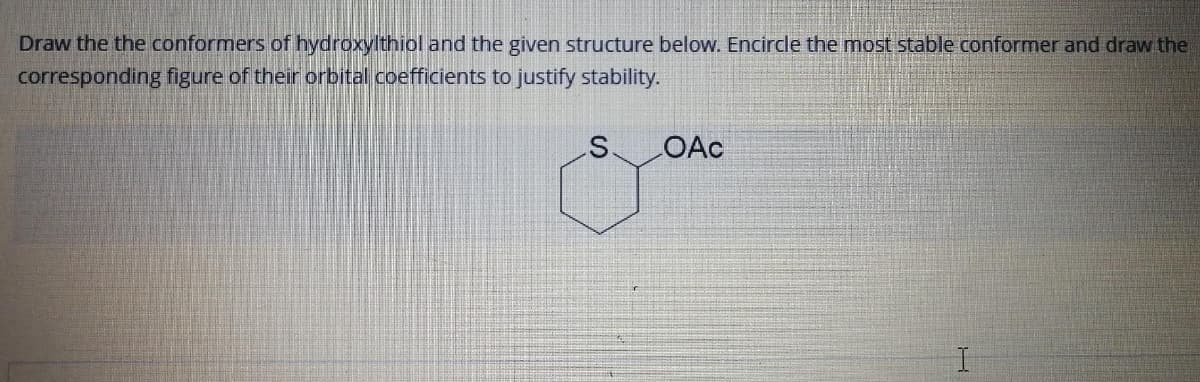
Draw the the conformers of hydroxylthiol and the given structure below. Encircle the most stable conformer and draw the corresponding figure of their orbital coefficients to justify stability. OAc
Q: d) ОН
A: The most stable conformation of cyclohexane ring is called as chair conformation. The reason behind…
Q: SECH3 CH3 CH3 SECH3 CH3 H3C H3C H3C. `SECH3 SeCH3 HзC. SECH3 ČH3 CH3 ČH3 A SECH3 В C H3C CH3
A: The given molecule is: The molecule can be converted into various forms of its chair conformations.…
Q: SA. For chains, draw Newman projections of all conformation= determine their energy OCSL 6.1 6.14…
A:
Q: What are the chair conformations of 1,2-dichlorocyclohexane and their IUPAC name using R/S or…
A: The chair conformations are the two most stable conformations between which a cyclohexane rotates…
Q: Adding a methyl group to propene, as 1-butene doubles the number of eclipsed and bisected…
A: The 4 conformations of 1-butene are eclipsed-1,2 and bisected-1,2.
Q: CI
A:
Q: How many energetically different eclipsed conformations does 1-bromo-2-methylpropane, viewed along…
A: The Newmann projections are stable when two bulky groups or atoms have a decent separation between…
Q: Given that the free energy of the twist-boat conformer of cyclohexane is 5.3 kcal/mol greater than…
A:
Q: How many energetically different eclipsed conformations does 1-bromo-2-methylpropane, viewed along…
A: The given Compound is 1-bromo-2-methylpropane. To find: The number of eclipsed conformation viewed…
Q: Molecular Formula: C8H03 Compute for the DU (degree of unsaturation) Then draw the structure of the…
A:
Q: Draw the more stable chair conformation of each of the following compounds in which an…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: (b) Consider the following tetra-substituted cyclohexane: он i. Draw the alternative chair…
A: A stable chair confirmation is one where the bulky groups are at equatorial position.
Q: a. Convert the following Newman projection of compound G to a three dimensional line structure in…
A:
Q: Nhen compound X undergoes ring-flipping, which compound below represents its ring-flipped conformer?…
A: We have given the organic compound i.e. 1,2diethyl-3-methyl cyclohexane ring we have to gives its…
Q: Which Newman projection for the conformation of 2-methylbutane will have energy Il on the potential…
A:
Q: Draw the two chair conformations for the following compound. Explain by calculation of 1,3-diaxial…
A:
Q: (i) Gauche conformation of ethane, (ü) felipsed conformation of ethene, (iii) Staggered conformation…
A:
Q: Make a model of cyclooctatetraene in the tub conformation. Draw this conformation, andestimate the…
A: Cyclooctatetraene is non-polar in nature and is non-aromatic compound. It adopts tub shaped…
Q: 4. Draw the most stable chair conformation for each of the following compounds: ОН (а) (b) (c) (d)…
A: In the chair conformation, the substituent may present at the axial or equatorial position. If the…
Q: Write the most stable chair conformation for each cyclonexane below. Br.
A: More the number of groups in equitorial position, more will be the stability of the conformation.…
Q: Which molecule do you expect to be more stable, a or b)? Use both chair conformations of each…
A: In order to draw the chair conformation of the given cyclohexane molecule. First draw the chair form…
Q: What are their 3D structures for all their possible constitutional isomers and/or stereoisomers.…
A:
Q: Sighting down the C1-C2 bond axis of 1-chloropropane, draw the Newman projection of, a) the anti…
A: Given compound: 1-chloropropane We have to draw the Newman projection of the: anti staggered…
Q: Draw 2 of the chair conformations for the compounds shown below and then cricle which are most…
A: Chair confirmation is one of the most stable conformation of the cyclohexane. In chair conformation…
Q: Draw chair conformations of cis-1-ethyl-3-methylcyclohexane. Is one isomer favored over the other?…
A:
Q: Calculate the energies of the 4 conformers below:
A: The 4 different conformations of the molecule are given of which the relative energies have to be…
Q: 3. Draw the Newman projections of the three possible staggered conformations of 2,3-dimethylbutane,…
A: Newman projection
Q: Discuss and compare two possible chair conformations of cis-1,3-dibromocyclohexane compound. Which…
A:
Q: CH3 CH3 CH3 CH3 Compound I Compound II (i) Are the above cyclohexane derivatives cis- or…
A:
Q: How many gauche interactions are present in the most stable chair conformation of…
A: Two conformers can be drawn for 1,1,2-trimethyl cyclohexane. In one conformer, C-1 and C-2 methyl…
Q: How many chlorines are axial in the most stable chair conformation of the molecule below? (enter 0,…
A: The equatorial position will be mainly for the bulky group while the axial position will be for the…
Q: 2. Draw the two chair conformations of cis-1-ethyl-4-methylcyclohexane, and determine which of the…
A:
Q: Draw every stereoisomer for 1-bromo-2-chloro-1,2-difluorocyclopentane. Use wedge-and-dash bonds for…
A: Please find your solution below : Stereoisomers are the compounds which have same composition but…
Q: (f) a Newman projection of the following molecule looking down the C2-C3 bond in the anti-…
A: Newman projection visualizes the conformation of a chemical bond from front to back, with the front…
Q: a. Convert the following Newman projection of compound G to a three dimensional line structure in…
A:
Q: The anti-conformation is the most stable n-butane conformation. If you could take a snapshot of a…
A: Anti-conformation of n-butane is the most satble conformation. To determine, for 1 mole of n-butane…
Q: Show the Newman projections for the staggered and eclipsed conformations of ethane ?
A: The structures of Newman projections are given in the following steps.
Q: Draw the most stable chair conformation possible for this compound. Are there any 1,3-diaxial…
A: Note: Put bulky group like isopropyl in equatorial position and write the most stable chair. Here in…
Q: Is there a difference in the stability of the two cyclohexane conformations?
A: The structure of chemical compound can be represented as structural formula. A structural formula…
Q: a) State two (2) types of stereoisomer. b) Explain briefly, which compound hexane or…
A: a. Stereoisomerism is the arrangement of atoms in molecules whose connectivity remains the same but…
Q: Are the two conformers identical? If not, circle the one with lower energy. Are the two methyl…
A: 1,3-dimethyl cyclohexane conformers
Q: B AG = 18.3 kJ/mol AG = 5.9 kJ/m. %3D ion
A: It is happen due to stearic relation between group In axial position group are bear to each other…
Q: The diaxial conformation of cis- 1,3-dimethylcyclohexane is approximately 23 kl/mol (5.4 kcal/mol)…
A: We have been asked to draw the chair conformation and show the unstability of diaxial conformation.
Q: Explain why following molecule is achiral despite having no symmetry in structure shown. HI N H3C
A:
Q: Create Newman projections depicting conformations along the carbon-carbon bond described above in…
A: Given:
Q: How do you account for the difference in energies between the two staggered conformations of…
A: Staggered and eclipsed conformations of 1,2 - dichloroethane
Q: In the lowest energy chair conformation of cis-1,3-dimethylcyclohexane, how many axial positions are…
A: We have to predict the number of methyl group on lowest energy confirmation.
Q: draw a model of cyclooctatetraene in the tub conformation. Draw this conformation, andestimate the…
A: Cyclooctatetraene is non-polar in nature and is non-aromatic compound. It adopts tub shaped…
Q: Molecular Formula: CHgO3 Compute for the DU (degree of unsaturation) Then draw the structure of the…
A: The degree of unsaturation or Double bond equivalence of any organic system represented by their…
Step by step
Solved in 2 steps with 2 images

- Consider the Newman projection below. a. Draw a full Lewis structure of this molecule with R1=Me,R2=Et , and R3=iPr . b. Given the sizes of these R groups (R3R2R1) , does the Newman projection above show thelowest potential energy conformation of this bond? If not, draw a Newman projectionshowing the lowest P.E. conformation (sighting down this same bond). c. To draw a Newman projection in the lowest P.E. conformation, the following rule of thumbusually applies: Place the largest group on the front carbon anti to the largest group on theback carbon. Is your answer to the previous question consistent with this rule of thumb?Fill in the blanks: cis-1,3-Dimethylcyclohexane has two different chair conformations: one withboth methyl groups in __________ positions and one with both methyl groups in ____________ positions.Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.
- Build a model of methylcyclohexane, and use the model to complete the following Newmanprojections of methylcyclohexane in the chair conformation: a. When the methyl group is in an axial or equatorial (circle one) position, the molecule is inits lowest potential energy conformation. b. Label one Newman projection above anti and the other gauche to describe the relationshipbetween the methyl group and C3 of the ring. c. In general, which is a lower PE conformation, anti or gauche? d. Explain how your answer to b and c provide an explanation for why it is more favorable fora large group to be in an equatorial than an axial position.Given the compounds below, which is the more stable one based on their chair conformation?Is the 1st compound ( more, less or equally) stable than B? Pls show through illustrations of chair conformations
- Draw both chair Conformers of chlorocylohexane. How much higher in energy is the less stable Conformer?a) i) Draw the Newman projections for the conformations of ethane (staggered and eclipsed) Which is higher in energy? Which is more stable? Next to each conformer, draw the equivalent ‘sideways’ view, using wedged bondsa. Draw Newman projections of the two conformers of trans-1,3-dimethylcyclohexane. b. Which of the conformers predominates at equilibrium?
- translate the bond-line notation structure to the Newman projection by filling int the missing groups (A, B, C, D or E) on the lines in the Newman projection so they match the conformation given in the original structure. Circle if the conformation is a staggered or eclipsed.a) Enter the name of the following compound. B) Write down all the chair conformations. c) Which conformation is the most stable and explain why. d) Which conformation is the least stable and explain why.draw a model of cyclooctatetraene in the tub conformation. Draw this conformation, andestimate the angle between the p orbitals of adjacent pi bonds.



