Identify each of the labeled points (indicated with letters) or changes (indicated with two letters separated by an arrow) shown on the phase diagram. 1.00- Pressure (atm) 0.50 F 0.10- -150 -75 75 150 Temperature ("C) Match the words in the left column to the appropriate blanks in the sentences on the right. Make certain each sentence is complete before submitting your answer. • View Available Hint(s) Reset Help sublimation 1. The transition shown by the line from F to E is freezing 2. The transition shown by the line from E to F is melting 3. The transition shown by the line from C to D is triple point critical point 4. The transition shown by the line from E to C is deposition 5. The point indicated by B is the boiling 6. The point indicated by A is the condensation 7. The transition shown by the line from D to C is 8. The transition shown by the line from C to E is
Identify each of the labeled points (indicated with letters) or changes (indicated with two letters separated by an arrow) shown on the phase diagram. 1.00- Pressure (atm) 0.50 F 0.10- -150 -75 75 150 Temperature ("C) Match the words in the left column to the appropriate blanks in the sentences on the right. Make certain each sentence is complete before submitting your answer. • View Available Hint(s) Reset Help sublimation 1. The transition shown by the line from F to E is freezing 2. The transition shown by the line from E to F is melting 3. The transition shown by the line from C to D is triple point critical point 4. The transition shown by the line from E to C is deposition 5. The point indicated by B is the boiling 6. The point indicated by A is the condensation 7. The transition shown by the line from D to C is 8. The transition shown by the line from C to E is
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter6: Equilibria In Single-component Systems
Section: Chapter Questions
Problem 6.47E
Related questions
Question
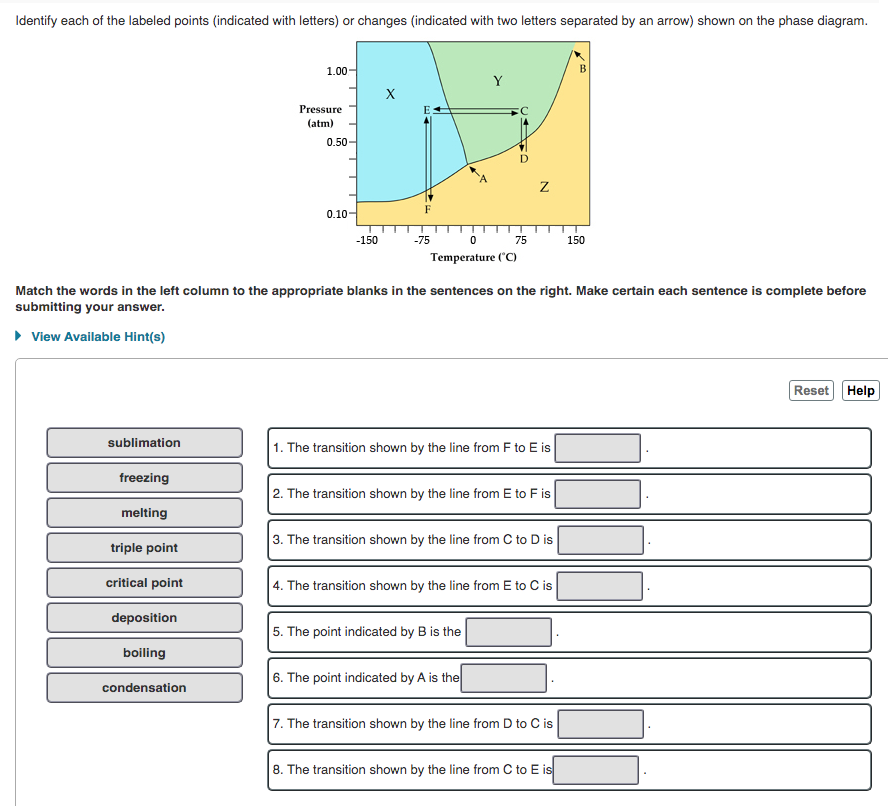
Transcribed Image Text:Identify each of the labeled points (indicated with letters) or changes (indicated with two letters separated by an arrow) shown on the phase diagram.
1.00
Y
Pressure
(atm)
0.50
F
0.10-
-150
-75
0 75 150
Temperature ("C)
Match the words in the left column to the appropriate blanks in the sentences on the right. Make certain each sentence is complete before
submitting your answer.
• View Available Hint(s)
Reset Help
sublimation
1. The transition shown by the line from F to E is
freezing
2. The transition shown by the line from E to F is
melting
3. The transition shown by the line from C to D is
triple point
critical point
4. The transition shown by the line from E to C is
deposition
5. The point indicated by B is the
boiling
6. The point indicated by A is the
condensation
7. The transition shown by the line from D to C is
8. The transition shown by the line from C to E is
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning