Q: Experiment 3. Cómplementary Colors Filter color Color absorbed Red Orange Yellow Green Blue Purple
A: Metal complexes Undergo d-d transition and absorbs energy at differeret wavelength. But when the…
Q: Module Five Lesson Three Activity Two.pdf 2. Several of Smith's full or partial fingerprints were…
A: Calculation empirical formula: Step 1: % of atom / atomic mass. Step 2 : above value / minimum…
Q: 70 Ga 31 Express your answers as integers separated by commas. ? VO AE Np, Nn, Ne = Submit Request…
A: Given that , the element is gallium (Ga) with atomic number 31 and mass number 70. Now, Atomic…
Q: Part A SrO O Sr O Sr=0 OSr :0: Sr: Submit Request Answer
A: Ionic compounds are formed by the transfer of electrons. The cation donates the electrons and the…
Q: Condition Column I Column II Number of t2g electrons Rh0: [/Ar] 5s2 4d7 [RuCle]3- [RhCla]2- Ru:…
A:
Q: OH 1. CH,-PPh; (excess) 1. CH3CH,MgBr A 1. PCC B 2. CH;CHMgBr 3. NH,CI/H,0 2. PCC 2. H,О+
A: First the wittig reagent will react with the ketone to form alkene and then the product react with…
Q: +1 charge O charge -1 charge Answer Bank HO HN Он Он NH, NH, NH, H,N N H NH, O NH, но HN HO но H,N.…
A: Following are the appropriate answers of the given questions.
Q: AH°F (kJ/mol) S (J/K-mol) Substance Охуgen 205.0 O218) 69.91 H2O) -285.83 Sulfur 31.88 Sulfur(s,…
A: (a) 2S (s, rhombic) + 3O2(g)-----> 2SO3(g) (b) S (s, rhombic) + O2(g)-----> SO2(g) (c) 2SO3…
Q: NO3 ball & stick labels Answer Retry Entire Group No more group attempts remain
A: Molecular geometry of given compounds are:
Q: Instruction: Find the following AH for the reaction below. 1. 2SO2 + 2P + 5C12 → 2SOCI2 + 2POCI3 AH…
A: Hess's law of heat summation states that for a chemical equation that can be written as the sum of…
Q: Data Table Trial 2 54.1mL Hal 12.3173 unknown 20.9°C HoO bufoe Trial 1 49.7mL HaO miŚ of HaO massh…
A: This question is related to finding out the specific heat of the metal. That can be solved by using…
Q: From Experiment Correct Value % error AHdissolving |(kJ/mol) -24
A:
Q: Experiment Number 2 A(g) + B (8) Initial [A] (M) - 3 C (8) Initial [B] (M) Initial ra formatic C (M
A:
Q: CHEM 181 Experiment #5 Part B Calculate the approximate volume of vinegar to react with 25 mL of 0.1…
A: Molarity (M) = Numer of moles of soluteVolume of solution (in L)Number of moles can be calculated…
Q: H. Application: Choose which test/s does the following structure of compounds will give positive…
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: ZnSO4 + Cu → MgSO4 + Cu → FeCl3 + Cu → AgNO3 + Cu → HCl + Cu → CuSO4 + Zn → MgSO4 + Zn → FeCl3 +…
A: More reactive substance displaces less reactive element from its compound.
Q: Date Page Anouer each in 5-6 sentonces- G-l Compare tho buropenties of dolid and ligjuidee sech as…
A: The properties of solid (Salt) and liquid (water).
Q: 2. AClsle) → ACI3(e) + Clz{e) AH = ? Given: Aa(s) + 6CI2(e) → 4ACI3{g) AH = -2439 kJ 4ACIS(e) →…
A:
Q: Discussion: 1- How would you prepare a- 500 ml of 0.1 M HC1 ? b- 1 L of 0.9 N HC1 ? Note : atomic…
A: Given that, Molar mass of HCl = 36.5 g/mol Density of HCl = 1.18 g/mL %w/w HCl = 37%
Q: Hopefull
A: Acid: Acids give H+ ions in the solution , acid solutions are sour in taste , acid solution pH…
Q: Oiron-59 O phosphorus-32 Othallium-201 O iodine-131 Ofluorine-18 Submit Request Answer
A: Answer is iron-59 Radiotracer Fe-59 is used to study problems with spleen.
Q: What mass of HCl, in grams is neutralized by a dose of milk of magnesia containing 3.5g Mg(OH)2
A: The estimation of amount that gets used up or formed in the reaction is done by utilizing the…
Q: Pear deck Exercise uset#@ Hi(^2) + F (a2) K-3.5xio -4 10
A: Equilibrium constant k= [H+][F-]/[HF]
Q: 1) b Br. a The number of lines exhibited by hydrogen(s) a is The number of lines exhibited by…
A: Lines observed by perticular carbon atom attached hydrogen, due to splitting. The splitting is…
Q: 5. Judy's data plot has a lesser slope than Nancy's. Which student will have the more positive AH°…
A: The equation for Gibbs free energy is written in equation (1).The Gibbs free energy in equilibria is…
Q: Ksp for this compound? er should be entered with 3 significant figures; do not enter units; give…
A: It is a ACl2 type salt. So, Solubility equilibrium is as ACl2(s) ----> A2+(aq.) + 2Cl-(aq.)…
Q: sid ate X OWLV2 | Onlinet X al 112021 X C Ais*war…
A:
Q: proceed? 1. 2CaO(s)→2Ca(s)-Ogg) 2. CO(g)-3H.(g)=CH₂(g) + H₂O(g) 3. Na(g)+31(g) →2NH3(g) 4.…
A: According to bartleby guidelines we're supposed to answer first three subparts of a multipart…
Q: l Verizon 4:05 PM O 75% Question 4 of 7 Submit Formula Ksp BaF, 1.7 x 10-6 PbF2 3.6 x 10-8 SrF2 4.3…
A: Solubility equilibrium is a type of equilibrium that exists when a chemical compound in the solid…
Q: What is the Minimum Ni-S distance in alpha-NiS to 4 decimal places in Angstroms? What is the…
A: The bond distance is calculated by adding the covalent radius of both the atoms which are bonded.
Q: IReview I Co (Pages 420- 425) Section 10.4 while Part A A syringe containing 1.66 mL of oxygen gas…
A: According to Charles Law,
Q: <W5 Assignment 1: MyMastering Chapter 7 Homework Problem 7.56 Enhanced - with Feedback Part A S and…
A: Since, Numeric number present before the chemical compound is known as stoichiometry. Thus,
Q: I Review I Part A The molecular weight of methanol (CH3OH), rounded to one decimal place, is amu.…
A: Mass of a given molecule: is called molecular weight of that molecule. Different molecules of the…
Q: excess Na Ob. OH Oc. OH Od. CH e. none of the other answers
A: The question is based on the concept of organic reactions. We have to identify the product formed…
Q: POSSIBLE POINTS: 2 Using the following information, calculate the AHsnfor the reaction between…
A:
Q: Part C Calculate the average charge on arginine when pH = 9.25. (Hint : Find the average charge for…
A:
Q: 1st attempt Feedback Jl See Periodic Table O See Hint COAST T What is the energy associated with the…
A:
Q: . I want to convent wt% tool. %. If the ata alpha phage is 0.865 wt%., the density is 8.94 g/cm³,…
A: Wt% means weight percent which is sometimes written as w/w i.e. [ weight of solute/ weight of…
Q: TMS. Ng HO. HO, CHO excess LDA/TMSCI 1 then workup ŠIMog OHC Bro ŠIME, OTBS 4 3 2.
A: Organic reaction mechanism.
Q: RESULTS AND CALCULATIONS: RUN 2 RUN 1 PART A (density of H20 = 1.00 g/mL) 29tHoomL= LOng 1029 %3D…
A:
Q: -1 T :0: S :0 What is the overall charge on the structure? -3 -4 +2 Answer Bank +3 0 -2 +1 7 +4
A:
Q: 4th attempt Part 1 Ag (aq) + C (aq) →→→ AgCl(s) -33 J/K Part 2 CH₂CH₂CH3(g) +50₂(g) → 3CO₂(g) +…
A: Entropy is a state function. Standard entropy change is difference in standard entropy between…
Q: ▼ Part A Name the following: Fe[AICI6] O iron(II) hexachloroaluminum O iron(III) hexachloroaluminate…
A: Here we are required to name the complex
Q: nswer 4th part only Both the photos are sam
A: The naming of the compounds is done as per the IUPAC nomenclature. It provided a set of rules which…
Q: Nitrogen N2 (g) 191.6 r answer to the ne N2H4 (g) 95.4 159.4 238.5 N2H4 (I) 50.6 149.3 121.2 NH3 (g)…
A:
Q: moles of water produced in HCl(aq) + NaOH(aq) -> H2O(l) + NaCl(aq)?
A: This question can be solved using stoichiometry of reaction. Moles of water produced = moles of…
Q: Balance the following ionic Lewis structures by placing the appropriate coefficients. Leave a target…
A: The ionic compounds given are,
Q: Problem 3.104 - Enhanced - with Feedback 5 of 5 A mixture containing KCIO3, K2CO3, KHCO3, and KCl…
A: Given information, Total mass of reaction mixture = 100.0 g Mass of H2O produced = 1.90 g Mass of…
Q: Question 6 What is the actindy of Lo my of 124,("% = 1.7 KIo pars)? of 1.0
A: Here we are required to find the activity of 1mg of radioactive element .
Q: Exercises Complete the table below. Find the % of C in CO2 Find the % of H in H20 Find the % of Zn…
A: The percentage of an atom in the compound can be calculated from the below formula:% of atom or…
Complete these structures by adding electrons, in the form of dots, as needed.
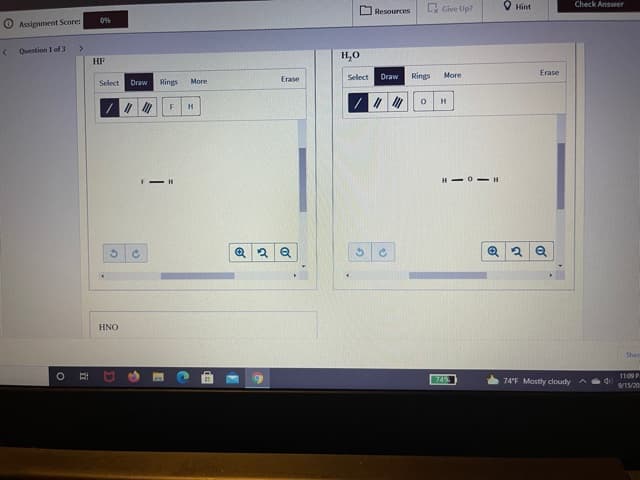

Step by step
Solved in 2 steps with 2 images

- Find oll doune The raction. 50g of CgCO wis racted. at Latm If Temp = 25C° and iatiel p = 0 inside reatim. v = 11. Au = q + w.What is ΔHsys for a reaction at 16.9 °C with ΔSsurr = -159 J mol-1 K-1 ? Express your answer in kJ mol-1 to at least two significant figures. (Please type answer no write by hend)Aleks data for PbCO3 is 7.40 x 10^-14.
- Which of the following will you use to find the Ka value for bromocresol green? Select one: a.The graph of Abs vs [H3O+]. b. A pipette. c. The value of pH. d. The Beer Lambert Law .(d0 9 with explanation )Which of the following is a spontaneous reaction.? a. Rxn with ΔH =- 10Kj/mol ΔS= -5J/mol T= 300K b. NaCl +H20 -> NaOH + HCl 25C c. H20(l) -> H2O(s) Temp: 25C d. Dissolution of 100g of solid sugar in 100 mL ice tea. Consider following reaction: HgO (s) -> Hg(l) + ½ O2 (g) Delta H = +90.7 kj/mol. What quantity of heat in kj/mol is required to produce one mole HgO? Write your answer without units. Given the following data 2ClF(g) + O2(g) --> Cl2O(g) + F2O (g) Delta H= 167.4 kJ I 2ClF3(g) + 2O2(g) --> Cl2O(g) + 3F2 O (g) Delta H= 341.4 kJ II 2 F2(g) + O2(g) ---> 2F2O (g) Delta H= -43.4 kJ III Calculate the delta H in kJ for below reaction: ClF(g) + F2(g) ---> ClF3(g)



