Question 7 The uncoupling agent 2,4-dinitrophenol (DNP) has a pk, of about 7.2. Which of the following describes how this molecule works to uncouple the proton gradient? NO2 O2N- 2,4-Dinitrophenol (DNP)
Question 7 The uncoupling agent 2,4-dinitrophenol (DNP) has a pk, of about 7.2. Which of the following describes how this molecule works to uncouple the proton gradient? NO2 O2N- 2,4-Dinitrophenol (DNP)
Biochemistry
9th Edition
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Chapter1: Biochemistry: An Evolving Science
Section: Chapter Questions
Problem 1P
Related questions
Question
100%
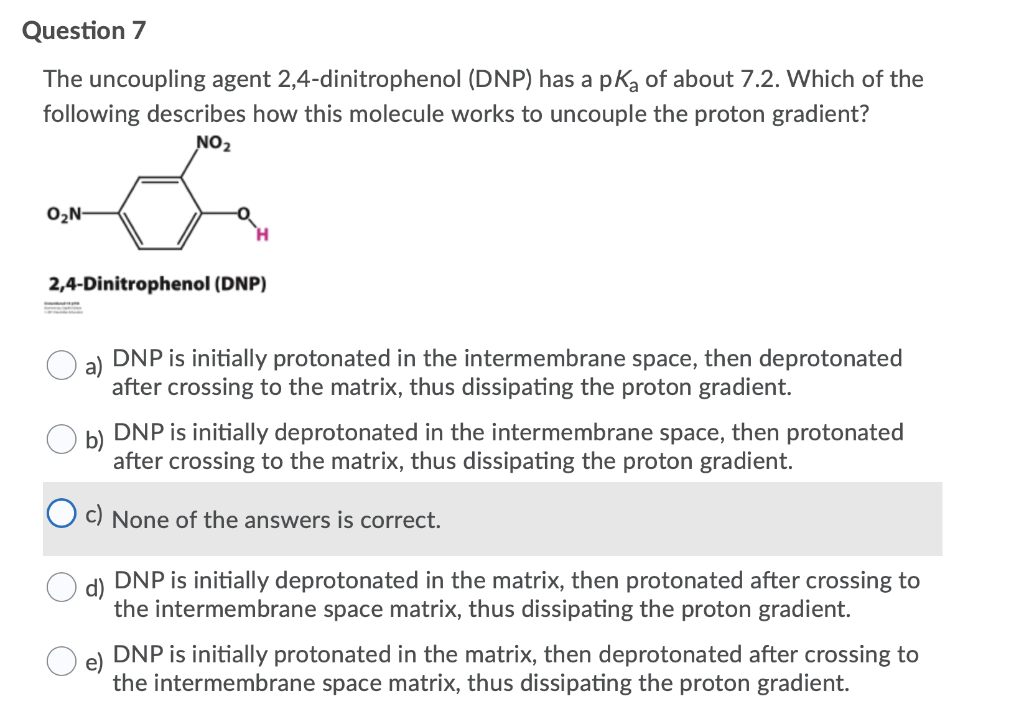
Transcribed Image Text:Question 7
The uncoupling agent 2,4-dinitrophenol (DNP) has a pką of about 7.2. Which of the
following describes how this molecule works to uncouple the proton gradient?
NO2
O2N-
2,4-Dinitrophenol (DNP)
DNP is initially protonated in the intermembrane space, then deprotonated
after crossing to the matrix, thus dissipating the proton gradient.
DNP is initially deprotonated in the intermembrane space, then protonated
after crossing to the matrix, thus dissipating the proton gradient.
O c) None of the answers is correct.
DNP is initially deprotonated in the matrix, then protonated after crossing to
the intermembrane space matrix, thus dissipating the proton gradient.
DNP is initially protonated in the matrix, then deprotonated after crossing to
the intermembrane space matrix, thus dissipating the proton gradient.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781319114671
Author:
Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:
W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:
9781464126116
Author:
David L. Nelson, Michael M. Cox
Publisher:
W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul…
Biochemistry
ISBN:
9781118918401
Author:
Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:
WILEY

Biochemistry
Biochemistry
ISBN:
9781319114671
Author:
Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:
W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:
9781464126116
Author:
David L. Nelson, Michael M. Cox
Publisher:
W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul…
Biochemistry
ISBN:
9781118918401
Author:
Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:
WILEY

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Fundamentals of General, Organic, and Biological …
Biochemistry
ISBN:
9780134015187
Author:
John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:
PEARSON