hydrolysis of sucrose and starch
Q: Consider a mixture of two proteins with molecular weights of 20,000 and 200,000. For simplicity of c...
A: Sedimentation Coefficient is the rate per unit centrifugal field experienced by the particle undergo...
Q: Chlorophyll can only be found in plant cells. True or False
A: Photosynthesis is carried out by the plant, which is a multicellular eukaryotic organism. Photosynth...
Q: Qualitative Analysis of Proteins Tests
A: Proteins are large biological molecule composed of amino acid, amino acids contain both amino (nh2) ...
Q: What are the concepts of specificity, competition, and saturation as they relate to enzymes. Include...
A: Enzymes are biocatalyst that increases the speed of reaction by lowering the activation energy....
Q: 1. In one sentence describe the general structure and functions of the following biomolecules in a l...
A: Biomolecules or Biological molecules are the molecules present in living organisms and are essential...
Q: If a liver extract capable of carrying out normal metabolic reactions (including gluconeogenesis) is...
A: Gluconeogenesis is the metabolic route that leads to glucose production from non-carbohydrate carbon...
Q: What is the basic skeleton structure for flavonoids? What are the different subgroups of flavonoids?...
A: The food that we get from plants contains a variety of natural nutrients. These are called phytonutr...
Q: People suffering with sickle cell anemia have a structural defect in hemoglobin (HB). The major reas...
A: Hemoglobin is the iron-containing pigment in the red blood cells which helps the blood to carry or t...
Q: Read through each scenario. Under the scenario, write which lab safety rule is being broken and indi...
A: Hi! Thank you for the question, as per the honor code, we are allowed to answer the first 3...
Q: Why did Okazaki propose that lagging strand synthesis proceeds through the synthesis of short fragme...
A: The effective copying of double-stranded chromosomal DNA is required for cellular DNA replication. T...
Q: For the following, calculate pka, and pka, of 1 Glycine, determine the 2 buffering regions, and calc...
A: Glycine is an amino acid with H-atom as the side chain. The isoelectric point of a amino acid (pI) i...
Q: Describe the mechanism of action of salivary amylase. Explain how it works and on what type of molec...
A: Carbohydrate along with protein, fat and water comprise macro-molecules which are required in larger...
Q: Discuss the different structures (primary, secondary, tertiary, and Quaternary structures) of protei...
A: Proteins are polymers of amino acids linked by peptide bonds (carboxyl group of one amino acid with ...
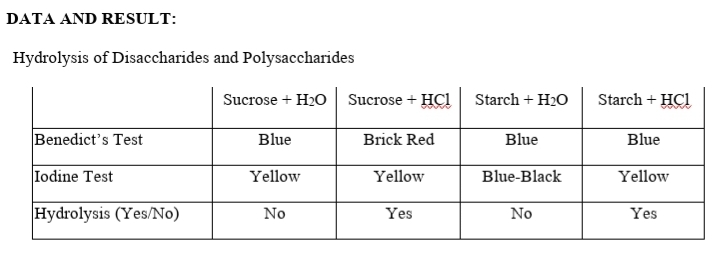
Q: Hydrolysis of Disaccharides and Polysaccharides Sucrose + H,O Sucrose + HCI Starch + H,O Starch + HC...
A: The disaccharides and polysaccharides are broken into their component sugar molecules by the process...
Q: Why do you think DNA is the genetic material used by eukaryotes instead of RNA?
A: Deoxyribonucleic acid (DNA): Nucleic acids are molecules that store hereditary information for perf...
Q: At a pH of 7.40, the carbonic acid ratio is, a. 35:1 b. 4:1 C. 20:1 d. 3:1
A: Given Values: pH = 7.4 pKa = 6.1
Q: aw the processes that happen in Glycoly
A: GLYCOLYSIS- It is a metabolic pathway that converts glucose into pyruvic acid. in this process, glu...
Q: Enumerate and describe the classes of enantiomeric drug Discuss briefly the different methods of ena...
A: All the objects & compounds around us are made up of the basic unit called atoms. Atoms combine ...
Q: CH3CH2OH +HCOOH
A: Alcohol reacts with many kinds of acid to form esters. When alcohol and carboxylic acid reacts, the ...
Q: Ceramide is synthesized biologically from sphingosine. 1. What type of reaction in this synthesis re...
A: Ceramide is a type of lipid, which is composed of sphingosine and fatty acids. Ceramide forms glycol...
Q: Explain why saturated (flat) fatty acids cause more plaque in blood vessels than unsaturated (gnarle...
A: In an aqueous environment the polar heads of the fatty acids will engage in favorable interaction be...
Q: You discover that one of your novel class IV transcription factors contains TBP. Describe an experim...
A: The TAFs are transcription factors associated with the TATA-binding protein, and until recently they...
Q: The proteins and other substances that bind to the DNA rely mostly on non-covalent interaction to de...
A: Proteins are an important class of biomolecules that are found in all living organisms and are compo...
Q: 10. The patients with a genetically determined medium-chain acyl-CoA dehydrogenase (MCAD) deficiency...
A: MCAD (medium- chain acyl- CoA dehydrogenase) deficiency is inherited disorder which prevents our bod...
Q: 3. A patient has got excess carbohydrate meal for the years and gain the weight. To explain this: a)...
A: A.) SCHEME OF TAG SYNTHESIS IN LIVER
Q: Complete the table below using the textbook or other information sources, if necessary. How will the...
A: A functional group is a group of atoms in a molecule that gives particular characteristics and chemi...
Q: Translate the following mRNA into protein, starting from the first initiation codon: 5'-CCGAUGCCAU...
A: Translation is the process of synthesis of protein from an mRNA. mRNA synthesized through transcript...
Q: 4. Draw the -forms of the following sugars. CHO CHO H- OH но H- HO H- HO. OH OH CH,OH CH;OH 5. Draw ...
A: A biomolecule is a molecule produced by living organisms or cells. Carbohydrates, proteins, lipids, ...
Q: You are working in the lab with a polypeptide that is present in solution at 1 mg/mL. The polypeptid...
A: Molarity (M) is the amount of a substance in a certain volume of solution. Molarity is defined as th...
Q: 21) Compute the moles of FADH2, NADH, GTP generated and NADPH consumed in the dissimilation of one m...
A: Lipids are easily soluble in nonpolar solvents such as benzene, ether, ...
Q: what is a hydration number
A: Hydration refers to the interaction of a molecule or substance with water. Water interacts with seve...
Q: Create a concept map for Glycosides.
A: Glycosides are molecules in which a sugar is attached to another functional group present in another...
Q: How can you show that σ (sigma factor) does not really accelerate the rate of transcription elongati...
A: Transcription: It is the first step of gene expression where a particular segment of DNA is copied t...
Q: Which one of the following correctly illustrates peptide bond formation in the peptidyltransferase c...
A: The ribosomal peptidyl transferase center residing in the domain V of 23S rRNA (of the 50S subunit o...
Q: What is the monomoric form of actin referred to as? a. F-actin b. G-actin c. M-actin ...
A: Actin exists in two forms. They are G-actin (or globular actin) and F-actin (or fibrous actin). Acti...
Q: Make a comparison between a prokaryotic cell and a eukaryotic cell as to: 1. genetic material 2. siz...
A: The three domains of life includes are categorized into Archaea, Bacteria, and eukarya. There are tw...
Q: Draw the biosynthetic pathway with explanations of each step for the following secondary metabolite ...
A: Polyketides are naturally found molecules of a large and diverse group of secondary metabolites whic...
Q: LUT3 iš homologous to human GLU 25. Rabbi threonine, two (2) serine and one (1) asparagine. Based on...
A: Since the environment within the lipid bilayer is nonpolar, the transmembrane regions of the the mem...
Q: new protein of unknown structure has been purified. Gel filtration chromatography reveals that the n...
A: native purification: in this type of purification, the protein structure is not disturbed. denaturan...
Q: Topoisomerases can cut phosphodiester bonds and the DNA ligases will have to seal the nicks whenever...
A: DNA ligases are enzymes that catalyze formation of a phosphodiester bond at a single-strand break in...
Q: Connection between Electron Transport & Phosphorylation Task: 1. Define P/O ratio 2. Explain its...
A: Our body is always working, continuously doing various metabolic activities even when we are sleepin...
Q: What is the differences between competitive, noncompetitive, mixed noncompetitive and uncompetitive ...
A: Enzyme inhibition is of two types reversible and irreversible. During irreversible inhibition the in...
Q: It is estimated that the most common amino acid in a protein is leucine. What is the relative leucin...
A: Leucine is an aliphatic hydrophobic amino acid residue. These residues populate the interior hydroph...
Q: lease describe a strategy to correctly balance a centrifure rotor if you have 4 samples. 2 samples w...
A: Centrifuge: It is an important tool in the biochemistry lab that through a rapid spinning process u...
Q: ive the functions of the following ingredients, then name a branded/commercial skin or hair care pro...
A: 1. BETAINE: It is also called as betaine anhydrous, or trimethylglycine (TMG), which is synthesized...
Q: Explain how PEMBA is used to isolate, differentiate and enumerate Bacillus cereus from food sample.
A: Microscopic organisms such as bacteria, fungi (mold and yeast), protists, archaea, alga...
Q: [N] NO MATCH 1. yields 2 moles of fatty acid, phosphoric acid, glycerol and choline upon hydrolysis ...
A: Since we only answer up to 3 sub-parts, we'll answer the first 3. Please resubmit the question and s...
Q: What are the advantages and disadvantages of viable cell count and the turbidimetric methods?
A: In turbidimetric determination of cell growth ,after inoculation of the bacterial culture the optica...
Q: Q21. What regulators of gene expression bind the lac promoter region if E. coli is grown in media co...
A: In the absence of lactose the repressor binds to the operator sequence adjacent to the promoter and ...
Q: The conversion of succinate to fumarate in the TCA cycle is shown below. Need carbon a and b ans...
A: The series of chemical reactions that occur inside the living body for the production of energy are ...
Step by step
Solved in 4 steps with 2 images

- Explain these results. Why did glucose result in the highest rate? Why did sucrose result in a lower rate than fructose, and starch even lower? Recall that sucrose is a disaccharide composed of glucose and fructose, and that starch is a polysaccharide composed of many glucose molecules.Circle and label the hemiacetal functional group and the acetal functional group in each of thefollowing carbohydrates:Carbohydrates: More than Just Energy: Concept Card Mapping Carbohydrates Create a mind map for carbohydrates conecept map
- Humans can digest starch, maltose, sucrose and lactose but not cellulose. What structural feature do the digestible carbohydrates have in common? (Biochemical structure related)Long explanations are not needed. Direct answers would suffice. a. Which of the following is NOT an example of structural polysaccharides? I. amylose II. cellulose III. chitin b. Which of the following is the epimer of glucose at C-2? I. gulose II. galactose III. mannose IV. fructose V. taloseMoore's Test We did an experiment about carbohydrate chemistry and the professor did not elaborate on the details. She just gave the positive result and reagent. I would like to ask the following: What type of reaction occurred when the samples (enumerated) reacted with the MOORE'S reagent? Give the chemical equation for each and the principle/mechanism of the reaction. Glucose Sucrose Fructose
- Create an illustrated step-by-step schematic diagram of the following Analysis of Lipids: Huble's testBoth reducing monosaccharide and disaccharide give positive results in Barfoed's test. Which of the following indicators would you be able to distinguish the two?* A. Monosaccharides will react more slowly than disaccharide. B. Disaccharides are easily oxidized than monosaccharide, thus gives red precipitate more quickly than monosaccharide. C. Monosaccharide is more reactive thus gives red precipitate later than disaccharide D. Disaccharide undergoes hydrolysis first, thus takes more time to form red precipitate than monosaccharide.Answer questions 4, 5, and 74. The only sugar structure that does NOT contain chiral carbon atoma. Erythroseb. Erythrolusec. Glyceraldehyded. Dihydroxyacetone5. The simplest class (with an example) of carbohydratesa. Oligosaccharides :Trehalose c. Disaccharides : Lactoseb. Monosaccharides : Sorbose d. Polysaccharides : Starch6. What form must all carbohydrates be in for cells to use them as an energy sourcemaking it the most abundant inside the body?a. Glycogen c. Glucoseb. Fructose b. Ribose7. Polysaccharides area. Sachharide units that contain 3 to 10 unitsb. the simplest sugar unitsc. Saccharides with 2 sugar units joined through glycosidic linkaged. class of carbohydrates is considered as non-sugar8. Which of the following glycosidic linkage is found in maltose?a. Glucose (α-1 – 2β) Fructoseb. Glucose (α1 – 4) Glucosec. Galactose (β1 – 4) Glucosed. Glucose (β1 – 4) Glucose9. Choose the statement/s that is/are correcta. cellulose are…
- Give 5 carbohydrate-containing samples and identify the specific name of carbohydrate present in each of them.Match the following compounds with choices (a) monosaccharide, (b) disaccharide, or (c) polysaccharide:SucroseGlucoseGlycogenFructoseStarchIn detail, describe the function of storage polysaccharides vs. structural polysaccharides.

