Sp eSantaFe | Santa Fe College | Ga X SE This is the link to your ALEKS Ho X A ALEKS - Natalie Drury - Learn G A major component of gasoline x A www-awn.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IJgt8PIGunmDn7WsVrRAXK6XnHkiRvH2tl800vdbuH6lkCnssocifJxkN3ax2TWa6AXXqcyFaLE6ceOfbJICanm28fOOHz O CHEMICAL REACTIONS Solving for a reactant using a chemical equation Natalie A major component of gasoline is octane (C,H,8). When octane is burned in air, it chemically reacts with oxygen gas (0,) to produce carbon dioxide (CO,) 18 and water (H,0). What mass of water is produced by the reaction of 3.05 g of octane? Round your answer to 3 significant digits. x10 Explanation Check 2020 McGraw-Hill Education. All Rights Reserved, Terms of Use | Privac
Sp eSantaFe | Santa Fe College | Ga X SE This is the link to your ALEKS Ho X A ALEKS - Natalie Drury - Learn G A major component of gasoline x A www-awn.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IJgt8PIGunmDn7WsVrRAXK6XnHkiRvH2tl800vdbuH6lkCnssocifJxkN3ax2TWa6AXXqcyFaLE6ceOfbJICanm28fOOHz O CHEMICAL REACTIONS Solving for a reactant using a chemical equation Natalie A major component of gasoline is octane (C,H,8). When octane is burned in air, it chemically reacts with oxygen gas (0,) to produce carbon dioxide (CO,) 18 and water (H,0). What mass of water is produced by the reaction of 3.05 g of octane? Round your answer to 3 significant digits. x10 Explanation Check 2020 McGraw-Hill Education. All Rights Reserved, Terms of Use | Privac
Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
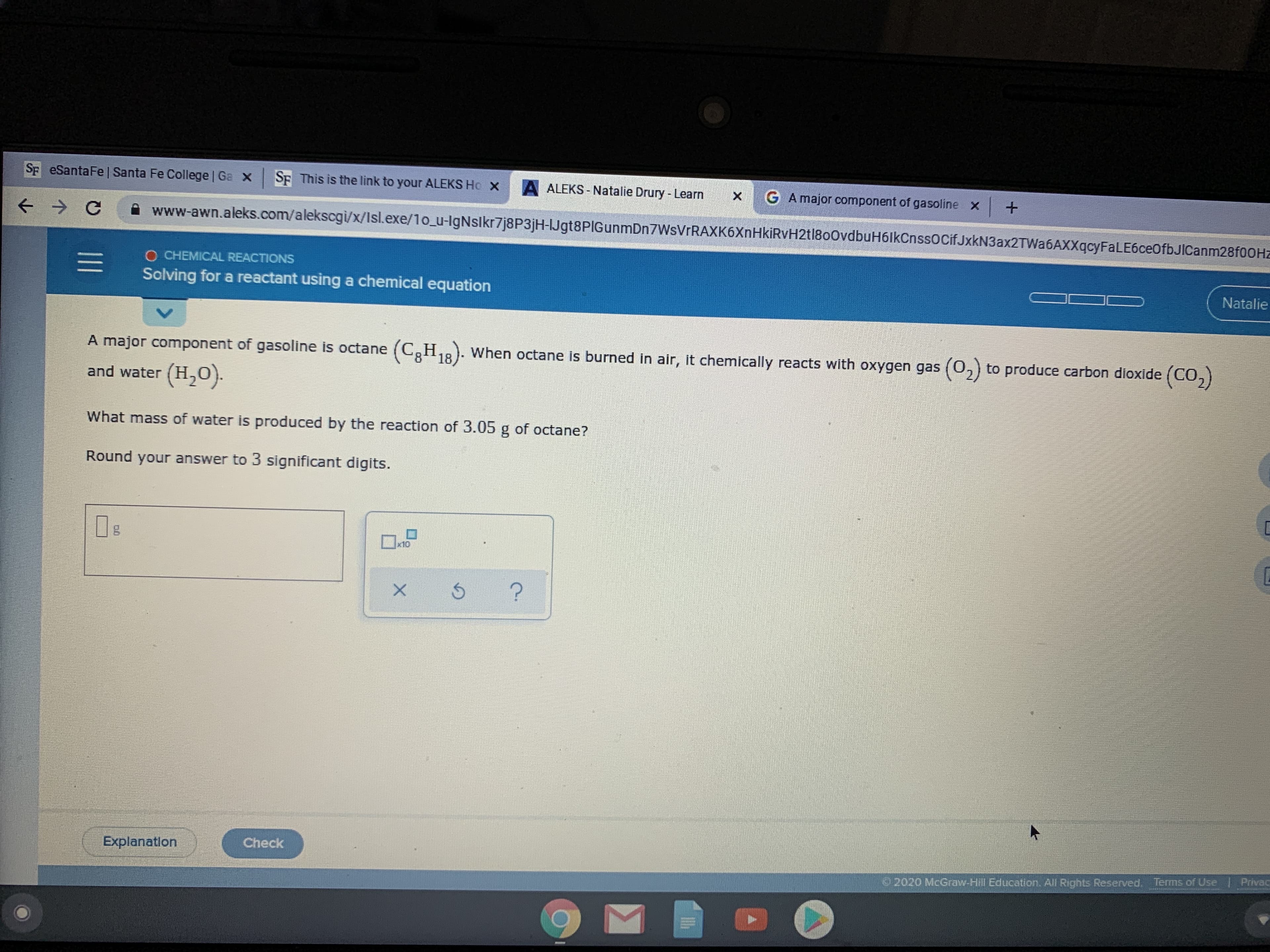
Transcribed Image Text:Sp eSantaFe | Santa Fe College | Ga X
SE This is the link to your ALEKS Ho X
A ALEKS - Natalie Drury - Learn
G A major component of gasoline x
A www-awn.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IJgt8PIGunmDn7WsVrRAXK6XnHkiRvH2tl800vdbuH6lkCnssocifJxkN3ax2TWa6AXXqcyFaLE6ceOfbJICanm28fOOHz
O CHEMICAL REACTIONS
Solving for a reactant using a chemical equation
Natalie
A major component of gasoline is octane (C,H,8). When octane is burned in air, it chemically reacts with oxygen gas (0,) to produce carbon dioxide (CO,)
18
and water (H,0).
What mass of water is produced by the reaction of 3.05 g of octane?
Round your answer to 3 significant digits.
x10
Explanation
Check
2020 McGraw-Hill Education. All Rights Reserved, Terms of Use | Privac
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning