I. Qualitative Analyses of Proteins Complete the table below. Chemical/reagent used (be specific) Final expected Test Sample Interpretation visible result after the experiment (color change, precipitation, etc.) Egg albumin Gelatin dispersion Biuret test 0.5% urea 0.5% glycine Egg albumin Xanthoproteic test Gelatin dispersion 0.5% lysine 0.5% tryptophan Egg albumin Gelatin dispersion 0.5% glycine 0.5% asparagine Egg albumin Ninhydrin test Millon's test Gelatin dispersion 0.5% tyrosine 0.5% glycine
I. Qualitative Analyses of Proteins Complete the table below. Chemical/reagent used (be specific) Final expected Test Sample Interpretation visible result after the experiment (color change, precipitation, etc.) Egg albumin Gelatin dispersion Biuret test 0.5% urea 0.5% glycine Egg albumin Xanthoproteic test Gelatin dispersion 0.5% lysine 0.5% tryptophan Egg albumin Gelatin dispersion 0.5% glycine 0.5% asparagine Egg albumin Ninhydrin test Millon's test Gelatin dispersion 0.5% tyrosine 0.5% glycine
Chapter10: Reconstitution Of Powdered Drugs
Section: Chapter Questions
Problem 7.2P
Related questions
Question
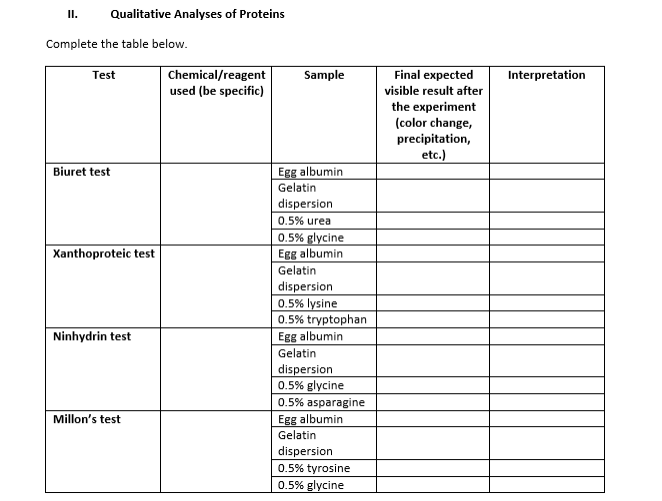
Transcribed Image Text:I.
Qualitative Analyses of Proteins
Complete the table below.
Chemical/reagent
used (be specific)
Final expected
Test
Sample
Interpretation
visible result after
the experiment
(color change,
precipitation,
etc.)
Egg albumin
Gelatin
dispersion
Biuret test
0.5% urea
0.5% glycine
Egg albumin
Xanthoproteic test
Gelatin
dispersion
0.5% lysine
0.5% tryptophan
Egg albumin
Gelatin
dispersion
0.5% glycine
0.5% asparagine
Egg albumin
Ninhydrin test
Millon's test
Gelatin
dispersion
0.5% tyrosine
0.5% glycine
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you

