-deficient molecule that may take…
Q: What does an acid based reaction that doesn't contain carbonate or bicarbonate produce?
A: When an acid and a base are placed together, they react to neutralize the acid and base properties,…
Q: What three important properties do catalyst have?
A: Catalyst are used in chemistry and biocatalysts or enzymes are used in biological system. It helps…
Q: A reaction at 23°C has ∆G = 1 kJ mol–¹. Why might this reaction become spontaneous at 37°C?
A: A reaction at 23 degree celecius has 1 KJ mol-1 This would make the G value negative and hence the…
Q: For the following reactions, identify the atom(s) being oxidized and reduced:
A: A redox reaction is one in which both oxidation and reduction take place simultaneously. When a…
Identify the major and minor product(s) of the following reaction:
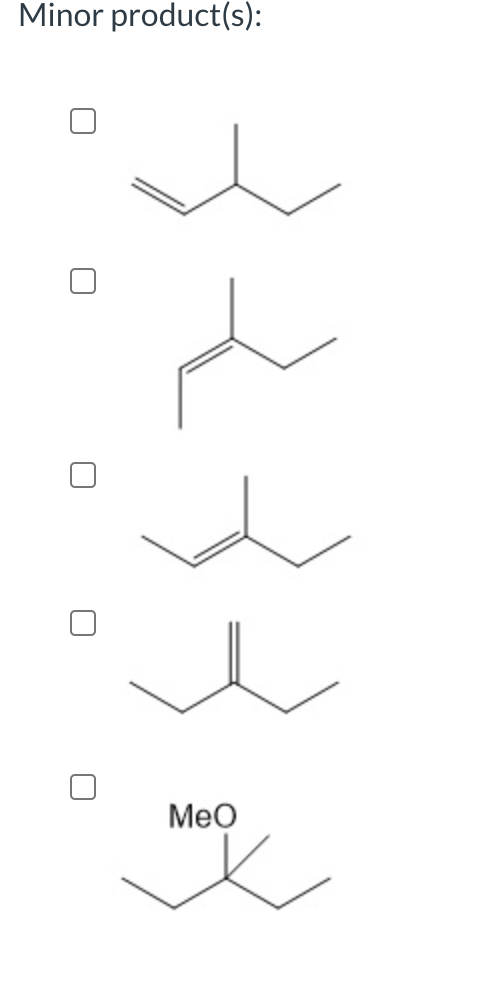
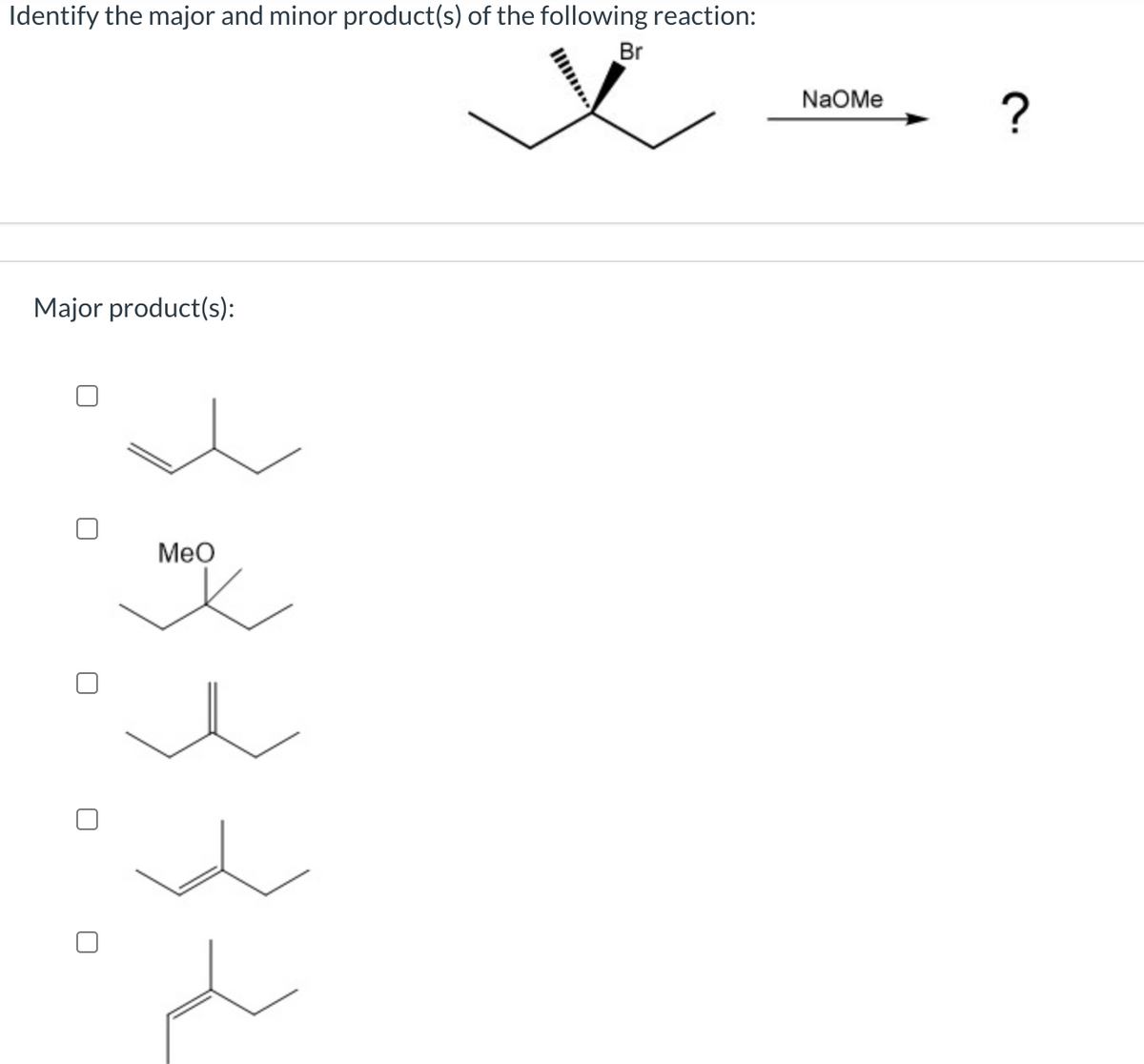
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Draw the product AND propose a reasonable, detailed stepwise mechanism, using curved arrow notation to show the flow of electrons, for the following reaction.If the phosphorus atom in 3-phosphoglycerate is radioactively labeled, where will the label be when the reaction that forms 2-phosphoglycerate is over?Determine whether or not reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. HBr(aq) + KOH(aq)--------> H2O(l) + KBr(aq)
- What are the possible sources of error when experimenting with sodium bicarbonate and hydrochloric acid experiment in yielding the reaction?In the partial reactions shown below, is the reactant undergoing oxidation or reduction?Write the reaction, occurring in the liver, that causes theoxidation of ethanol. What is the product of this reactionand what symptoms are caused by the product?



