Q: Which of these reagents cannot be used in transforming nitro containing compounds into amines? O…
A: The reagent SnCl2/HCl is a reducing reagent which helps in the reduction of the functional groups.…
Q: a. b. C. N-H+ HC1 N- -H + CH3I N-CH3 + HONO
A: Detail mechanistic pathway is given below to find out the product
Q: 5. Draw the starting material in preparation of the amines below: „NH2 (a) `NH2 (b)
A: 1-Aminobutane and 2-Phenylethan-1-amine are primary amine. 1-Aminobutane is colorless liquid. It has…
Q: Amines A. Predict the major product: CH3 Sn, HCI 1. O2N КОН `NO2 + H. 2. 0 °C phenol .NH, + HONO 3.…
A:
Q: Which amine below is an primary aromatic amine? a. N-methyl-2-phenylethanamine b. All of the given…
A: We have to choose the following given amine below is an primary aromatic amine as follows in step 2:
Q: Be sure to answer all parts. Be sure to include the ion charges. What products are formed when the…
A: A neutralization reaction occur when an acid and base react
Q: Question attached
A: Amines are the organic compound having -NH2 functional group. Amines are classified into three…
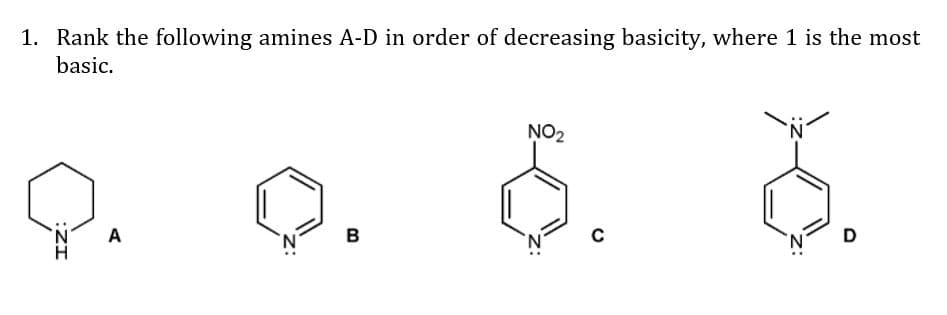
Q: Rank the amines in each set in order of increasing basicity. H ot (b)bims n N (a) H. NH2 NH2 (c) H H…
A: ***Note: Since you have posted a question with multiple subparts, we will solve the first three…
Q: Draw the products formed when each amine is treated with [1] CH3I (excess); [2] Ag:0; [3] A.…
A: Hofmann elimination is a reaction in which amines are eliminated from the molecule to form alkenes.…
Q: Oxytocin is a naturally occurring hormone that induces labor and stimulates milk production in…
A: The organic compound is formed by two main elements, carbon, and hydrogen. The specific group of…
Q: The Gabriel Amine Synthesis is a useful method to make primary amines. That said, which one of the…
A: The Gabriel synthesis is a chemical reaction that transforms primary alkyl halides into primary…
Q: 1. What amine and what carbonyl compound are required to make the following molecule:
A:
Q: Identify all amines below that can NOT be made by reduction of an amide by LAH. -NH2 NH NH2 (A) (B)…
A:
Q: Rank the following amides from greatest reactivity to least reactivity toward acid-catalyzed…
A: Given amides are represented as follows:
Q: Give a systematic name for each amide
A: (a) Systematic name: N-ethylmethanamide (b) Systematic name: N,N-dimethylbenzamide
Q: 1. Rank the following nitrogens or amines in order of decreasing basicity (strongest to weakest,…
A: In case of basicity only to check the availability of loan pair of electron on nitrogen atom when…
Q: The reaction shown below is specific for the preparation of: CH,N2 Ph он A. Amides B. Imines O C.…
A: In this question we have to tell the product of the reaction.
Q: Amines in aqueous solutions are less reactive than amines in gaseous solutions. Select one: O True O…
A: Amines are Lewis’s base due to the presence of a lone pair of electrons on the nitrogen atom of…
Q: Determine the hybridization around the N atom in each amine, andexplain why cyclohexanamine is 106…
A: Hybridization is a hypothetical concept which involves mixing of two or more pure orbitals. When…
Q: What is the name of the major product of the following synthetic scheme? OH 1. CH,CH,NH, 275 C 2.…
A: In this reaction acid amine coupling takes place which results in formation of amide bond which…
Q: Rank the following amides from greatest reactivity to least reactivity toward acid-catalyzed…
A:
Q: H;C. CH3 'N' 1 2 3 4
A: Basicity of amines:
Q: a. b. С. (CH3CH2)2NH NH2
A: Amides can be reduced to amines on treatment with reducing agents like LiAlH4. RCONH2 + LiAlH4…
Q: Draw the products formed when each carbonyl compound reacts with the following amines: (1]…
A: The process is given below:
Q: show how you might prepare each of the following amines through reductive aminations…
A:
Q: Which of the N atoms are basic in ciprofloxacin? Explain A) N1 B.) N9 C.) N12
A: The basicity of a base depends on the availability of lone pairs. The higher the availability of…
Q: Rank the following amines in order of basicity.
A: The given compounds are: A. B.
Q: Why are tertiary amines more basic than primary and secondary amines? Because tertiary amines are…
A: Chemical compounds are classified on the basis of attached functional groups. The compounds…
Q: 1. Name this amine properly. Is it a primary, secondary, or tertiary amine? Which of the following…
A:
Q: Which of the following amines is more basic? Explain why. NH2 NH2 A B
A: An acid is a chemical that gives off the hydrogen ions in water and forms salts by combining with…
Q: NH NH2 NH2 WXY ZAB CDE FGH IJK least basic amine Choose... + most basic amine Choose... + most…
A:
Q: Draw the structures of the missing substances in each of the following reactions involving amides.…
A: The amide functional group is denoted as follows: R-CO-N(R1)(R2), Where R, R1, and R2 can be alkyl…
Q: Which of the following is the correct order of basicity of amines? :NH2 A B
A:
Q: ON 16 Which has the lowest boiling point? OA. CH3(CH2)5CONH2 O B. CH3CH2CON(CH2CH3)2 O C.…
A: The boiling point of a compound is determined based on the interaction among the molecule. The…
Q: Arrange the following amines in order of decreasing base strength. Rank from strongest to weakest…
A: We can see Basicity of amines according to stability of conjugate acids. ->As the electron…
Q: Question is attached
A: The amide functional group is denoted as R-CO-NH2, where R can be alky, aryl, or hydrogen. When the…
Q: How could you prepare the following amides using an acid chloride and an amine or ammonia? (a) CH…
A: We have to synthesize the given amides using an acid chloride and an amine or ammonia. The given…
Q: This question contains 3 parts. Indicate your answers clearly for each part. For amines A, B, and C…
A: Basicity of a nitrogenous base depends on the availability of l.p. present on nitrogen atom. Greater…
Q: N. carbonyl compound amine Select one: NH2 a. „NH2 O b. HN O d. + ZI
A: When a ketone or Aldehyde (carbonyl group containing Compound) is treated with amine , then imine is…
Q: What is the product of the following reductive amination? CH, CH, NHz mild acd Nafity or Na BH3CN…
A: This is an typical example of reductive amination. The details solution for this reaction is…
Q: What products are obtained when the following tertiary amines react with hydrogen peroxide followed…
A:
Q: Z-I Give an acceptable name for each amine. a. CH;CH,CHCH3 -N(CH3)2 C. NH2 H. b. CH3CH,CH,NHCH3 d.…
A: Note : As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost…
Q: 1. Give the names of the following amines and classify them as primary, secondary or tertiary: a)…
A: In organic chemistry there are many functional group that show various properties in the organic…
Q: O Which reagents can reduce amide? 1. LIAIHY 2. H20, H30* NOBH ETOH H2, Pd /CLIAIHO+-Bi); 78C
A:
Q: Which of the following compounds would form Azo-dye from primary aromatic amines? Indicate YES or NO…
A: Azo dye is a test to distinguish between an aromatic amine and aliphatic amine. Only aromatic amines…
Q: Select the best answer that identifies the compound(s) below that would furnish an amine on…
A: Following organic compounds will give amine when reacts with excess LiAlH4.
Q: How could you prepare the following amides using an acid chloride and an amine or ammonia? | (a)…
A: To find: The preparation method of the following amide from acid chloride and an amine or ammonia a)…
Q: Arrange the following amines in order of decreasing base strength. Rank from strongest to weakest…
A: Bases are electron donors. The molecule is more basic if it easily the donate electrons to the other…
Q: (b). Many amines are occurring naturally with a wide range of biological activity. For instances,…
A: Reductive amination is reaction in which carbonyl compounds are converted into amines via imine as…
Q: (a) NCH3 (b) NH2 -
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Rank the following amines in order of basicity from highest to lowest.Account for the following :(i) Primary amines (R-NH2) have higher boiling point than tertiary amines (R3N).(ii) Aniline does not undergo Friedel – Crafts reaction.(iii) (CH3)2NH is more basic than (CH3)3N in an aqueous solution.Arrange the following amines in order of increasing basicity. For example, an answer of 1234 would indicate that you think 1 is least basic and 4 is most basic
- Which of the following is the correct order of basicity of amines? A. B > C > AB. A > C > BC. B > A > CD. C > A > BE. A > B > CExplain how you would rank the amines given below according to their basicity, together with the reasons.(1) Which is the most basic amine and which is a secondary amine? (2) Which can undergo hydrolysis? (3) Which gives a diazonium salt upon reaction with HNO2, HCl at 0oC?
- Each pair of compounds below can be separatedusing acid/base extraction. Provide the common acid or base that could be used to effect the separation. Amines can be converted into ammonium salts using acid, but neutral amines are very weak acids (pKa~ 35)and thus cannot be extracted by an aqueous base.Which amine below is an primary aromatic amine? a. N-methyl-2-phenylethanamine b. All of the given c. 3-methylbenzamine d. N-ethylbenzamineRank each set of compounds in order of increasing basicity.aniline, p-methylaniline, p-nitroaniline

