"he table and graph below display data from the Fresh 4 Less farm stand in A /19/2020 from https://data.world/cityofaustin/cfer-vyii. 4000 3500 3000 2500 2000 1500 1000 500 200 250 300 350 400 450 Number of Customers Number of Customers 216 254 316 397 Total Sales $1900 $1995 $2606 $3482 When answering the questions below, maintain exactly four decimal p Total Sales ($)
"he table and graph below display data from the Fresh 4 Less farm stand in A /19/2020 from https://data.world/cityofaustin/cfer-vyii. 4000 3500 3000 2500 2000 1500 1000 500 200 250 300 350 400 450 Number of Customers Number of Customers 216 254 316 397 Total Sales $1900 $1995 $2606 $3482 When answering the questions below, maintain exactly four decimal p Total Sales ($)
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter33: Automated Methods Of Analysis
Section: Chapter Questions
Problem 33.4QAP
Related questions
Question
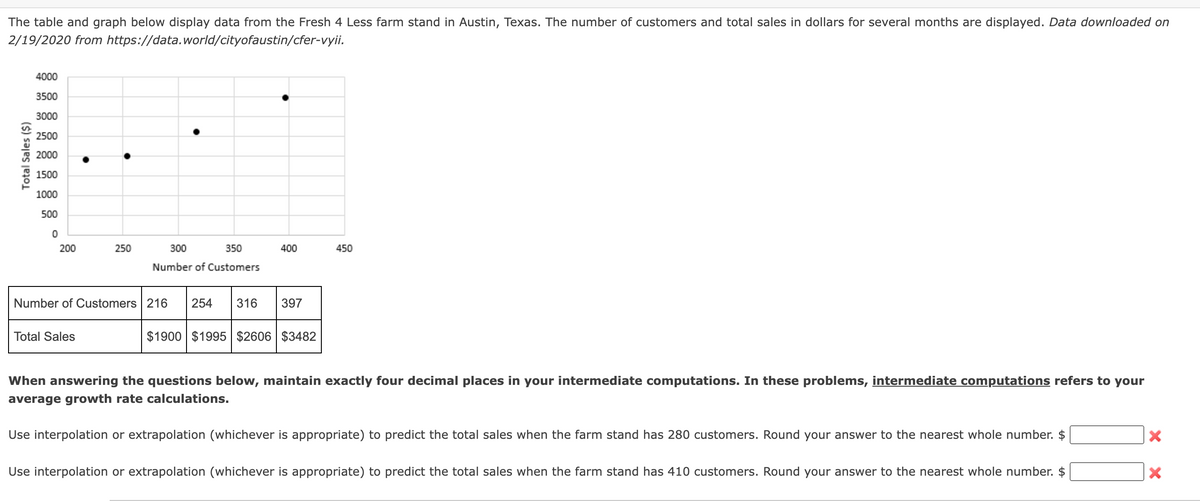
Transcribed Image Text:The table and graph below display data from the Fresh 4 Less farm stand in Austin, Texas. The number of customers and total sales in dollars for several months are displayed. Data downloaded on
2/19/2020 from https://data.world/cityofaustin/cfer-vyii.
4000
3500
3000
2500
2000
1500
1000
500
200
250
300
350
400
450
Number of Customers
Number of Customers 216
254
316
397
Total Sales
$1900 $1995 $2606 $3482
When answering the questions below, maintain exactly four decimal places in your intermediate computations. In these problems, intermediate computations refers to your
average growth rate calculations.
Use interpolation or extrapolation (whichever is appropriate) to predict the total sales when the farm stand has 280 customers. Round your answer to the nearest whole number. $
Use interpolation or extrapolation (whichever is appropriate) to predict the total sales when the farm stand has 410 customers. Round your answer to the nearest whole number. $
Total Sales ($)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning