Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Complete the percent error
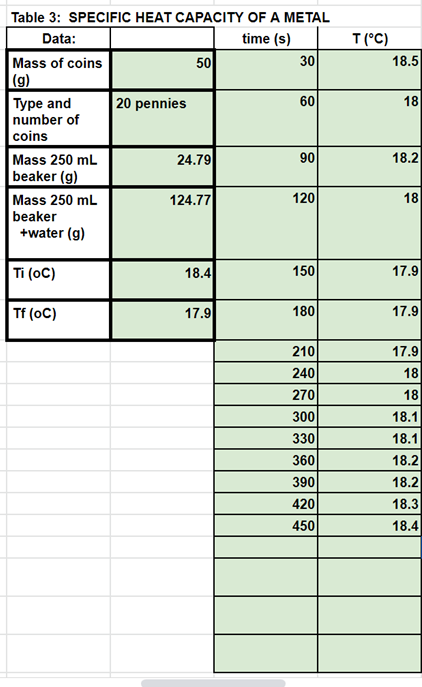
Transcribed Image Text:Table 3: SPECIFIC HEAT CAPACITY OF A METAL
Data:
time (s)
T (°C)
Mass of coins
50
30
18.5
(g)
Туре and
number of
20 pennies
60
18
coins
Mass 250 mL
24.79
90
18.2
beaker (g)
Mass 250 mL
124.77
120
18
beaker
+water (g)
Ti (oC)
18.4
150
17.9
Tf (oC)
17.9
180
17.9
210
17.9
240
18
270
18
300
18.1
330
18.1
360
18.2
390
18.2
420
18.3
450
18.4
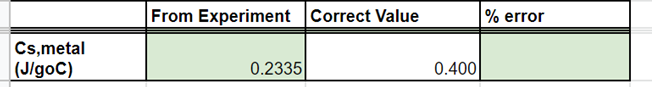
Transcribed Image Text:From Experiment Correct Value
% error
Cs,metal
|(JlgoC)
0.2335
0.400
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

