Q: Write the net ionic equation for the precipitation of magnesium carbonate from an aqueous solution: ...
A: Net ionic equations should be balanced by both electronic charge and mass. Balancing by mass means m...
Q: Given the following: pH = 1.88, calculate the [H3O+](note: Please use 3 sig figs. in your answer and...
A: All the required calculation to find out concertration of hydronium ion is provided in a attached im...
Q: Rank the following compounds in order of increasing frequency of the C=O absorption in their IR spec...
A: IR frequency of carbonyl group is present in the range of 1710 cm-1 and it varies depending upon gro...
Q: What is the full electron configuration of Mg?
A:
Q: A student ran the following reaction in the laboratory at 1098 K:2SO3(g) 2SO2(g) + O2(g)When she int...
A: Initial concentration of SO3= 8.35×10-2 moles ÷ 1L = 8.35×10-2 M.
Q: You mix 208 ml of 0.291 M LIOH with 208 ml of 0.536 M HF in a coffee-cup calorimeter, and the T of t...
A: Calorimeters are used for the determination of heat of reaction. For a coffee-cup calorimeter the en...
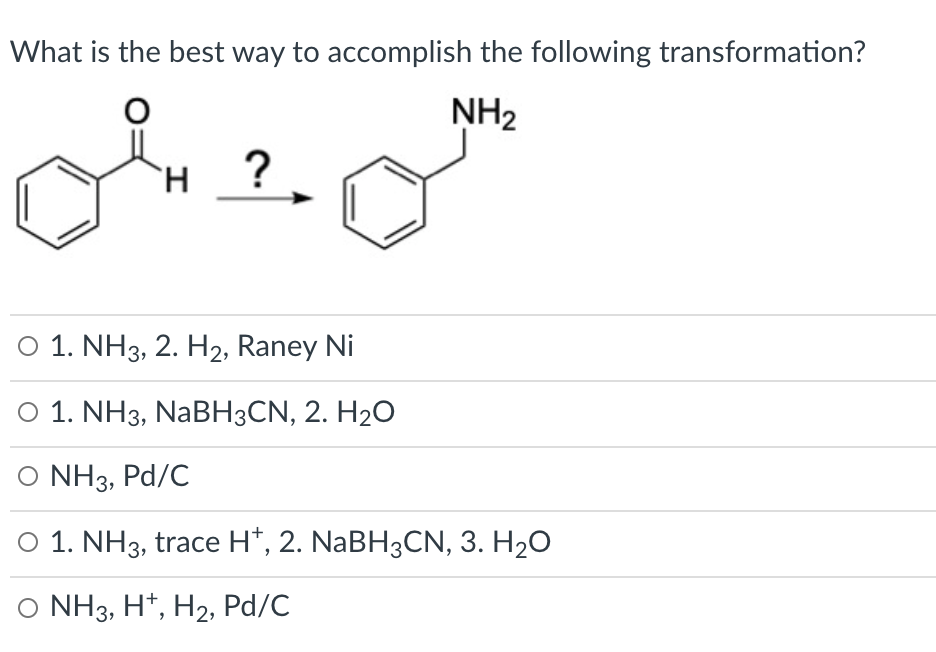
Q: 1. Propose a mechanism and predict the product. NaBH4 H. МеОн
A: The reducing agents, like NaBH4 and LiAlH4, react with the compound containing carbonyl group (aldeh...
Q: Which of the following are important stabilizations for the structure of collagen? (check all that a...
A: Collagen is a rod-shaped molecule, consisting of three helical polypeptide chains coiled around one ...
Q: Consider the following balanced equation: 2 H2 + O2 → 2 H2OWhat mass of water (H2O) will be collecte...
A: The provided balanced equation: 2 H2 + O2 → 2 H2O 2 mol 1 mol 2mol Molar Mass...
Q: Using your knowledge of buffer systems , calculate the pH of blood with its carbonic acid buffer sys...
A: H2CO3 ↔ H+ + HCO3- 0.0024M ...
Q: Arrange these compounds in order of increasing magnitude oflattice energy: KCl, SrO, RbBr, CaO.
A: Lattice energy is energy required for formation ionic compounds through ionic bond .The heat for for...
Q: What is the relationship between these compounds? H OH HO H- HO H- HO- H- HO- HO H но- - H- HO- HO C...
A: Enantiomers: Optical isomers which are non-superimposible mirror images of each other. They have opp...
Q: Draw a stepwise mechanism for the conversion of lactone A to ester Busing HCl in ethanol. B is conve...
A:
Q: Using cyclooctyne as your starting material, show how you would synthesize the following compounds. ...
A: Given compound is: Cyclooctyne. Required product from cyclooctyne is: Cyclooctanone.
Q: Given the following: [H3O+] = 6.330 x 10-5, calculate the pH: (note: for pH always please use 3 sig...
A: pH = - log[H3O+] = - log(6.330×10-5) =5-log(6.330) = 4.20
Q: Draw a stepwise mechanism for the following imine hydrolysis.
A: The hydrolysis of imine leads to the formation of corresponding amine and a carbonyl-containing comp...
Q: Provide the systematic name for the following molecule. -CH
A:
Q: Provide the name of the compound below using either systematic or common nomenclature. A derived nam...
A:
Q: How many nanometers are in 1.33 centimeters? Note: 1 meter = 100 centimeters and 1 meter = 1a 10° nm
A:
Q: calculate the molar solubility of AgBr in 0.10 M NaBr solution.
A: Solubility is defined as the amount of solute dissolved in the particular amount of solvent at a cer...
Q: Predict the sign of AS° and calculate AS° for each of the following reactions: a. H2(g) + V½ 02(g) →...
A: Entropy is randomness of any state. Entropy order of various forms of states in increasing order:- S...
Q: Determine whether HI can dissolve metal sample. If it can, write a balanced chemical reaction showi...
A: The standard reduction potential determines the direction of a reaction. The standard reduction pot...
Q: What is the mass of copper and the mass of chlorine sample?
A: Given that, Mass of hydrated sample, m1 = 1.02 g Mass of dehydrated sample, m2 = 0.91 g Mass of wat...
Q: At 298 K a cell reaction has a standard cell potential of +0.17 V. The equilibrium constant for the ...
A: Given: T = Temperature in K = 298 K E = standard emf = 0.17 V K = equilibrium constant = 5.5 X 105 T...
Q: Provide the name of the compound below using either systematic or common nomenclature.
A:
Q: Gaseous ethane (CH,CH) reacts with gaseous oxygen gas (02) to produce gaseous carbon dioxide (CO,) a...
A:
Q: The enthalpy of vaporization of chlorine at the standard (1 atm) boiling point (284 K) is 20.41 kJ/m...
A:
Q: A protein structure is solely determined by the sequence of amino acids in the primary structure. Su...
A: The protein forms from the polypeptide chain after large amount of primary and secondary interaction...
Q: Please calculate the pH of a buffer solution made up of citric acid and potassium citrate. The solut...
A:
Q: Consider the following balanced equation: 2 H2 + O2 → 2 H2OWhat mass of water (H2O) will be collecte...
A: From the given data,it is clear that 2 mol H2 +1 mol O2 → 2mol H2O we know that ,1 mol of a compoun...
Q: Decide whether each pair of elements in the table below will form an ionic compound. If they will, w...
A: The required answer for the completion of above table is provided in a attached image.
Q: A synthesis reaction takes place when carbon monoxide (CO) and hydrogen gas (H2) react to form metha...
A:
Q: The following reaction shows sodium hydroxide reacting with sulfuric acid.4NaOH + 2H2SO4 → 2Na2SO4 +...
A:
Q: Identify the compound that does NOT have dipole-dipole forces as its strongest force. Choose from on...
A: the correct ans is D
Q: A pill that is 91.5% aspirin is found to contain 50.21 mg of aspirin. What is the mass of the pill i...
A: % w/w = (mass of a component/total mass) × 100%
Q: Question attached
A: Significant figures are the digits of value which carry meaning towards the resolution. They represe...
Q: Suppose that a metal oxide of formula M2O3 were soluble inwater. What would be the major product or ...
A: Given- Metal oxide formula- M2O3
Q: 2518-25 General, Settings Help Discussions List > View Topic Gas_Laws Cho 7 Unlocked: Monday, Februa...
A: In the given question we have to calculate temperature after change in pressure and volume of gas, b...
Q: In the following reaction, oxygen is the excess reactant.SiCl4 + O2 → SiO2 + Cl2The table shows an e...
A: The given reaction is, SiCl4 + O2 -----> SiO2 +Cl2 Molar mass of SiCl4 = 28+(35.5×4)=170 g/mol. M...
Q: 3) Complete the following chemical equations. Make sure to clearly indicate the stereochemistry in e...
A:
Q: For each of the following unbalanced equations, indicate how many moles of the second reactant would...
A:
Q: If all of the SCN−SCN− was not converted completely to FeNCS2+FeNCS2+ when the calibration curve was...
A: Fe3+(aq) + SCN- (aq) <------------> FeNCS2+ (aq) Keq = [FeNCS2+ ]/([Fe3+ ] [SCN- ])
Q: When would Zn metal is put into an aqueous solution of CuSO4, solid Cu metal and a solution of ZnSO4...
A: A single replacement reaction, sometimes called a single displacement reaction, is a reaction in whi...
Q: Answer clearly and quick
A: Given that, At 25°C, Volume of water in the flask = 250 mL Volume of octanol in the flask = 200 mL V...
Q: Q# 20. Complete the table by providing the structural formulas of the organic product of the reactio...
A: (a) Reaction a is the addition of water to the alkene. (b). Reaction b is the dehydration reaction o...
Q: Determine the formal charges on all the atoms in the fol- lowing Lewis diagrams. H-N=ö and H-ö=N Whi...
A: The formal charge on all the following structure
Q: represent the same element?
A: we know,In atom the number of protons and electrons are equal but different neutrons is possible.so ...
Q: K. 5.6 x 10 at 500 K for the dissociation of iodine molecules to iodine atoms. L(8) 2 I(s) A mixture...
A: Given: The concentration of I2 and I in the reaction mixture is 0.020 mol/L and 6.0×10-8 mol/L, resp...
Q: Write the net ionic equation for the precipitation of manganese (II) hydroxide from an aqueous solut...
A: Mn(OH)2 is an inorganic compound with one central metal atom and two hydroxyl ions. Mn has +2 oxidat...
Q: Decide whether each pair of elements in the table below will form an ionic compound. If they will, w...
A: Ionic compounds are formed by the combination of an electropositive and an electronegative element. ...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Which of the following reagent best accomplish this transformation below? a. BH3, THF, H2O2 b. NaNH2, NH3 c. H2SO4, H2O, HgSO4 d. H2, LindlarWhich of the following reagent best accomplish this transformation below? Which of the following reagent best accomplish this transformation below? (1st) HCl, (2nd) NaNH2, NH3 (1st)Br2, CCl4, (2nd)2 NaNH2, NH3 (1st) BH3, THF, (2nd) H2O2 (1st) H2, Lindlar, (2nd) H2O2Consider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:
- Please help. Questions in images. Solvent Rf of A Rf of B Rf of C 10% ethyl acetate-90% hexanes .12 .07 .01 20% ethyl acetate-80% hexanes .34 .29 .10 30% ethyl acetate-70% hexanes .53 .42 .22 40% ethyl acetate-60% hexanes .72 .61 .35Which of the following reagent best accomplish this transformation below? a. Li/NH3 b. H2SO4, H2O, HgSO4 c. BH3, THF d. potassium permanganateWhich set of reagents would be the bestchoice for the transformation shown below?A. 1. HgBr2/H2O 2. NaBH4B. Br2/H2OC. 1. Br2 2. H2OD. 1. Br2 2. NaOHE. 1. BH3 2. Br2 3. NaOH, H2O2, H2O
- Which of the following would accomplish the transformation below? a) 1. LiAlH4, 2. H3O+ b) 1. CH3MgBr, 2. H3O+ c) 1. LiAlH(OtBu)3, 2. H3O+ d) CH3OH, H+Please help me draw 9-Methoxy-2-phenethyl-4,4-diphenyl-4H-naphtho[2,3- d][1,3]dioxine, preferably in ChemDraw7. Choose the best reagent from the list below for carrying out each transformation. Place the letter of the reagent in the blank to the left of the reaction. (see attached screenshot). a. 1. O3 2. Zn, H3O+ b. 1. BH3, THF 2. H2O2, NaOH, H2O c. H2O, H2SO4, heat d. 1. OsO4 2. NaHSO3, H2O e. KMnO4, acid f. 1. Hg(OAc)2, H2O 2. NaBH4
- 1. Which of the following is the strongest nucleophile? Refer to the diagram below with an MeOH, MeSH, etc. 2. Which of the following is true about the stereochemistry of SN1 reaction?a.retention of configuration at the electrophilic centerb.50:50 mixture of retention and inversion of configuration at the electrophilic centerc.inversion of configuration at the electrophilic centerd. slightly more inversion than retention at the electrophilic center1-ethylcycloheptene + cold, dilute KmnO4What is the locant of the methoxy substituent? 4-methoxy 1-methoxy 5-methoxy 3-methoxy



