Q: From what you have learned about enols and the hydration of alkynes, predict what product is formed…
A:
Q: A. How many double bonds does ß-ocimene have? B. Is ß-ocimene conjugated, isolated or cumulated…
A:
Q: ) Propose a reasonable synthesis for the formation of nonane from CH3CH2CH2I and any other…
A: The question is based on the concept of organic reactions. we have to convert the reactants provided…
Q: Devise a stepwise synthesis of each compound from dicyclopentadiene using a Diels–Alder reaction as…
A: A stepwise synthesis of the compounds from dicyclopentadiene using a Diels–Alder reaction as one…
Q: an oxidizing agent KMNO4/H2SO4 and resulting product is treated with NH3. (i) Ethanal is oxidized by
A: In other words, the oxidising agent is a material that has the ability to oxidise other compounds to…
Q: Ibufenac, a para-disubstituted arene with the structure HO2CCH2C6H4CH2CH(CH3)2, is a much more…
A:
Q: Which carbonyl groups in the anticancer drug Taxol will undergo nucleophilic addition, and which…
A: The nucleophilic addition is a characteristic reaction of carbonyl compounds. It is sensitive…
Q: Which of the following set of reagents will yield a vicinal diol when reacted with an alkene? a…
A: ->Vicinal diol is Compound in which -OH group attached at adjacent carbons.
Q: Draw the structure of a hydrocarbon that reacts with 2 equivalents of H2 on catalytic hydrogenation…
A: Catalytic hydrogenation- H2 can be added across a double bond or triple bond in presence of…
Q: The ionones are fragrant substances present in the scent of iris and are used in perfume. A mixture…
A: Ionones are a group of compounds that are cyclic in nature and they have a specific aroma in their…
Q: Synthesize each compound from cyclohexanone and organic halides having s4 C's. You may use any other…
A: a)
Q: 4-Methylpyridine reacts with benzaldehyde (C6H5CHO) in the presence of base to form A. (a) Draw a…
A: (a) The step-wise mechanism for the formation of A is shown as follows:
Q: Devise a synthesis of each compound using CH3CH, as the only source of carbon atoms. You may use any…
A:
Q: An organic compound A (C2H3N) is used as a solvent of choice for many organic reactions because it…
A: Oxidizing agent is a substance that has the ability to oxidizes other substance. It is also known as…
Q: Devise a synthesis of each compound from benzene. You may use any other organic or inorganic…
A: a.
Q: Devise a synthesis of each compound from benzene. You may use any other organic or inorganic…
A: Benzene is an aromatic hydrocarbon with the molecular formula, C6H6. The molecule is aromatic since…
Q: Oximene and myrcene, two hydrocarbons isolated from alfalfa that have the molecular formula C10H16,…
A: The structure of 2,6-dimethyloctane is as follows:
Q: Draw the structure of a hydrocarbon that reacts with 2 equivalents of H2 on catalytic hydrogenation…
A:
Q: Atorvastatin (Lipitor) is used to decrease patient serum cholesterol levels. It works by inhibiting…
A:
Q: In addition to using CHX3 and base to synthesize dihalocarbenes (Section 26.4), dichlorocarbene…
A: The molecule contains neutral carbon-atom and if neutral C-atom carrying two valence electrons then…
Q: Draw a stepwise mechanism for the formation of A from an alcohol and acid chloride. A was converted…
A: Since the alcohol group is having lone pair electrons and hence can attack on electrophile. Since,…
Q: In addition to using CHX3 and base to synthesize dihalocarbenes, dichlorocarbene (:CCl2) can be…
A:
Q: Attenol A and pinnatoxin A are natural products isolated from marine sources. (a) Locate the…
A: The diether derivates of the aldehydes and ketones are known as acetals or ketals. The imine is a…
Q: (R)-Carvone, the major component of the oil of spearmint, undergoes acid-catalyzed isomerization to…
A: Acid-catalyzed isomerization of R carvone leads to the product carvacrol which is more stable than…
Q: oblem 19.22 Show how ethyl bromide can be used as a starting material in the preparation of each of…
A: Introduction: R-MgX is commonly known as Grignard reagent. It is used to prepare primary, secondary…
Q: An unknown compound (A) has a formula of C3H12. Treatment of A with H,/Pd- carbon gives B (CgH16).…
A:
Q: Propranolol, an antihypertensive agent used in the treatment of high blood pressure, can be prepared…
A:
Q: Starting with cyclohexanone, show how to prepare these compounds. In addition to the given starting…
A: Introduction: In the given conversion reaction firstly, cyclohexanone is reduced using sodium…
Q: Which compound will react fastest with aqueous acid (water with a small amount of H2SO4)? D. A, B…
A:
Q: What is the structure of cis-jasmone, a natural product isolated from jasmine owers, formed by…
A: Lindlar catalyst: The catalyst is used for the hydrogenation of alkenes in a syn manner. This means…
Q: Draw the organic products formed when cyclopentene is treated withfollowing reagent. [1] O3; [2]…
A: Cyclopentene reacts with many reagents to form different products. Cyclopentene can undergo many…
Q: غير مجاب علیه بعد سؤال 7 الدرجة من 2.00 Acetylene reacts with water in the presence of sulfuric acid…
A: Hydrocarbons are defined as organic compounds which are solely made up of carbon and hydrogen atoms.…
Q: What steps are needed to convert benzene into p-isobutylacetophenone, a synthetic intermediate used…
A: First we need to react the benzene with the below mentioned compound in presence of AlCl3 Than the…
Q: What is the structure of cis-jasmone, a natural product isolated from jasmine flowers, formed by…
A: Since the treatment of any alkyne with H2 in the presence of the Lindlar catalyst reduces the…
Q: b) Arrange the following compounds according to the reactivity towards nitration. Give reasons for…
A: Given compounds are :
Q: Draw a stepwise mechanism for the nitration of a benzene ring.
A: A Stepwise mechanism for the nitration of benzene ring has to be given.
Q: What is the structure of cis-jasmone, a natural product isolated from jasmine flowers, formed by…
A: When an alkyne is treated with the lindlar catalyst to give alkene, this reaction is called the…
Q: What is the structure of cis-jasmone, a natural product isolated from jasmine flowers, formed by…
A: What is the structure of cis-jasmone, a natural product isolated from jasmine flowers, formed by…
Q: 2. How many different ß-hydroxyaldehydes and ß-hydroxyketones, including constitutional isomers and…
A:
Q: Draw a stepwise mechanism for the formation of A from an alcohol and acid chloride. A was converted…
A: A species with a larger size can easily accommodate negative charge. In other words, in a large size…
Q: A dialkyl-substituted benzene, C14H22, is treated with basic potassium permanganate, followed by…
A:
Q: (a) Draw the structure of a compound of molecular formula C6H10 that reacts with H2 in the presence…
A: H2 in the presence of Pd-C reacts with alkenes or alkynes to reduce them to alkanes or alkenes…
Q: The reaction between 2-methyl-2-pentanol and sulfuric acid to yield 2-methyl-2-pentene goes via a(n)…
A:
Q: Alkynes generally undergo nucleophilic addition reactions. Ozonolysis of alkynes with O3, Zn and…
A:
Q: (R)-Carvone, the major component of the oil of spearmint, undergoes acid-catalyzed isomerization to…
A: The protonation of (R)- carvone is show below.
Q: Arrange the following alkyl halide in order of increasing E1/ E2reactivity:
A: Elimination reaction occurs either via E1 mechanism or E2 mechanism. E1 mechanism occurs via 2 step…
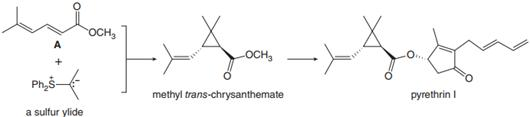
Q: Sulfur ylides, like the phosphorus ylides, are usefulintermediates in organic synthesis. Methyl…
A: Introduction: The ion containing -ve charge over C-atom as well as a +ve charge over the heteroatom…
Q: (vi) Ethoxyethane is hydrolyzed to produce a compound A which dehydration produces B. B on…
A:
Q: Draw the product formed when C6H5N2+Cl− reacts with each compound.
A: Diazonium salt is formed by the reaction of aniline with NaNO2 in the presence of HCl and this…
Sulfur ylides, like the phosphorus ylides of Chapter 21, are useful intermediates in
Step by step
Solved in 2 steps with 3 images

- 1. Complete the following diagram so that it represents (R)-2-bromobutane.Determine the Anti-Markovnikov Product prop-1-ene + H2O—> _________ (For Alkene and Alkyne Naming, follow this format “Branch-Carbon Prefix-Functional Group Suffix”) e.g. hex-2-eneThe methyl groups can be described as: A) both axial B) both equitorial C) one axial one equitorial
- Why (a) is cis 3,4dimethylhex2ene? Why (b) is trans 6 Methylhept3ene?What is a molecule that fits these three criteria: It should have a molar mass between 180 and 280 g/mol, contain a substituted cyclohexane like e. g. 5,6-dihydroxy-bicyclo[2.2.2]octane-2-carboxylic acid methyl ester, and be optically active.is diketone cyclohexadiene antiaromatic or nonaromatic?
- How many products of 2-hexene+Cl2 are thereDraw and name a constitutional isomer of C9H18 that has cis/trans isomers and is named a “3-hexene”. (Label your structure with the proper E/Z nomenclature).Can someone help me estimate the percentage of ethylcyclohexane molecules that have their substituents in an axial orientation at room temperature? I know how to draw a ethylcyclohexane molecule but I don't know how to go about this problem.

