Express your answer as a chemical equation. Identify all of the phases in your answer. > View Available Hint(s) ΑΣΦ OA chemical reaction does not occur for this question. Submit Previous Answers X Incorrect; Try Again; 5 attempts remaining Your answer is not a balanced chemical equation. Part B Cr(s) + Fe²+(aq) –→ Cr** (aq) + Fe(s) Express your answer as a chemical equation. Identify all of the phases in your answer. > View Available Hint(s) ΑΣΦ O IE A chemical reaction does not occur for this questic Submit Part C BrOg (aq) + N2H4 (g) → Br (aq) +N2(g) Express your answer as a chemical equation. Identify all of the phases in your answer. • View Available Hint(s)
Express your answer as a chemical equation. Identify all of the phases in your answer. > View Available Hint(s) ΑΣΦ OA chemical reaction does not occur for this question. Submit Previous Answers X Incorrect; Try Again; 5 attempts remaining Your answer is not a balanced chemical equation. Part B Cr(s) + Fe²+(aq) –→ Cr** (aq) + Fe(s) Express your answer as a chemical equation. Identify all of the phases in your answer. > View Available Hint(s) ΑΣΦ O IE A chemical reaction does not occur for this questic Submit Part C BrOg (aq) + N2H4 (g) → Br (aq) +N2(g) Express your answer as a chemical equation. Identify all of the phases in your answer. • View Available Hint(s)
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter4: Reactions In Aqueous Solution
Section: Chapter Questions
Problem 58QAP: The iron content of hemoglobin is determined by destroying the hemoglobin molecule and producing...
Related questions
Question
100%
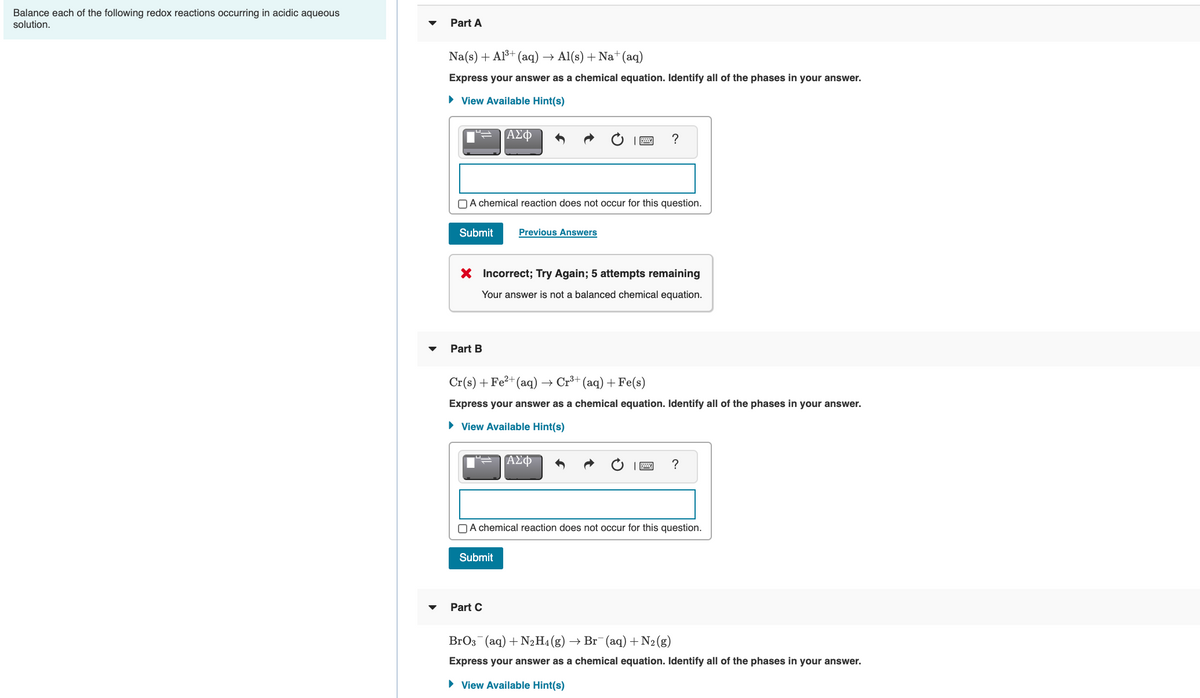
Transcribed Image Text:Balance each of the following redox reactions occurring in acidic aqueous
solution.
Part A
Na(s) + Al³+ (aq) → Al(s) + Na+(aq)
Express your answer as a chemical equation. Identify all of the phases in your answer.
• View Available Hint(s)
ΑΣφ
?
OA chemical reaction does not occur for this question.
Submit
Previous Answers
X Incorrect; Try Again; 5 attempts remaining
Your answer is not a balanced chemical equation.
Part B
Cr(s) +Fe²+(aq)
+ Cr+ (aq) + Fe(s)
Express your answer as a chemical equation. Identify all of the phases in your answer.
• View Available Hint(s)
ΑΣΦ
OA chemical reaction does not occur for this question.
Submit
Part C
BrO3 (aq) + N2H4 (g)
→ Br (aq) +N2(g)
Express your answer as a chemical equation. Identify all of the phases in your answer.
• View Available Hint(s)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
