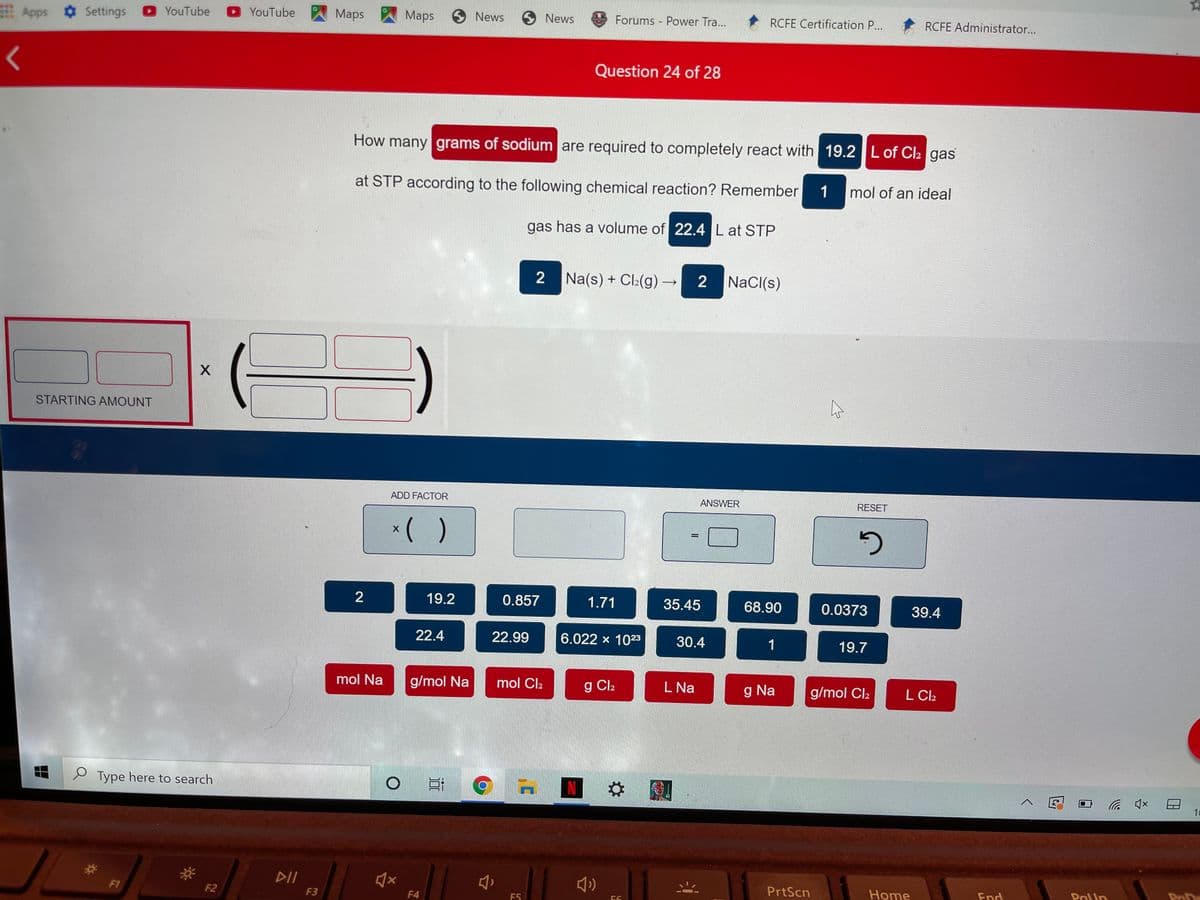
How many grams of sodium are required to completely react with 19.2 L of Cl: gas at STP according to the following chemical reaction? Remember 1 mol of an ideal gas has a volume of 22.4 L at STP 2 Na(s) + Cl:(g) 2 NaCl(s)
How many grams of sodium are required to completely react with 19.2 L of Cl: gas at STP according to the following chemical reaction? Remember 1 mol of an ideal gas has a volume of 22.4 L at STP 2 Na(s) + Cl:(g) 2 NaCl(s)
Chapter5: Gases
Section: Chapter Questions
Problem 74E: Urea (H2NCONH2) is used extensively as a nitrogen source in fertilizers. It is produced commercially...
Related questions
Question
100%
can you answer the questions by filling in the blank with the provided equations and answers
Transcribed Image Text:% Apps Settings YouTube
DYouTube
G.
Maps
Maps
S News
S News
Forums - Power Tra...
RCFE Certification P...
RCFE Administrator...
Question 24 of 28
How many grams of sodium are required to completely react with 19.2 L of Cl2 gas
at STP according to the following chemical reaction? Remember
1
mol of an ideal
gas has a volume of 22.4L at STP
2
Na(s) + Cl:(g) –
2 NaCI(s)
STARTING AMOUNT
ADD FACTOR
ANSWER
RESET
*( )
2
19.2
0.857
1.71
35.45
68.90
0.0373
39.4
22.4
22.99
6.022 x 1023
30.4
1
19.7
mol Na
g/mol Na
mol Cl2
g Cl2
L Na
g Na
g/mol Cl2
L Cl2
P Type here to search
N
xp ツ
10
DII
F1
F2
F3
F4
PrtScn
Home
End
PDD
F5
Palln
道
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning