Four test tubes with 20mL of water and cut flowers were set up. Three of the test tubes had food coloring added. You observ the flowers that were placed in the test tubes for the following five days. You record your observations each day at 10:00am. On the fifth day, the flowers in the colored test tubes began to turn the color of the liquid in which they were placed. You hypothesis was: IF cut flowers are placed in colored water, THEN the flower petals will absorb that color throughout the petals. Experimental Design In this experiment, one of the constants was: O 10:00am O The test tube without food coloring O The ending color of the flowers O The color of the water
Four test tubes with 20mL of water and cut flowers were set up. Three of the test tubes had food coloring added. You observ the flowers that were placed in the test tubes for the following five days. You record your observations each day at 10:00am. On the fifth day, the flowers in the colored test tubes began to turn the color of the liquid in which they were placed. You hypothesis was: IF cut flowers are placed in colored water, THEN the flower petals will absorb that color throughout the petals. Experimental Design In this experiment, one of the constants was: O 10:00am O The test tube without food coloring O The ending color of the flowers O The color of the water
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter2: Matter And Energy
Section: Chapter Questions
Problem 2.4TC: Specific gravity is a physical property. Beakers hold three clear, colorless liquids A, B, and C....
Related questions
Question
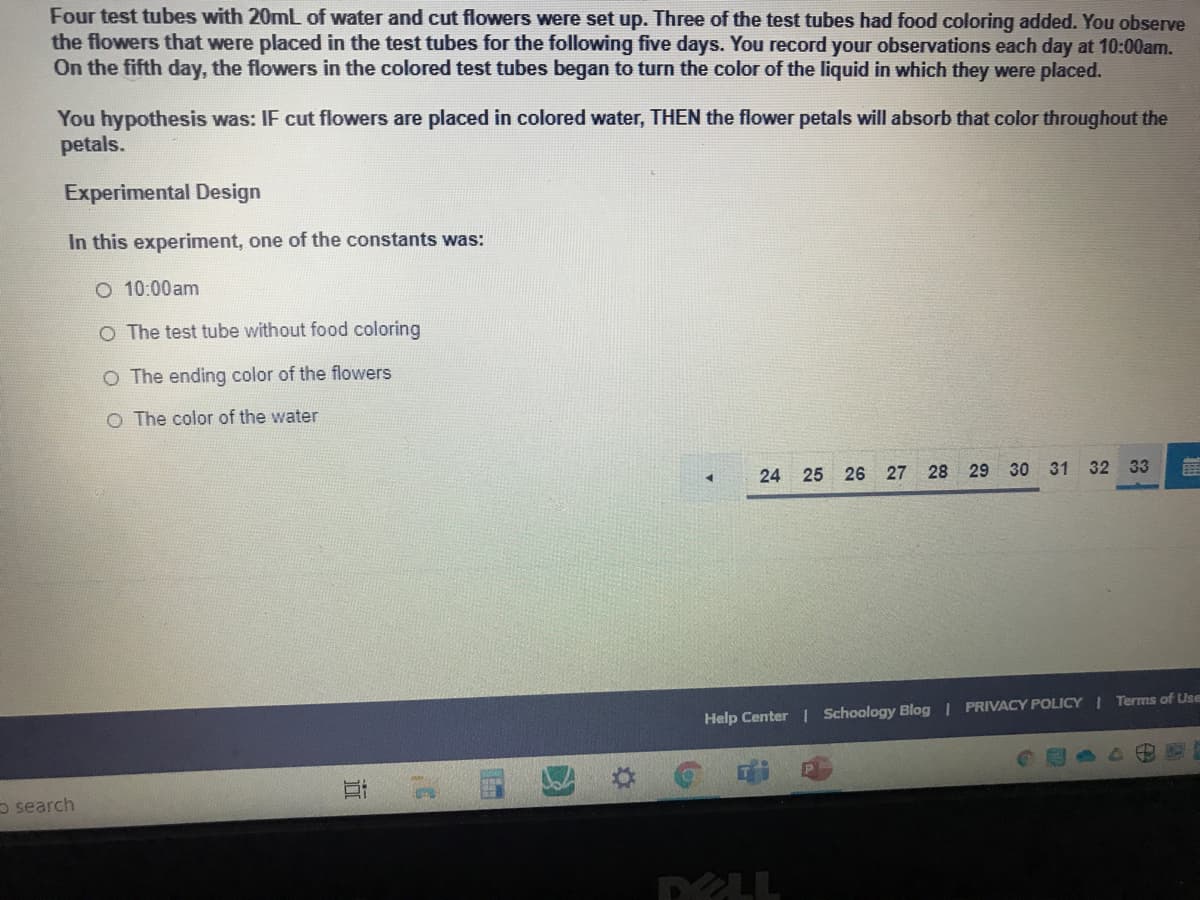
Transcribed Image Text:Four test tubes with 20mL of water and cut flowers were set up. Three of the test tubes had food coloring added. You observe
the flowers that were placed in the test tubes for the following five days. You record your observations each day at 10:00am.
On the fifth day, the flowers in the colored test tubes began to turn the color of the liquid in which they were placed.
You hypothesis was: IF cut flowers are placed in colored water, THEN the flower petals will absorb that color throughout the
petals.
Experimental Design
In this experiment, one of the constants was:
O 10:00am
O The test tube without food coloring
O The ending color of the flowers
O The color of the water
24 25
26 27 28 29 30 31 32 33
Help Center | Schoology Blog I PRIVACY POLICY Terms of Use
5 search
DLL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co


Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning