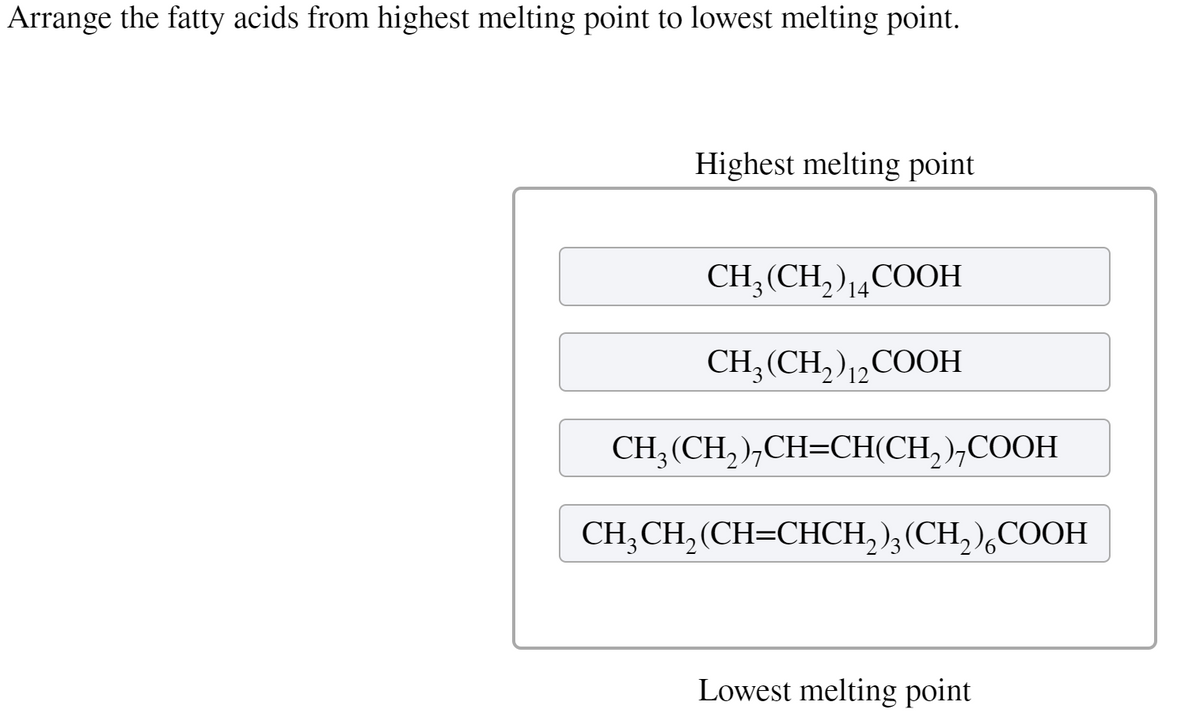
Highest melting point CH; (CH,)COOH CH;(CH, ),,COOH 12 CH; (CH, ),CH=CH(CH, ),COOH CH;CH, (CH=CHCH, );(CH, ),COOH Lowest melting point
Q: Predict which fatty acid is most likely to be a solid at room temperature. CH₂(CH₂),CH=CH(CH₂)₂COOH…
A: Food contains fat, which is necessary for survival. They give energy and basic materials for our…
Q: CH2OH CH2OH CH2OH CH2OH он он OH H OH OH + H2O → OH OH HO Но но OH OH OH OH OH OH The reaction above…
A: The simple sugars called the monosaccharides can be joined together to form the disaccharides and…
Q: Which of the following substances will have the greatest lattice energy? MgS NaF MgO CaO
A: Lattice energy can be defined as the energy required to convert one mole of an ionic solid into…
Q: number of protons to number of neutrons ratio in
A: Every element in the universe has a proton, neutron, and electron. The number of protons is similar…
Q: Which of these atoms has an oxidation number of 2-? O Sulfur O Chlorine O Aluminum O Neon O Silicon…
A: Oxidation State/number And Valency Valency is defined as the number of electrons present in…
Q: What does a higher viscosity result in?
A: Anything that has mass and occupies space is known as matter. There are three main states of matter…
Q: compounds are the products? Carb
A: Carbon dioxide can be defined as a chemical compound that is composed of one carbon and two oxygen…
Q: In hydrolysis of triglycerides what are the reactants and products
A: The question asks to determine the reactants required and the products generated during the…
Q: Water can absorb a large amount of heat without much change in temperature because it has a high…
A: An important property of water is that it is a polar molecule. The hydrogen atoms and the oxygen…
Q: How much solute do you need to make 250 ml of a 3M stock of glucose?
A: The Molarity of a solution is the number of moles of a solute dissolved in a liter of solution. That…
Q: If burning 1 gram of cornflake (in a calorimeter) caused the temperature of 23.0°C water (100 mL) to…
A: Calorie: The calorie is the energy required to raise the temperature of 1 gram f water by 1°C. Its…
Q: Why can water absorb a great amount of heat with little increase in its temperature? What is this…
A: Heat capacity is defined as the amount of heat provided to the material to bring out a unit change…
Q: Name colour Reactions okAmino Acidd t puoteine.
A: The presence of amino acids can be detected using some specific reagent known as colour reactions…
Q: A chemist combined chloroform (CHCI,) and acetone (C, H,O) to create a solution where the mole…
A: Hi, Thanks For Your Question. Ans :
Q: Which of the following is NOT TRUE about water? It has a high specific heat. O Ice floats in liquid…
A: Water is the most abundant compound that is present on Earth’s surface(71%). In nature, it can exist…
Q: Match each fatty acid with its melting point.
A: Lipids are macromolecules that play an important role in the structure and function of the living…
Q: The average fuel value of fats is 9kcal / g . A 1 oz French fries typically contains 4.5 g of fat.…
A: All living organisms require energy to carry out cellular functions for the growth and repair of the…
Q: A pound of body fat stores an amount of chemical energy equivalent to 3500 Cal. When sleeping, the…
A: Calorie It is a measuring unit of energy described as the energy obtained from a specific food. It…
Q: What is the Isoelectric point of Y-R-S (pH 7-11) What is the isoelectric point of N-G-L (pH 1-5)
A: Isoelectric point (PI) is the pH at which the molecule has no net electrical charge. Each amino acid…
Q: Which would have a higher boiling point - a sample of sugar water (sucrose dissolved in water)or a…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is a mineral? Briefly describe the functions of the following minerals: calcium, phosphorus,…
A: In biology , minerals can be defined as any set of inorganic substances that are required for the…
Q: If the cells of your body oxidize 1 mole of glucose, by how much would the temperature of your body…
A: Specific heat capacity is defined as the amount of energy required to raise the temperature of 1 kg…
Q: In general, electrons far from the nucleus have more potential (chemical) energy than electrons…
A: The lowest energy would be in electrons nearest to the nucleus. Higher energy will be provided to…
Q: Which will consume highest amount of 1₂ to become saturated? Which has the highest melting point?…
A: Saturated and Unsaturated fats Saturated fats are those that have single binds between their carbon…
Q: HH нно н но HHO H-N-CC-OH H-N-C C-N-C-C-OH H-NC C-OH CH2 CH2 HC-CH3 CH3 CH2 CH2 HC-CH3 CH3 CH3 ČH3…
A:
Q: what is the basic unit of matter
A: Anything that has mass and is capable of occupying space is considered as matter. Different or…
Q: Which of the following best describes the molecule below? CH OH H. CHOH H H. OH H OH CHOH H OH OH
A: Carbohydrate is one of the biomolecules among protein and lipid and disaccharide belong to…
Q: The production of glycogen within our liver and skeletal muscle cells requires what type of…
A: A molecule synthesized by living organisms or cells is referred to as a "biomolecule."…
Q: Trans-oleic acid (18:1,D9) has a melting temperature of (44.5C) and cis-oleic acid (18:1,D9) has a…
A: Oleic acid is an 18 carbon long mono-unsaturated fatty acid which bears double bond at carbon number…
Q: Which of the following has the highest iodine content? Which of the following has the lowest…
A: Fatty acid: These are the most common lipids. Based on the type of bonds they are classified into…
Q: What is not valid for aerosols?
A: Aerosols are basically a suspension that is composed of fine solid particles or liquid droplets in…
Q: In the iodine clock experiment, if you increase the volume of vitamin C used, would the reaction…
A: The iodine clock experiment or reaction is the very famous experiment for the understanding of…
Q: 1.1 Which of the following statements is true for the chemical depicted below?…
A: A hydrocarbon chain is an organic compound made up solely of carbon and hydrogen atoms linked…
Q: (i) MnO is basic whereas Mn207 is acidic in nature. Why? (ii) Transition metals form alloys. Why?…
A: Chemical compounds are the important basis of life. The compounds that are obtained from living…
Q: If the body can use alcohol as a fuel, why can’t we live on alcohol and a multivitamin/mineral…
A: Nutrients provide energy to the body and maintain the different cellular structures.
Q: In H2O how many hydrogen (h) atoms are present
A: Answer- 2 atoms. In H2O 2 hydrogen atoms are present.
Q: Which of the following is a glycerophospholipid? H,C-O HC-O H,C-O а. b. он HO NH2 C. d.
A: Glycerol is a colorless, odorless, viscous polyol. Glycerol is used in the treatment of wounds and…
Q: Water has unusually high specific heat. What does this mean?
A: The question is saying that "water has unusually high specific heat".We need to determine the…
Q: So you have a solid substance. So you start to add heat to it. What happens as you start to do this?…
A: There are 3 forms of matter Solid, Liquid and Gas. when increasing the temperature or giving heat…
Q: which of the following solvents would NOT dissolve lipids and why? a. Hot ethanol - because this…
A: Biological macromolecules are those important cellular components that are necessary for the…
Q: HHHH HHHHH с-с-с-с-с-с-с-с-с-с-н HHHHH H H Using the graphic above, answer the following question.…
A: Biochemistry is a branch of science that deals with the study of chemical processes related to the…
Q: A catalyst does not alter the difference in the energy contents of the reactants and products. True…
A: Catalysts are substances such as solids, liquids, and gases that speed up chemical processes and are…
Q: Which of the following terms describes when weak forces are broken, drastically changing the shape…
A: Proteins are biomacromolecules which play very important role in maintaining most of the cell…
Q: 11
A: Ans- B
Q: нннн ннннН с-С-с-с-с-С-С-С-С-с-н HHHH HHHH H H. Ising the graphic above, answer the following…
A: Introduction: Life is made up of lifeless chemical molecules. The natural compounds known as the…
Q: Glycerol could react with an excess of which of the following to form a fat or an oil? Choose all…
A: Glycerolipids are one significant type of lipids. Glycerolipids consist of fatty acids and glycerol.…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Rank the fatty acids in each group in order of increasing melting point. A<C<B B<C<A B>C>A A>C>BHow can the fatty acid release energy in the cells?Butter can become rancid as a result of hydrolysis by microorganism. Which of the fatty acids are responsible for the bad odor associated with rancidity?
- How are insulin injection and fatty acid synthesis related? Insulin injection would decrease glucose and increase glycogen synthesis but aren't these carbohydrates compared to lipid storage?Which of these fats, phospholipids, or steroids would you predict would be on the outside of a lipoprotein?Why do unsaturated fatty acids have lower melting points than their saturated counterparts? What is relationship of the degree of unsaturation and melting or boiling point?
- List the properties of fatty acids?Fatty acids have many important functions in the body. What are they?Lipid digestion and absorption:if starting lipid molecule is dietary fat or oil, what will happen to the triacylglycerols of fats and oils once we ingest them? (include location/ site where digestion/ absorption occurs, enzymes, products generated at each location/ site)

