i Melting point of Nacl is lower than Mgo why? ii. Br is less reactive than F, why? I iil. Complete and balance the following two reactions and derive your conclusion, what do they indicate? a. Al(OH)3s) + H b. Al(OH)as) *+ HO (aq) (ag)
i Melting point of Nacl is lower than Mgo why? ii. Br is less reactive than F, why? I iil. Complete and balance the following two reactions and derive your conclusion, what do they indicate? a. Al(OH)3s) + H b. Al(OH)as) *+ HO (aq) (ag)
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter20: Transition Metals And Coordination Chemistry
Section: Chapter Questions
Problem 98IP
Related questions
Question
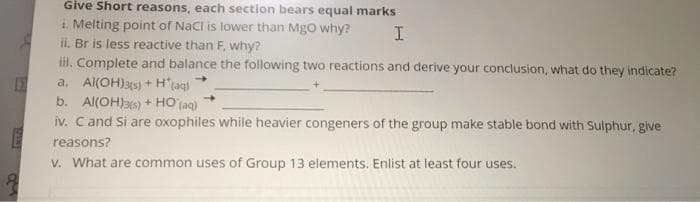
Transcribed Image Text:Give Short reasons, each section bears equal marks
i Melting point of Nacl is lower than MgO why?
ii. Br is less reactive than F, why?
il. Complete and balance the following two reactions and derive your conclusion, what do they indicate?
a. Al(OH)3(s) + H'(aq)
b. Al(OH)3s) + HO (aq)
iv. Cand Si are oxophiles while heavier congeners of the group make stable bond with Sulphur, give
reasons?
V. What are common uses of Group 13 elements. Enlist at least four uses.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning