Focusing Lenses Radiation Source Wavelength Selector Detector Atomizer Signal Processor Amplifier Sample 1. What is the stationary phase? 3 1. Why is it important to prevent air bubbles from forming in the column? * 2. What are the advantages of capillary column over packed columns in GC?
Focusing Lenses Radiation Source Wavelength Selector Detector Atomizer Signal Processor Amplifier Sample 1. What is the stationary phase? 3 1. Why is it important to prevent air bubbles from forming in the column? * 2. What are the advantages of capillary column over packed columns in GC?
Chapter4: Least-squares And Calibration Methods
Section: Chapter Questions
Problem 8P
Related questions
Question
6
Transcribed Image Text:Nitrate levels can be reported in several different units. Report the
conc. of nitrate in your tap water unknown in unit mg N\L, mg NO 2 \L
and as mol\L .
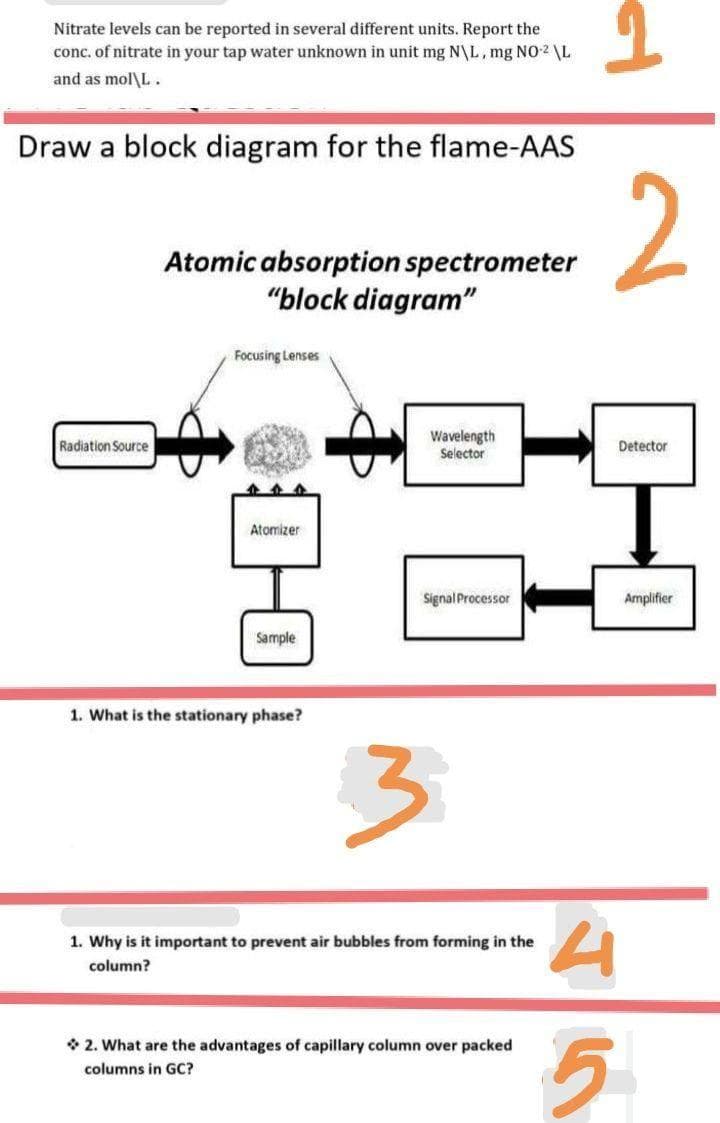
Draw a block diagram for the flame-AAS
2
Atomic absorption spectrometer
"block diagram"
Focusing Lenses
Wavelength
Selector
Radiation Source
Detector
Atomizer
Signal Processor
Amplifier
Sample
1. What is the stationary phase?
1. Why is it important to prevent air bubbles from forming in the
column?
* 2. What are the advantages of capillary column over packed
columns in GC?
00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning