Q: Consider 1-bromopropane, CH3CH2CH2Br. (a) Which of these is the lowest energy conformation?
A: The newman projection of 1 bromopropane shows the lowest energy conformation is Anti staggered form…
Q: Does 2-pentyne have enantiomers? Explain.
A: Enantiomer is one of two stereoisomers that are mirror images of each other that are…
Q: H3C Which structure(s) below represent(s) the diastereoisomer(s) of the compound on the picture?…
A: Given that : We have to identify which of the following structures below represents the…
Q: OH CH3
A: In a chair conformation where the bigger substituents are present in equatorial position that…
Q: Add wedge-and-dash bonds to indicate stereochemistry. H CH3 H,C + Br -Br CH, H3C H Br Br
A: When trans alkene react with Br2 gives which gives anti addition formed meso product Complete…
Q: h) CH3-CH2-CH2-CH2- ċ=ċ-CH3 i) CH3 -CH3 CH3 CH3-CH,–ĊH ČH3 HIC
A:
Q: ball & stick v -+ labels Which of the Newman structures below represents this conformation of…
A: Different forms of a compound formed by the rotation around the single bond between the…
Q: Which is more stable, cis-2-methyl-3-hexene or trans-2 methyl-3-hexene?
A: The given compounds are cis-2-methyl-3-hexene and trans-2 methyl-3-hexene. The two compounds are…
Q: Shown below is one of the chair conformations of trans-1-bromo-3-methylcyclohexane, a disubstituted…
A:
Q: Create a 3D model of the most stable chair conformation of (1R,3S)-3-chlorocyclohexan-1-ol
A: In substituted cyclohexanes, stable conformation has the maximum number of substituents on the…
Q: please answer both name of highlighted side chain compound H C=0 CH, CH- CH,- CH2 CH, H C=0 CH, CH,…
A: According to Bartlby guidelines I solve first question. You post second question separately.
Q: Give the IUPAC name of the following: la) CH3 CH3 (b) CO2H (c) Br CHCH2CH2CHCH3 Br H3C CH3 (d) Br…
A: The IUPAC name of the following compounds has to be given below. (a) (b) (c) (d) (e) (f)
Q: By considering viewed through the C-2-C-3 bond, which below conformations of 2.3- dibromabutane show…
A: Conformation- III Most stable as it is a Staggered Conformation and All groups are anti to each…
Q: Draw all the alcohol having the formula C5H12Land which are choral
A:
Q: Q1:- Which of the isomeric alcohols having formula CSH120 are chiral? Which are achiral? Draw the…
A: The question is based on the concept of isomers. molecules having same molecular formula but…
Q: CI CH,CI H3C C= CH3 H H3C H.
A: The numbering will be as follows: There is 'Cl' substituent at 6th and 7th position.
Q: Why is this chair conformation the most stable trans-1-chloro-3-flurocyclohexane? Wouldn't the…
A: This question is related to stability of conformations
Q: Citing down the C3-C4 bond, and using the drawings below, complete the Newman projections for all…
A: Here, three staggered and three eclipsed forms are present. I, III and V are eclipsed forms as bond…
Q: 2. Decide whether to use E/Z nomenclature or cis or trans (or if neither are applicable). Then, draw…
A:
Q: Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that…
A:
Q: 3. For each pair of the following molecules, indicate whether its members are identical, structural…
A: Note :- Since you have asked multiple question, we will solve the first question with first three…
Q: Br -CHC=CH -ĊCH,CH3 ČH3 ČH3 (S-Isomer) (Racemate)
A:
Q: Which conformation in the picture attached represents the most stable ECLIPSED conformation of…
A: The arrangement of dihedral angle between the front bond and the back bond is 00 is known as…
Q: H3C CH2CH3 H. CH,CH3 Но Br H3C HO. Br CO,H -H- CH2CH3 H- OH Но CO,H CH,CH3
A:
Q: H3C,,, + enantiomer CH3 H3C CI
A: ->First of all there occur elimination reaction in presence of base and alkene formed. -> Then…
Q: (R)-2,2-dimethylcyclopentan-1-ol (а). bicyclo[4.1.0]heptane (b). cis-
A: To solve this problem we have to write the structure of the given compounds .
Q: CH3 CH3 `CH CH, CH, H3C-CH2 -ĊH-C-CH2-CH-CH-CH2-CH2-CHg CH3 CH3 CH2 CH3
A: Given, Hydrcarbon with chain of ten Carbon atoms. Numbering should be done following the IUPAC…
Q: /hat is the most stable chair conformer for the compound shown (left)? Use the labels indicated the…
A: In chair conformation most stable conformation is one in which more bulky group is equatorial.
Q: Which of the statements below correctly describes the chair conformations of cis-1,4-…
A: Note: In both chair conformations one ethyl at axial, one ethyl at equatorial. Therefore both chari…
Q: 7. CH3(CH3)CH=CH2 3. CH3CH2CH2C(CH3)=C(CH3)CH2CH3 э. СНЗCH2CH(CHз)C-CH 10. CH3CH=CHCH2CH=CH2
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Draw curved arrows to show the movement of the electrons that result in formation of the given…
A: a.-OH acts as a base and it has tendency to abstract proton. The curved arrows that show the…
Q: #1. CH3 CH3 H,C CH3 Name #2. H, C. CH3 Name H3 C #3. 5-cyclopropyl-3-heptene diagram
A: For IUPAC name you have to 1st consider the longest carbon chain containg the functional group and…
Q: ace the conformations of cis-1-isopropyl- ble to most stable). А
A: Stablity of these molecule when bulkyl group is at equitorial position not axial position. At…
Q: H,C -Br H3C
A:
Q: 3. A) Draw the most stable conformation of trans-1-ethyl-3-methylcyclohexane. Br B) Complete the…
A: 3 (A) The most stable conformation of any compound is formed when the repulsion between the groups…
Q: In the least stable conformation of cis-1-isopropyl-4-methylcyclohexane. The isopropyl and methyl…
A: Option (B) " The isopropyl group in an axial position and the methyl in an equatorial position " is…
Q: Identify the equilibrium below that shows the two chair conformations of the following compound: Br…
A: In the cyclohexane system, the most stable one is chair form. The equilibrium is set up between…
Q: #1. CH3 CH3 H, C. CH3 Name #2. H,C. Name CH3 H3 C #3. 5-cyclopropyl-3-heptene diagram
A:
Q: CH, CH3 CH2-CH3 CH,-C-CH-CH-CH2 CH3 CH2-CH-CH, CH3 d) CH2-CH3 CH;-CEC-CEc-CH-CH-CEc-CH3 CI e) CH3…
A: Since you have posted multiple subpart question, we will answer the 3 subparts (d,e,f) for you.…
Q: Does 1-pentyne have enantiomers? Explain.
A: Enantiomer is one of two stereoisomers that are mirror images of each other that are…
Q: H H3CCH3 Br CI H3C, H3C' OCHS H3C CH3 'CH3 HA CH3 H, H II
A: Given conformations,
Q: Sighting down the Ci-C2 bond axis of 1-bromopentane, Br-CH2-CH÷CH»CH=CH®, draw the Newman projection…
A: Given that : For 1-bromopentane, we have to draw the Newman projection of : a) the anti staggered…
Q: Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that…
A: Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that…
Q: : does the stable anti-conformer of CH2CI shows an equivalency of CH3 H's? Why?
A:
Q: How many axial hydrogen atoms are there on the most stable conformation of…
A: The chair form of the conformation is most stable conformation then the others as there is no any…
Q: By considering viewed through the C-2-C-3 bond, which below conformations of 2,3- dibromobutane show…
A: In alkanes, due to free rotation around carbon carbon single bond infinite number of structure are…
Q: Which conformation(s) show(s) the bromines in a gauche orientation for 2,3-dibromobutane, viewed…
A:
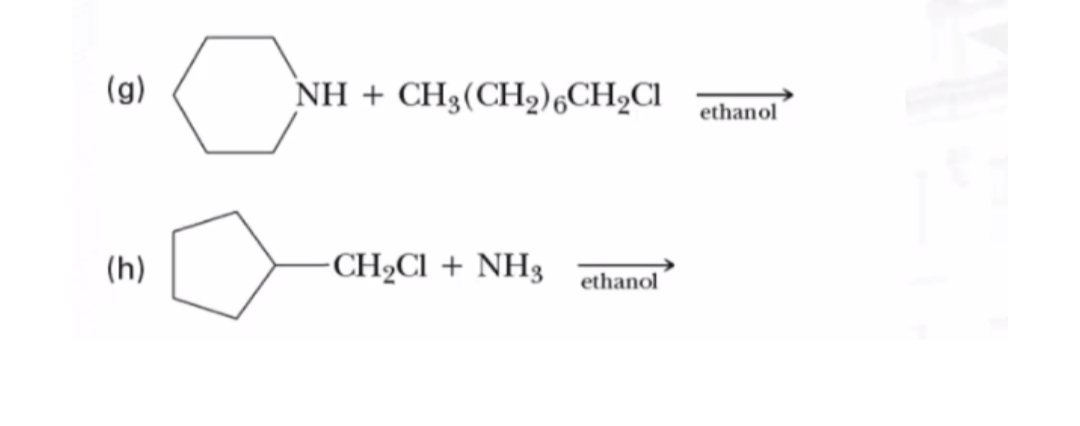
Step by step
Solved in 2 steps with 2 images

- Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.)E,Z isomerism but not chirality1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.

