Q: Suggest two possible experimental errors that may result in affecting the precise determination of…
A: Two possible experimental errors that may affect the precise determination of boiling point are:
Q: To convert an alcohol to an alkene, one would use a hydrolysis reaction. Answer: FALSE, can you…
A: The given statement: Alcohols are converted to alkenes by hydrolysis reaction. The given statement…
Q: If you find that a 69.8 mL sample of vinegar requires 120.31 mL of a 0.504 M solution of NaOH for…
A: To calculate the amount of acetic acid in the vinegar sample, we can use the following balanced…
Q: 3) Given the reactions: CO2(g) → C + O2(g) AH =393 KJ S(s) + O2(g) → SO2(g) AH =-296.4 KJ CS2 (1) +…
A:
Q: Nitric acid is often manufactured from the atmospheric gases nitrogen and oxygen, and hydrogen…
A:
Q: Suppose 0.753 g of sodium chloride is dissolved in 100. mL of a 57.0 m M aqueous solution of…
A:
Q: 1a) Assuming standard conditions, answer the following questions. (Use the table of Standard…
A: The species with the standard reduction potential greater than H+ would oxidize the H2.
Q: A 1.00-L solution contains 3.75×10-4 M Cu(NO3)2 and 2.00×10-3 M ethylenediamine (en). The Kf for…
A:
Q: Identify the isomeric relationship between each pair of molecules below. The options are:…
A:
Q: Why does H not have a physical state
A: Hydrogen always exist in the form of molecular H2. Not as H in the atomic state. Hydrogen is known…
Q: Light fuel oil with an average chemical composition of C10H18 is burned with oxygen in a bomb…
A: This problem is based on Thermodynamics. The solution is given below.
Q: draw the structures of the geometric and optical isomers of the compound [Co(gly)3
A: We have been given an inorganic compound [Co(gly)3].We have been asked to draw the structures of…
Q: An unknown compound has the following chemical formula: PCI where x stands for a whole number.…
A:
Q: Over coffee and croissants at breakfast one day, your friend Jane (an expert chemist) says this:…
A: Answer:- This question is answered by using the simple concept of reaction of metal sulfide with…
Q: Draw the products of each acid-base reaction. EXAMPLE: a. b. CH3CH₂ propanoic acid OH + CH3CH₂CH₂ OH…
A:
Q: What is the pH of the solution with the following analytical concentrations: a) 0.0650M in H3PO4 and…
A: Answer: Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: Show which peaks associate with which carbons and hydrogens in the benzaldehyde
A: The question is based on organic spectroscopy. we need to analyse the spectra and identify the…
Q: The standard free energy of the hydrolysis of ATP to ADP: ATP (aq) → ADP (aq) + (aq) phosphate in…
A:
Q: Provide IUPAC name for the structure shown:
A:
Q: Consider the following data on some weak acids and weak bases: name hydrocyanic acid hydrofluoric…
A: Answer: This question is based on hydrolysis of salt, salt of strong acid and strong base doesn't…
Q: Draw electron-dot structures for the product. 4NH,+Zn® _ Zn(NH3)2 Draw the molecule by placing atoms…
A: While drawing electron-dot structure we should aware of the lone pair of electrons on the species.
Q: If 364 mL of sulfuric acid is diluted to 1.00 L to give a solution that is 1.94 M, what was the…
A:
Q: 10. If 223.0 mg of AgBr is completely dissolved in 184.0 mL of 0.14 M NH3, what is the equilibrium…
A: Given, The mass of the AgBr = 223.0 mg The concentration of ammonia (NH3) = 0.14 M The volume of the…
Q: C3H60(1)+402(g)→3CO2(g)+3H2O(g) AH.rxn=-1790kJ Part A If a bottle of nail polish remover contains…
A: The given reaction, C3H6O(l) + 4O2(g)→3CO2(g) + 3H2O(g) Volume of acetone = 265 mL Density of…
Q: Based upon the limiting reagent, calculate the theoretical yield of magnesium oxide, MgO, for the…
A: Given: Ignition of magnesium metal in air for this experiment To determine: Based upon the limiting…
Q: Consider the reaction below. Rank the following species in order of appearance in the reaction…
A:
Q: groups in aspirin absorbing at different wavenumbers? 3. Below is the NMR spectrum of aspirin with…
A: The question is based on the concept of organic spectroscopy. we need to analyse and interpret the…
Q: 2. Calculate the mass of 2-methylcyclohexanol that represents 0.033 mole
A:
Q: Draw the graphic from Figure 15.12. Explain how it helps you calculate any of the values of [H3 O+…
A: pH is the negative logarithm of the H+ ions. From the pH we can predict the concentration of H+ ions…
Q: MISSED THIS? Read Section 18.2 (Pages 788 - 799); Watch KCV 18.2B, IWE 18.2. Use the…
A:
Q: Answer the questions in the table below about the shape of the tetrachloroiodide (IC₁4) a anion.…
A: The question is based on VSEPR theory we have to determine the shape and bond angle of the given…
Q: In a reaction where a carboxylic ester and water react to produce an alcohol and carboxylate (or…
A: In a reaction where a carboxylic ester and water react to produce an alcohol and carboxylate (or…
Q: hello i am having difficulty with part a, b, and c A galvanic cell based on the above reaction is…
A: Galvanic cell
Q: 2. Briefly explain why it is essential that none of the yeast comes into contact with the H₂O2…
A: H2O2 should come into contact with yeast before the stoppers are firmly placed as the oxygen…
Q: Снзена сна-лідвь П + енз сна сня сна он ко усна сна-сна сна снагомдер H₂₂0 сна-сна снасна он + му…
A:
Q: Part F 4- V₂07¹ Express your answer as signed integers separated by a comma. View Available Hint(s)…
A: In the given compound, oxygen is in -2 oxidation state and vanadium is in its highest oxidation…
Q: B Current Attempt in Progress X Incorrect Draw the major product(s) for the following reaction. If a…
A: We have been given an organic reaction and we have been asked to draw the major product of given…
Q: D. E. F. _OH НО. нази зи OCH₂CH₂CH3 НО.
A: The question is based on organic synthesis. we need to synthesize the product from the reactants…
Q: QUESTION 13 Use the AH°f and AHxn information provided to calculate AH°f for IF: IF7(9) + 12(g) → IF…
A: Note: As per the guidelines solution of question 13 has been made. For the expert solution of other…
Q: Calculate the equilibrium constant of the reaction N2(g) + 3 H₂(g) = 2 NH3(g) at 25°C, given that AG…
A:
Q: QUESTION 7 Determine the final temperature of a gold nugget (mass = 376 g) that starts at 288 K and…
A:
Q: [References The mass spectrum of compound A shows the molecular lon at m/z 85, an M + 1 peak at m/z…
A: The question is based on the concept of organic spectroscopy. we need to analyse the spectral data…
Q: Draw a structural formula for the major organic product of the following SN2 reaction. -CH₂Br +…
A: Answer:- This question is answered by using the simple concept of chemical reactions of organic…
Q: Which of the following transport systems allows small molecules such as urea and O₂ to cross the…
A: By the passive diffusion process small molecule cross the cell membrane without expanding energy .…
Q: Consider the titration of 100.0 mL of 0.100 M H₂NNH₂ (Kb = 3.0 x 10-6) by 0.200 M HNO3. Calculate…
A:
Q: Which of the following two molecules is more basic? Justify your answer A B
A: basicity is directly proportional to electron donation, the compound which can donate electron…
Q: Give IUPAC names for the following compounds. a) b) CH₂ CH3CCO2H CH3 0 ОН
A: -CO2H represent carboxylic acid (-COOH) functional group. Numbering starts from functional group…
Q: INSTRUCTION: MAKE A CHEMICAL REACTION AND DRAW THE PRODUCT OF THE GIVEN COMPOUND. What product is…
A: K2Cr2O7 and Tollens reagent both acts as oxidising agents . But their reactivity is completely…
Q: . A buffer is prepared by mixing 48.2 mL of 0.196 M NaOH with 141.2 mL of 0.231 M acetic acid. What…
A:
Q: Draw the structure of the compound that is consistent with the ¹H NMR. (Assume that long-range…
A: Question is Based on the concept of organic spectroscopy. We need to analyse the spectre and…
ochem help
Rank the following radicals in the order of increasing stability (see attached image)
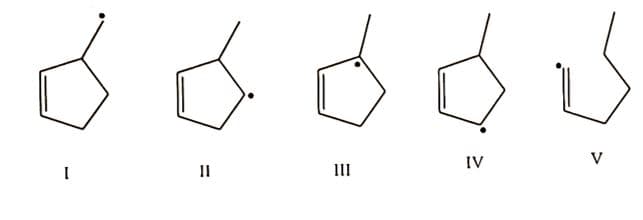
Step by step
Solved in 2 steps with 1 images

- HiiiiiiA concentration of carbon monoxide of 4000 ppm (0.400 % by mass) in air is lethal in about half an hour. What mass in grams of carbon monoxide in a car with an internal volume of 2.9 m3 is sufficient to provide that concentration? Assume the density of air is 1.29 g/L ; 1m3=1000L.A sample of ferrous oxide has actual formula Fe93O1.00. In this sample what fraction of metal ions are Fe2+ions? What type of nonstoichiometric defect is present in this sample?

