Give the correct conformation of the compound A and B. H. H. H. H. H. H. H H. H H. H. O A. s-cis and r-trans OB. Cis and trans C. r-cis and r-trans O D. s-cis and s-trans
Give the correct conformation of the compound A and B. H. H. H. H. H. H. H H. H H. H. O A. s-cis and r-trans OB. Cis and trans C. r-cis and r-trans O D. s-cis and s-trans
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter6: Alkanes & Alkenes
Section: Chapter Questions
Problem 2E
Related questions
Question
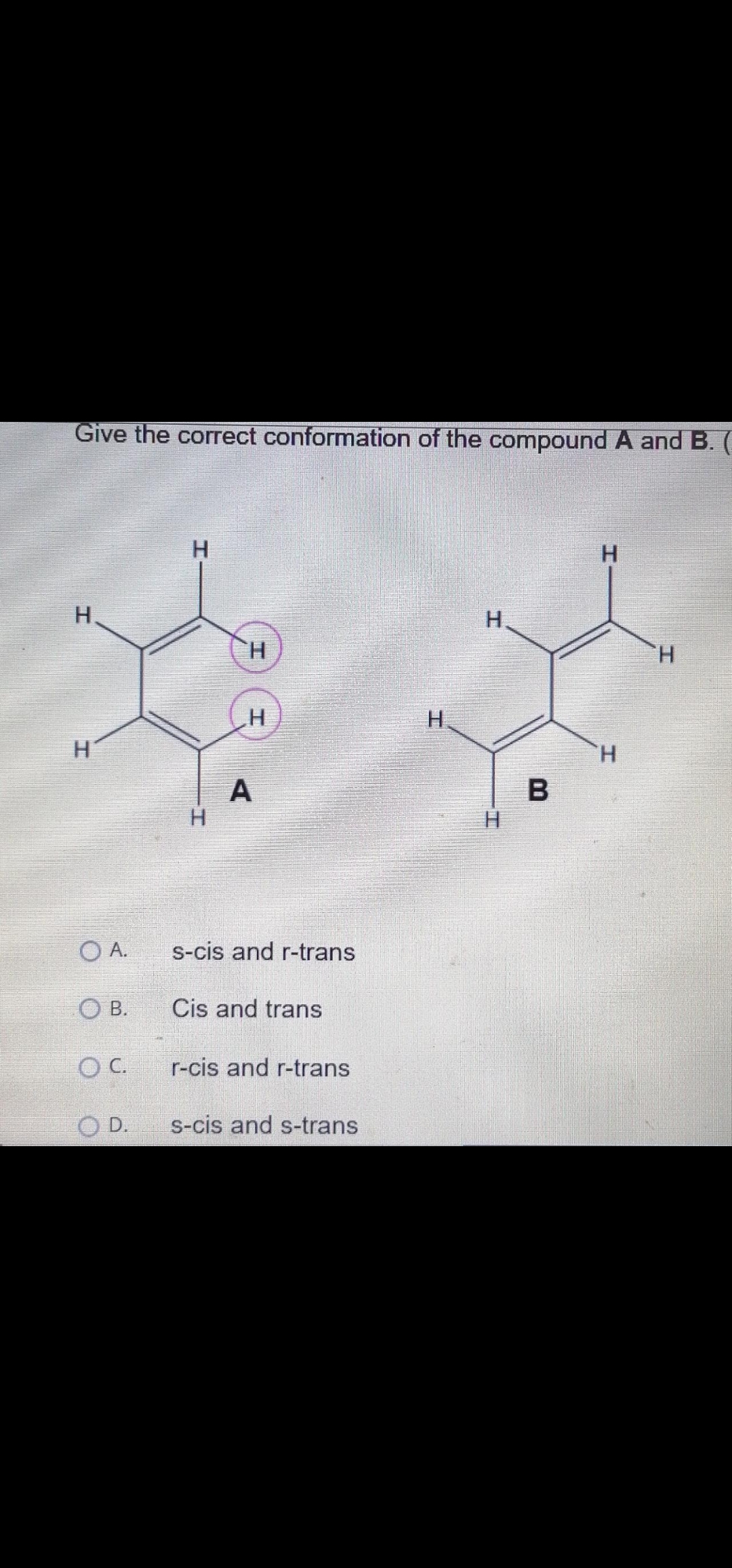
Transcribed Image Text:Give the correct conformation of the compound A andB.
H.
H.
H.
H.
H.
H.
H.
H.
A
O A.
s-cis and r-trans
O B.
Cis and trans
O C.
r-cis and r-trans
O D.
s-cis and s-trans
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning