Q: give the IUPAC name of the fllowing amine
A: 1. 3 degree Amine , so we used here N,N .
Q: Classify each amine in the following compounds as 1°, 2°, or 3°. NH2 a. H,N b. CH,CH20. N-CH,…
A: NH3 is ammonia R-NH2 is primary amine i.e. N attached to one alkyl and two hydrogen atoms forms a…
Q: 13. Which of the following structures contains a secondary amine? NH2 `NH `NH2 II II IV А) I B) II…
A: To Solve this problem first we have to know about primary, secondary and tertiary amines .
Q: Use this amine to answer the following questions: A) Is this amine primary, secondary or tertiary?…
A: (a) Is this amine primary, secondary or tertiary? (b) Draw the product when this amine reacts with…
Q: Which of the following compounds is not a heterocyclic amine? a. pyridine O b. imidazole O c. purine…
A:
Q: Amines in aqueous solutions are less reactive than amines in gaseous solutions. Select one: O True O…
A: Amines are Lewis’s base due to the presence of a lone pair of electrons on the nitrogen atom of…
Q: ethylamine NABH,CN H;C One problem with reductive amination as a method of amine synthesis, is that…
A: Here electrophile is benzaldehyde which is first activate by the acid and then it is attack by the…
Q: 1. Rank the following amines A-D in order of decreasing basicity, where 1 is the most basic. NO2 A…
A: The compounds given are,
Q: NH, M IZ IZ
A:
Q: Arrange the amines in order of increasing boiling point (lowest first). methylamine I ethylamine II…
A:
Q: H;C. CH3 'N' 1 2 3 4
A: Basicity of amines:
Q: Nor HN N. A OH F There are two nitrogen atoms highlighted and labelled A and B. Match the…
A:
Q: Why are tertiary amines not soluble in water? O a. Primary amine cannot hydrogen bond with water. O…
A: Correct Answer : Option C "Tertiary amine cannot form hydrogen bond with water".
Q: Which of the following amines are derivatives of 2-phenylethylamine? Label the atoms of…
A: The derivatives of the 2-phenyl ethyl amine are the compounds obtained from this compound. 2-phenyl…
Q: Predict the major products formed when the following amines undergo exhaustivemethylation, treatment…
A: The formation of major product, when the given amine undergoes exhaustive methylation, treatment…
Q: WHich of the following molecules are all amines based off? H20 CO2 NO3 NH3
A: In this question, we will see that which molecules are all amines based off. You can see details…
Q: Draw the structure of the following three isomeric amides with chemical formula CgH1¡NO. Amide #1:…
A: Isomers are defined as compounds having the same molecular formula but have a difference in the…
Q: What type(s) of amines can be made by the reduction of nitriles? You may choose more than one…
A: The species which have tendency to donate the electron to another species is known as reducing…
Q: be used to prepare following amine by reductive amination? NH2 NH2 NH2 NH2 (III) (IV) (I) (II)
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: 2. Which of the following is a tertiary amine. Encircle the substituents that make it tertiary. CH3…
A:
Q: Give a systematic name and a common name (if it has one) for each of the following amines and…
A: An amine is generally a functional group with a nitrogen atom having a lone pair. Amines resemble…
Q: Classify and describe the properties of the following nitrogen containing compound. C/O Classify the…
A:
Q: ement best described the ability of amines to hydrogen bond? A. Primary, secondary and…
A: B.Primary and secondary amines can hydrogen bond with molecules identical to themselves, but…
Q: Explain why the boiling point of propanamide, CH3CH2CONH2, is considerably higher than the boiling…
A: Physical properties like boiling point depend upon the molecular mass and intermolecular hydrogen…
Q: The compounds below can undergo reduction to form a primary amine EXCEPT for what compound? A. NO₂…
A:
Q: What amide(s) form amine X on treatment with LiAlH4?
A: General reaction for the conversion of amide to amine can be given by :…
Q: Classify each of the following amines as primary, secondary or tertiary. CH₂CH3 V CH₂CH₂NCH₂CH3 CH3…
A: The classification of amine is done on the basis of the number of alkyl groups attached to the…
Q: What amide(s) form amine X on treatment with LiAlH4?
A: General reaction for the conversion of amide to amine can be given by :…
Q: Arrange the amines in order of increasing boiling point (lowest first). methylamine I ethylamine II…
A: Greater is the intermolecular force, higher will be the boiling point. Greater is the molecular…
Q: What is the name of the major product of the following synthetic scheme? OH 1. CH3CH2NH2, 275 °C 2.…
A:
Q: You are trying to identify an unknown amine from a list of potential candidates. You have the…
A: Ammonia is a basic compound and reacts with acid. In the presence of acid like HCl it will react and…
Q: 5. Write an equation that would produce the following products a) „NH2 b) NH2 c) Classify each of…
A: Here we have to write the reactants that reacts to produce the target products and to find primary,…
Q: Classify each of the following amines as (1) primary, secondary or tertiary and as (2) aliphatic or…
A: 1) It is a primary or 1° in which one H- atom of NH3 is replaced by an alkyl group. It is an…
Q: alai 4 ich is Ammonia Acetic acid HCI NaOH Lläs 4 * N,N-Dimethylbutylamine is Symmetrical tertiary…
A: The answer to the following questions are as follows:
Q: 5. Write an equation that would produce the following products a) 0- NH2 b) `NH2 NH4 c) Classify…
A: Primary amine type is RNH2.. Secondary amine type is RNHR.. Tertiary amine is R3N...
Q: A tertiary amine is more basic compared to a secondary amine. (A) True B False
A: Amines are Lewis base due to the presence of a lone pair of electrons on the nitrogen atom of amine.
Q: hech of the fo llowing Compeund ở a Sz Secendary eromatic amine? CH3 NH2
A: The factors that confer aromaticity to an organic molecule are 1) The molecule should be having a…
Q: Which of the following statements is INCORRECT? a. Amines are electrophilic. Amines are stronger…
A:
Q: Edit H;C `NH2 (Structure 1) One of the two primary amines which contain only primary, secondary and…
A: The organic compounds are named as per IUPAC rules. IUPAC rules provide a unique name to an organic…
Q: Draw structural formulas for three isomer of C,H,N, and classify them as primary, secondary, or…
A:
Q: Identify the following as a primary (P), secondary (S), tertiary (T), or quaternary (Q) amines „NH2…
A: Primary amine is in which 1 carbon attached with nitrogen Secondary amine is in which 2 carbon…
Q: 1. Give the names of the following amines and classify them as primary, secondary or tertiary: a)…
A: In organic chemistry there are many functional group that show various properties in the organic…
Q: What is the common name of the amine show? (do not include any spaces) CH3-CH2-NCH2-CH3 CH3 What is…
A:
Q: NH2 CH;NH2 (CH3)2NH (CH:);N
A: Basicity can be defined as tendency to donate unshared pair/Lone pair of electrons present on the…
Q: "hich of the following is a tertiary amine? O CH3CH2N(CH3)2 (CH3)3CNH2 O CH3CH2NHCH3 O…
A:
Q: STATEMENTS TRUE FALSE 1. Butylamine is liquid at room temperature. 2. Nitrogen has the ability to…
A: 1.Butyl amine is liquid at room temperature. True (As it has strong interactions: H-bonding)
Q: Synthadotin is a promising anticancer drug in clinical trials. a.Identify the functional groups.…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: NHCH3
A: In NH3, when one of the H-atom is replaced by carbon atom then the amine formed will be Primary.…
Q: Classify each amine as primary, secondary, or tertiary and as aliphatic or aromatic
A: In Benzocaine, a primary aromatic amine is present.
Q: Predict the major products formed when the following amines undergo exhaustivemethylation, treatment…
A: Elimination reactions: Organic reactions contain the removal of a group of atoms from a molecule by…
How do we convert the compound below into a quaternary
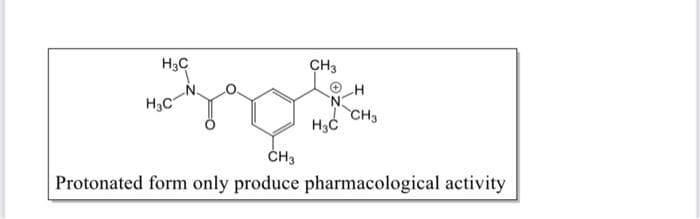
Step by step
Solved in 3 steps with 2 images

- 1. ) 2-Pentanol when introduced with H2SO4 will become ____________ . H2SO4 is a Dehydrating agent (used the concept of Zaitsev to come up with the major product)1-Pentene 1-Pentanal2-Pentene2- Pentanone2.) Diabetic breath has a mild sweet odor because of AroamticAldehydeAlkeneKetone3.) The following compounds contain a Carbonyl group, exceptAcetaminophenCitric AcidPhenolDacroonprovide a synthesis pathway for the molecule (left) using the boxed reagents. show illustration if possible.A diuretic is a compound that causes increased urination and thereby reduces fluid volume in the body. An important use of diuretics in clinical medicine is in the reduction of the fluid buildup, particularly in the lungs, that is associated with congestive heart failure. It is also used as an antihypertensive (i.e., to reduce blood pressure). Furosemide, an exceptionally potent diuretic, is prescribed under 30 or more trade names, the best known of which is Lasix. The synthesis of furosemide begins with treatment of 2,4-dichlorobenzoic acid with chlorosulfonic acid in a reaction called chlorosulfonation. The product of this reaction is then treated with ammonia followed by heating with furfurylamine. Q. Propose a mechanism for the chlorosulfonation reaction in Step (1).
- A diuretic is a compound that causes increased urination and thereby reduces fluid volume in the body. An important use of diuretics in clinical medicine is in the reduction of the fluid buildup, particularly in the lungs, that is associated with congestive heart failure. It is also used as an antihypertensive (i.e., to reduce blood pressure). Furosemide, an exceptionally potent diuretic, is prescribed under 30 or more trade names, the best known of which is Lasix. The synthesis of furosemide begins with treatment of 2,4-dichlorobenzoic acid with chlorosulfonic acid in a reaction called chlorosulfonation. The product of this reaction is then treated with ammonia followed by heating with furfurylamine. Q. Propose a mechanism for Step (3).A diuretic is a compound that causes increased urination and thereby reduces fluid volume in the body. An important use of diuretics in clinical medicine is in the reduction of the fluid buildup, particularly in the lungs, that is associated with congestive heart failure. It is also used as an antihypertensive (i.e., to reduce blood pressure). Furosemide, an exceptionally potent diuretic, is prescribed under 30 or more trade names, the best known of which is Lasix. The synthesis of furosemide begins with treatment of 2,4-dichlorobenzoic acid with chlorosulfonic acid in a reaction called chlorosulfonation. The product of this reaction is then treated with ammonia followed by heating with furfurylamine. Q. Is furosemide chiral? If so, which of the possible stereoisomers are formed in this synthesis?Give the name (common name or IUPAC name) of intermediate product (a) in the following reaction.
- please answer with deatils and fill in blanksThe cyclic hemiacetal is more stable than the open-chain form, so very little of the open-chain form is present atequilibrium. Will an aqueous solution of glucose reduce Tollens reagent and give a positive Tollens test? Explain.Explain this difference in potency and speed of onset by pointing out the main differences in functional groups between morphine and heroin.

