HCl is added to the epoxide below. On which carbon (A-D) is Cl most likely to add to form the major organic product? 0 0000 A H₂C A B HC C CH₂ D
HCl is added to the epoxide below. On which carbon (A-D) is Cl most likely to add to form the major organic product? 0 0000 A H₂C A B HC C CH₂ D
Chapter11: Reactions Of Alkyl Halides: Nucleophilic Substitutions And Eliminations
Section11.5: Characteristics Of The Sn1 Reaction
Problem 12P: 3-Bromo-1-butene and 1-bromo-2-butene undergo SN1 reaction at nearly the same rate, even though one...
Related questions
Question
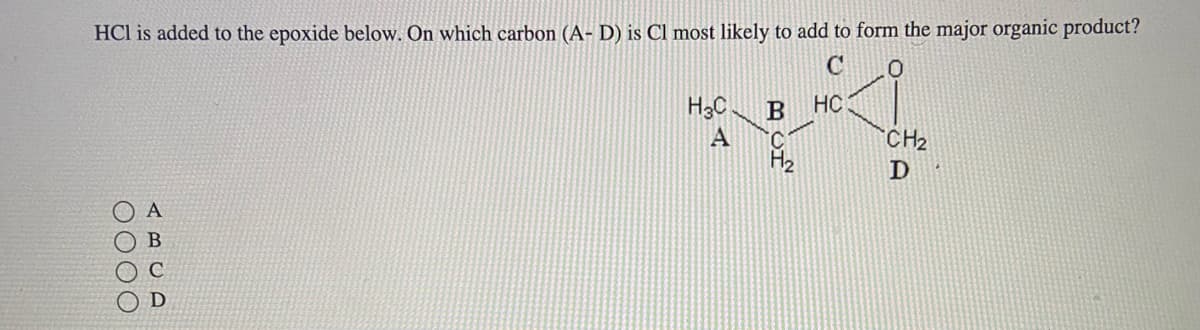
Transcribed Image Text:HCl is added to the epoxide below. On which carbon (A-D) is Cl most likely to add to form the major organic product?
0000
ABCD
ܐ .
C
H3C B HC
A C
CH₂
D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

