Q: 1. For the reaction, 2 NO (g) + 2 H₂ (g) following equilibrium concentrations: [NO]= 8.1 x 10-³ M…
A: Since you have asked multiple question, as per our company guidelines we are supposed to answer…
Q: The zinc content of a 1.03 g ore sample was determined by dissolving the ore in HCl, which reacts…
A: According to the question we have, The mass of the ore sample is = 1.03 gm The volume of the HCl…
Q: Part II: For each experiment, calculate the values of [BB-], [HBB] and [H+] to fill in the table…
A: Since concentration cannot be negative, in trial-A-2, [HBB] will be 4.2*10-5, and K = 3.27*10-8
Q: A new analytic instrument is capable of detecting picogram quantities of a chemi- cal substance. How…
A: We have given a new analytic instrument is capable of detecting picogram quantities of a chemical…
Q: If a pharmacist dissolves 1.2 g of a medicinal agent in 60 mL of a cough syrup having a specific…
A: Given, mass of medicinial agent = 1.2 g volume of cough syrup = 60 mL specific gravity = 1.20
Q: If a pharmacist combined 50-mL portions of three syrups having specific gravities of 1.10, 1.25, and…
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: 1. How many moles of NaOH (FW = 40g/mol) are there in 2.0 grams NaOH? 2. How many grams of Na2CO3…
A:
Q: Which chair conformer is the most stable/lower in energy: EXY Because axial positions are more…
A: Given chair conformations:
Q: Determination of the Empirical Formula of Magnesium Oxide Data Sheet ata Table 1: Empirical Formula…
A: #1 is correct. #2 , the mass of product can be calculated using the formula : Mass of product = (…
Q: A) To 0.5 mL of 3% (or 6% whichever is available in the lab) hydrogen per- oxide, H₂O₂, solution add…
A: For each reaction (test tube A,B, and C) write balanced half-reactions and a total reaction in ionic…
Q: reaction should occur in each of these?
A: In a double displacement reaction if one of the product formed is is in solid state then that double…
Q: A sugar crystal contains 1.8 * 1017 sucrose ( C12H22O11) molecules. What is the mass in mg?
A:
Q: 1. What is the enthalpy change when 12.8 g Hz (g) reacts with excess (lig) to form HCl (g) H₂ (g) +…
A:
Q: Use chemical equations to show how you would use any additional reagents to complete the following…
A:
Q: 5.9) How much energy would be required to vaporize (boil) 50.0 grams of each of the following…
A: Given data;
Q: Granulated sugar (sucrose, C12H22O11) has an average density of about 0.76 g/cm3. Calculate the…
A: Given, Granulated sugar : C12H22O11 Density of C12H22O11 = 0.76gcm3
Q: Determine the product(s) in the reaction below. A. D. A and C E D с CI OH + HCI B. OH E. B and C CI…
A: Here the reaction is a nucleophilic substitution reaction. Cl- acts as a nucleophile and the Spiro…
Q: This reaction represents the titration from today's lab. 2- 2- 2 S₂03² + 1₂ → S406²- + 21- In this…
A: Oxidizing agent: The species which oxidize the other elements by taking electrons from them . When…
Q: Which of the following will have an electron configuration ending with 4p? Choose ALL that apply. d.…
A: Which of the following will have an electronic configuration ending with 4P6. That is I have to…
Q: a) b) Me CN NO₂ Me Cl₂ AIC13 NO₂ Et Br₂ FeBr3 H₂SO4 HNO3
A: Aromatic compounds can give electrophilic substitution reaction. In presence of electrophile it can…
Q: this answer correct eve vs n for nano = 10^-9 a
A: Unit of measurement is a definite magnitude of a quantity. It is used as a standard for measurement…
Q: Given that 1 in. = 2.54 cm (exactly), how many inches are there in one meter, expressed to six…
A:
Q: Classify the given compound/ ion by writing: A (if acid), B (if base), or C (if amphoteric) 1.…
A: Introduction to acids, bases and Amphoteric compounds; Acids - Which can donate proton or accept…
Q: Determine AH, q, w, and AE at 298 K and 1 atm pressure for the reaction of 9.211 g of NH3(g) with…
A: Given, Pressure = 1 atm, Temperature= 298 K, Mass of NH3 = 9.211 g
Q: In the next figure, the results of the X-ray diffraction of nickel has FCC crystal structure:
A: Given: Miller indices for three peaks (hkl) =(111),(002), (022) λ=1.54A° To Calculate: Part a)…
Q: Crastin® (CAS# 24968-12-5) is a polybutylene terephthalate (PBT) thermoplastic engineering polymer…
A: The given structure of polymer, Crastin(PBT) is made of two different monomers. Hence Crastin is a…
Q: Chemistry 3. (6%) Ca(HCO3 ) 2 is called calcium bicarbonate. It is one of the most abundant…
A: •In first question, we have to calculate the the molar concentration of HCO3-, normal concentration…
Q: If the Mn of a batch of LDPE is 3.2x109 g/mol, what is the corresponding full contour length in…
A: Given: The molar mass of LDPE is 3.2 x 109 g/mol To calculate: Full contour length in Angstroms (Ao)…
Q: Differentiate the various types of EAS: halogenation, nitration, sulfonation, alkylation,…
A: EAS is referred as Electrophilic Aromatic Substitution. The reactions which involves substitution of…
Q: For the diprotic weak acid H2A, ?a1=3.6×10−6 and ?a2=8.5×10−9. What is the pH of a 0.0800 M…
A:
Q: For the chemical reaction HCIO (aq) + NaOH(aq) → H₂O(1) + NaClO4 (aq) write the net ionic equation,…
A: Given chemical reaction is, HClO4 (aq) + NaOH(aq) ---------> H2O(l) + NaClO4(aq) To find out net…
Q: Suggest two ways to reduce the errors of determining the molecular weight of a polymer(polystyrene…
A: Polymer: A polymer would be a substance and material made up of macromolecules, which are very big…
Q: Calculate the standard atmosphere (1.00 atm) in a) mbar b) kPa
A: The conversion of quantity from one unit to another unit is known as unit conversion. During the…
Q: Gaseous azomethane C2H6N2 decomposes in a first order reaction when heated. C2H6N2 (g) -> N2 (g) +…
A: C2H6N2(g) -> N2(g)+C2H6(g) Initial amount in the flask=2.00g Rate constant=40.8min-1…
Q: Draw the expected major product for the hydroiodination of hexa-1,5-diene. Do not show hydrogen…
A:
Q: Ammonium iodide, a solid, decomposes to give NH3(g) and HI(g). At 406 °C, some NH4I(s) is placed in…
A: Given: For equation: NH4I(s)→ NH3(g) + HI(g) Total pressure = 1.07 atm Partial pressure of NH3=…
Q: Write the chemical equation involved for a specific EAS reaction,-Electrophilic Aromatic…
A: We know, EAS means Electrophilic Aromatic Substitution. Electrophilic substitution involving…
Q: Chemistry 6/(Practice problem 17.14 page 800 Solomons 1th Edition) Provide the reagents required to…
A: Synthesis of nitriles from carboxylic acid is reduction process. In this reaction, the carbonyl…
Q: D Question 1 Which of the following is not true about As2 Te3 as a pure substance? O The atoms are…
A: Given in following question a
Q: S2082-(aq) + 21-(aq) → 2SO42-(aq) + 12 (aq) 2S2O32-(aq) + 12(aq) → S4062-(aq) + 21-(aq) Solve for…
A: To find rate and rate constant for the given reaction.
Q: Calculate the pH of 1.0 x 10-3M HCl solution. (Round-off your answer to two decimal place)
A:
Q: Don't forget to show Toma Charges where necessary 1) Using the principles from orbital…
A: The stretching frequency of various functional groups depends on the force constant(k) and reduced…
Q: a) Write the full electron configuration for the sodium atom. b) Label and provide a point form…
A: The given question is about electronic configuration of Sodium. We have to write correct and full…
Q: A mixture of three compounds A, B, and C spotted on an alumina TLC plate gives the following Re…
A: Given : Rf Value of various compounds are given. And also the Given methanol is polar solvents. To…
Q: Arrange these elements according to first ionization energy. Highest ionization energy Kr Br Se As…
A: Here we have to determine the correct decrease order of ionisation energy of the following given…
Q: Consider this molecular-level representation of a mixture of gases. 98 Po If the partial pressure of…
A: Dalton's law: Dalton's law of partial pressures states that in a mixture of non-reacting gases, the…
Q: Given that Kw for water is 2.4 x 10-14 at 37 °C, calculate the pH of a neutral aqueous solution at…
A: i) To calculate the pH for neutral solution , we would use relation between Kw , [H+] and [OH-] .…
Q: Polonium, with an atomic radius of 0.168 nm, crystallizes in a simple cubic structure. Determine its…
A:
Q: N 0. Name: c. Relationship between structures above: Conformational isomer or Structural isomer 2.…
A: The answer to the following question is
Q: What is the value of the equilibrium constant, K', at 25°C for the following reaction if the…
A:
Step by step
Solved in 2 steps with 1 images

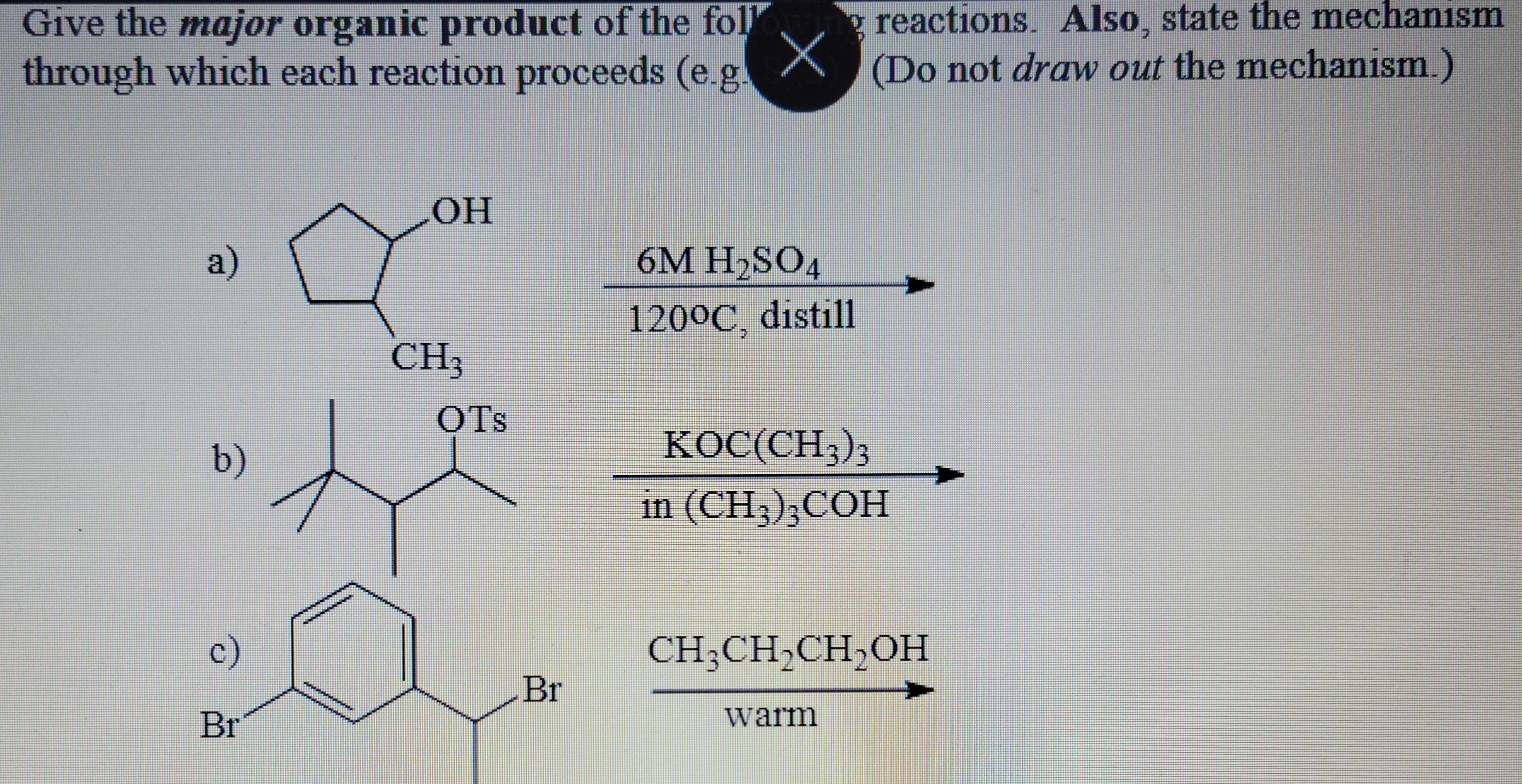
- Please give major organic products along with mechanism. Please also identify what type of reaction it is (SN1, SN2, E1, E2)(ii) The elimination reaction between 2-bromobutane and NaOCH2CH3 gives two organic products. Draw a mechanism for the reaction which produces the major organic elimination product and provide a rationale as to why that is the major product.(a) (i) 3-Methyl-2-butanol will react with sulphuric acid to give two isomeric alkenes in 3:1 proportions.(i) Write down the structures for these alkenes and assign appropriate systematic names to them. (ii) Name the most abundant isomer. (iii) Write down detailed mechanism for the formation of a minor product.
- Predict the major organic product formed when the compund shown below undergoes a reaction with H2O2 and then is heated in H2O. The 2nd photo is what I thought was the answer, but when I put it into Mastering Chemistry, it was counted as wrong and my feedback was, "What configuration is the C=C bond expected to haev?" I'm confused as to what this is referring to, so I appreciate any help given.I have this task in organic chemistry (book: Brown's introduction to organic chemisty, global edition). Task 10:42. In (a) I have to tell what the funcion of K2CO3 is in step 1. Is it that CO32- take the hydrogen atom in 1-napthol? Will it then be a SN2 mechanism? In (b) I have to name the amine used in step 2 to form Propanolol. But I can't really find out how to come up with an amine that will make that reaction. Here are two pictures of the task:help me please Please fill in the reagents,intermediates, and/or products of the following reactions. all the reactions shall be covered in Chapter 12-15 from Organic Chemistry, 6th Edition by Marc Loudon and Jim Parise.
- Give the major organic product(s) for each step of the following reactions (a,b,c)Fill in necessary products reactants or reagants of these reactions. Please note the existence of enantionmers in some cases.Chemistry please show detailed mechanisms for the reactions shown below. pleasemake sure to show all work like arrows, charges, and byproducts ect.



