Here is a graph of the molarity of hydrogen peroxide (H,02 in a reaction vessel during a certain chemical reaction. Use this graph to answer the questions in the table below. 0.3- 0.25- 0.209 0.170 0.15- 0.0947E 0.05 50 80.0 100 150 1810 250 300 seconds O created Is H,0, being created or destroyed by the chemical O destroyed reaction? O neither created nor destroyed Explanation Check O 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use I Privacy Acce PHED-T280-00T Spring 2021) gevor 20 stv MacBook Air DII DD F10 F9 F8 목 미 ×
Here is a graph of the molarity of hydrogen peroxide (H,02 in a reaction vessel during a certain chemical reaction. Use this graph to answer the questions in the table below. 0.3- 0.25- 0.209 0.170 0.15- 0.0947E 0.05 50 80.0 100 150 1810 250 300 seconds O created Is H,0, being created or destroyed by the chemical O destroyed reaction? O neither created nor destroyed Explanation Check O 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use I Privacy Acce PHED-T280-00T Spring 2021) gevor 20 stv MacBook Air DII DD F10 F9 F8 목 미 ×
Chapter8: Reaction Rates And Equilibrium
Section: Chapter Questions
Problem 8.14E
Related questions
Question
Transcribed Image Text:Calculating average and instantaneous reaction rate from a...
Hann
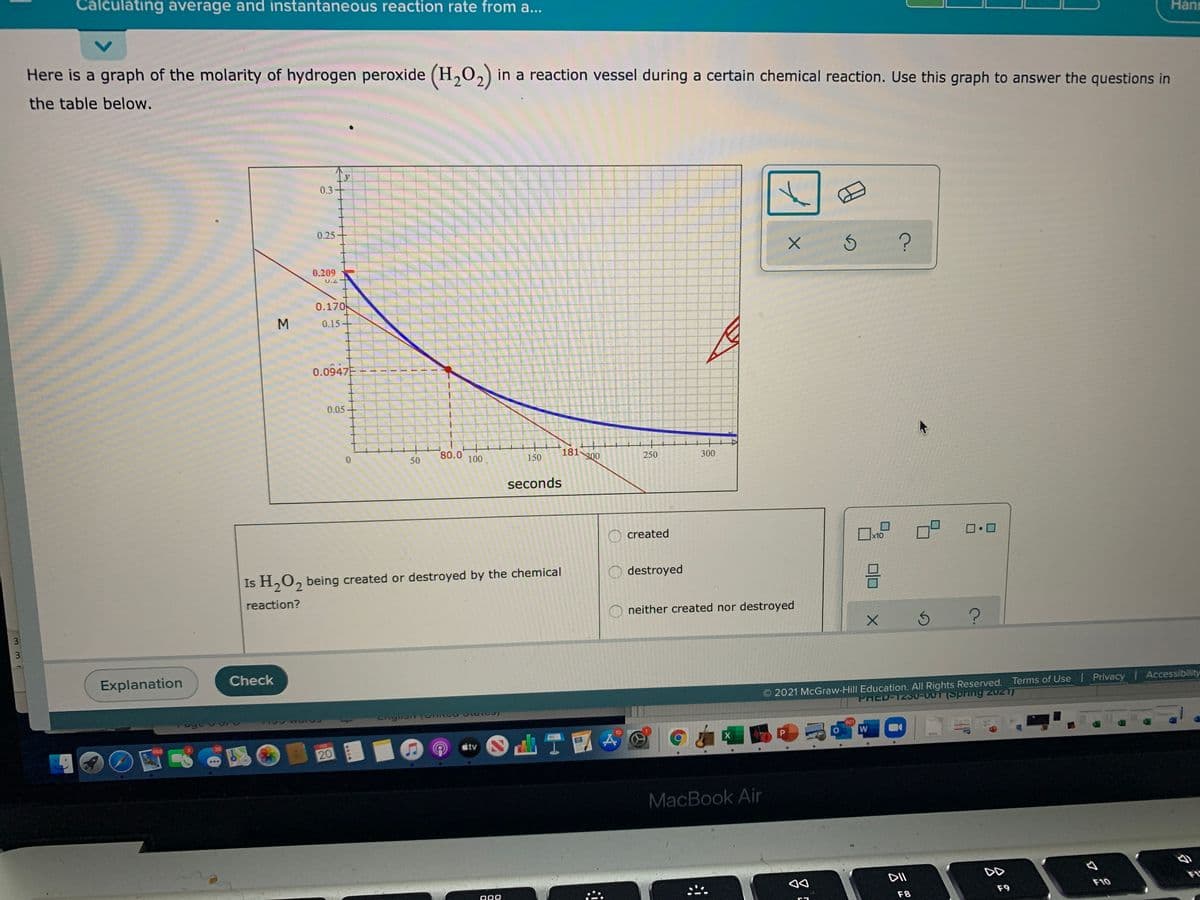
Here is a graph of the molarity of hydrogen peroxide (H,O2) in a reaction vessel during a certain chemical reaction. Use this graph to answer the questions in
the table below.
0.3
0.25
0.209
U.2
0.170
0.15
0.0947E
0.05
80.0
100
181 300
150
250
300
seconds
created
x10
Is H,0, being created or destroyed by the chemical
destroyed
reaction?
neither created nor destroyed
3
3
Explanation
Check
2021 McGraw-Hill Education. All Rights Reserved. Terms of Use I Privacy Accessibility
PRED-1230-001 (Spring 2021)
age
601
668
étv
DOCK
20
MacBook Air
DD
000
F10
F9
F8
50
Σ

Expert Solution
Step 1
As the graph shows that the molarity or concentration of H2O2 goes on decreasing with the time hence H2O2 being Destroyed.
The rest of the answers are shown in below image
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning