Grade an Module 3: Law of Direction: Read the Importance of Chemical Reaction in our body. Then answer the we questions below. All the time, our bodies are carrying out two processes: storing the energy that reactions. A chemical reaction changes one set of chemicals into another. A series of take in as food and spending that energy. Our bodies do this by creating chemical chemical reactions turn the food we eat into energy that can be used by our cells. Other series of reactions spend that energy by building cells, growing, and exercising. Six chemical elements make up 99% of the human body: oxygen, carbon, hydrogen. in the body: carbohydrates, lipids, nucleic acids, and proteins. These molecules form nitrogen, calcium, and phosphorus. These elements form the key types of molecules all our cells and carry out most of the chemical processes needed for life. https://www.q-files.com/life/human-body/body-chemistry#: 1. If there were no chemical reactions in the human body, would life be possible? Why or why not? How long can we survive without any chemical reactions happening in our bodies? 2. The reactants can change to the products, but the products cannot change back to t reactants. These reactions are called irreversible. Why are the chemical reactions in body regularly irreversible? There are trillions of chemical reactions that happen simultaneously in the body. Can. you name at least one chemical reaction in the body and explain its importance.
Grade an Module 3: Law of Direction: Read the Importance of Chemical Reaction in our body. Then answer the we questions below. All the time, our bodies are carrying out two processes: storing the energy that reactions. A chemical reaction changes one set of chemicals into another. A series of take in as food and spending that energy. Our bodies do this by creating chemical chemical reactions turn the food we eat into energy that can be used by our cells. Other series of reactions spend that energy by building cells, growing, and exercising. Six chemical elements make up 99% of the human body: oxygen, carbon, hydrogen. in the body: carbohydrates, lipids, nucleic acids, and proteins. These molecules form nitrogen, calcium, and phosphorus. These elements form the key types of molecules all our cells and carry out most of the chemical processes needed for life. https://www.q-files.com/life/human-body/body-chemistry#: 1. If there were no chemical reactions in the human body, would life be possible? Why or why not? How long can we survive without any chemical reactions happening in our bodies? 2. The reactants can change to the products, but the products cannot change back to t reactants. These reactions are called irreversible. Why are the chemical reactions in body regularly irreversible? There are trillions of chemical reactions that happen simultaneously in the body. Can. you name at least one chemical reaction in the body and explain its importance.
Chapter6: The States Of Matter
Section: Chapter Questions
Problem 6.78E
Related questions
Question

Transcribed Image Text:Name:
Parent's Signa
Grade and Section:
WRITTEN WORK NO. 3
Module 3: Law of Conservation of Mass
questions below.
Direction: Read the Importance of Chemical Reaction in our body. Then answer the
All the time, our bodies are carrying out two processes: storing the energy that we
reactions. A chemical reaction changes one set of chemicals into another. A series of
take in as food and spending that energy. Our bodies do this by creating chemical
chemical reactions turn the food we eat into energy that can be used by our cells.
Other series of reactions spend that energy by building cells, growing, and exercising.
in the body: carbohydrates, lipids, nucleic acids, and proteins. These molecules form
nitrogen, calcium, and phosphorus. These elements form the key types of molecules
Six chemical elements make up 99% of the human body: oxygen, carbon, hydrogen,
all our cells and carry out most of the chemical processes needed for life.
https://www.q-files.com/life/human-body/body-chemistry#:
1. If there were no chemical reactions in the human body, would life be possible? Why or
why not? How long can we survive without any chemical reactions happening in our
bodies?
2. The reactants can change to the products, but the products cannot change back to the
reactants. These reactions are called irreversible. Why are the chemical reactions in the
body regularly irreversible?
3. There are trillions of chemical reactions that happen simultaneously in the body. Can
you name at least one chemical reaction in the body and explain its importance.
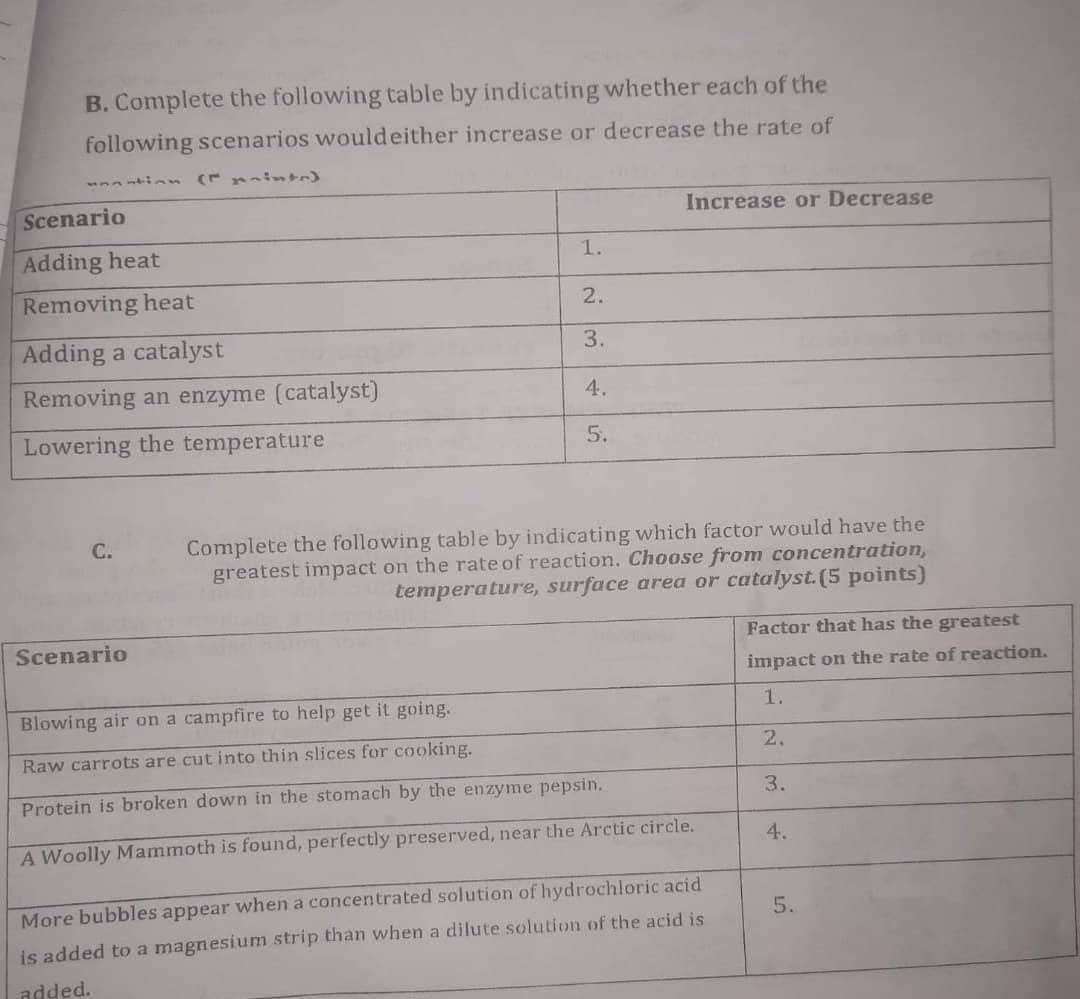
Transcribed Image Text:B. Complete the following table by indicating whether each of the
following scenarios would either increase or decrease the rate of
Scenario
Increase or Decrease
Adding heat
1.
Removing heat
2.
Adding a catalyst
3.
Removing an enzyme (catalyst)
4.
Lowering the temperature
5.
C.
Complete the following table by indicating which factor would have the
greatest impact on the rate of reaction. Choose from concentration,
temperature, surface area or catalyst. (5 points)
Scenario
Blowing air on a campfire to help get it going.
Raw carrots are cut into thin slices for cooking.
Protein is broken down in the stomach by the enzyme pepsin.
A Woolly Mammoth is found, perfectly preserved, near the Arctic circle.
More bubbles appear when a concentrated solution of hydrochloric acid
is added to a magnesium strip than when a dilute solution of the acid is
added.
Factor that has the greatest
impact on the rate of reaction.
1.
2.
3.
4.
5.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax



Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
