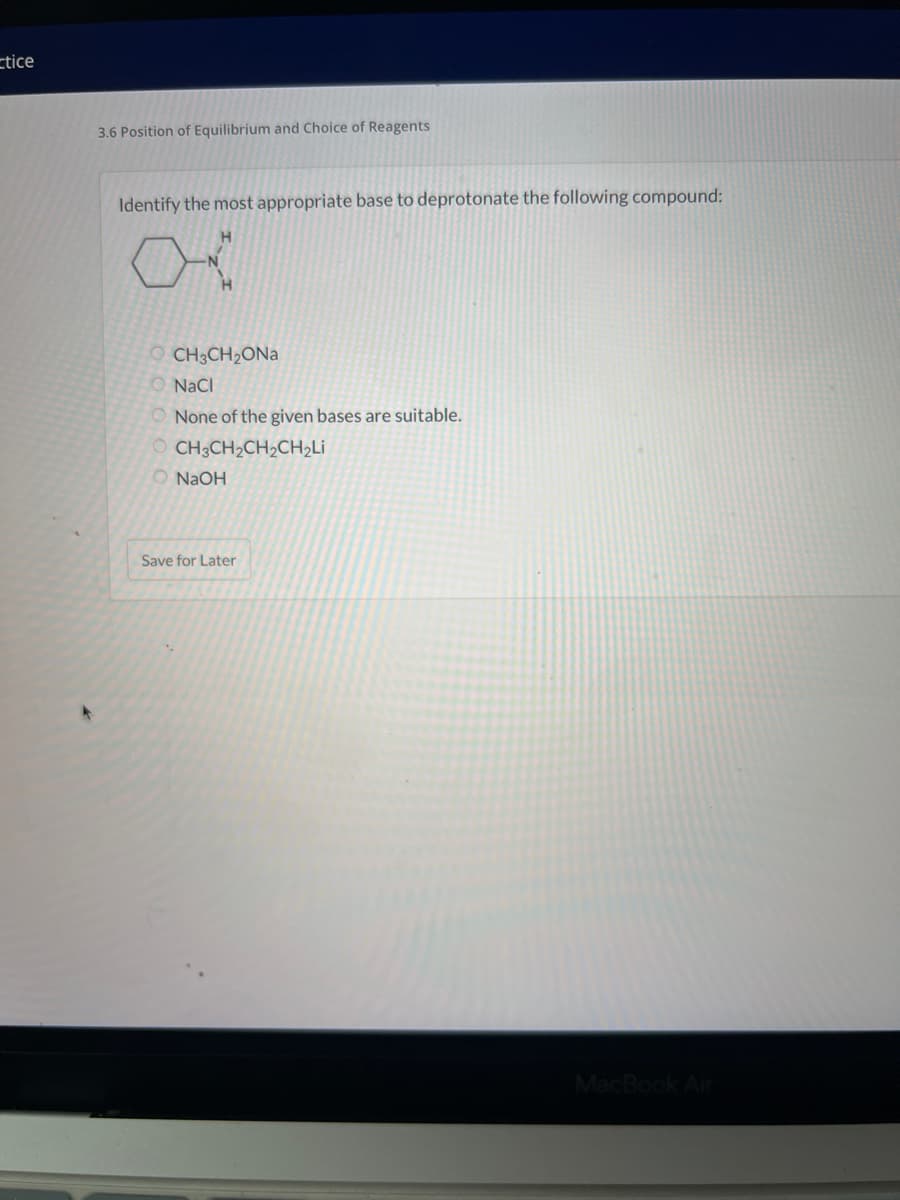
Identify the most appropriate base to deprotonate the following compound: H CH3CH₂ONa O NaCl None of the given bases are suitable. OCH3CH₂CH₂CH₂Li O NaOH Save for Later
Q: For each chemical reaction listed in the table below, decide whether the highlighted atom is being…
A:
Q: Ask me Delta H for the following reaction indicate exothermic endothermic.
A:
Q: Determine the solubility product of CaF2 if its solubility in water is 25.9 mg/L.
A: Given soluublity of CaF2 in water = 25.9 mg/L
Q: Suppose 720. mmol of electrons must be transported from one side of an electrochemical cell to…
A:
Q: Re-order each list in the table below, if necessary, so that the atoms or ions in it are listed in…
A: Atomic radius: The distance between the nucleus and the outermost electron is known as the atomic…
Q: Propose a synthesis and give detailed comments on each reagent you used and why you wouldn't use…
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts. H3O+, heat Drawing CN
A: In the above given reaction the starting material contains -CN (cyanide) functional group and it is…
Q: What compound will have the most conductivity in solution? A. Potassium bromide b. Glucose c.…
A: Conductivity is directly proportional to ions present in the solution. Ionic compounds formed by…
Q: This energy diagram shows the allowed energy levels of an electron in a certain atom or molecule:…
A: The energy diagram shown below is for any atom or molecule, the transition from ground state to…
Q: Calculate the mass of glucose (C6H₁2O6) that contains a trillion (1.00 × 10¹2) oxygen atoms. Be sure…
A:
Q: What quantity in moles of nitrogen gas at 130.0 °C and 13.3 atm would occupy a vessel of 23.4 L?
A: we have to calculate moles of nitrogen gas
Q: A and C are correct but B was incorrect
A: b. Given that, the Ka of HIO is 2.3×10-11. The number of moles of HIO and NaIO in a 3.4 L buffer…
Q: How many dissociated H+ ions are there in (3.1x10^-1) L of an aqueous solution whose pH is 9.68?
A:
Q: What is the common name for this compound? OH
A: Since, Common name are those name which is generally used to identify the chemical in the…
Q: The pressure in Denver, Colorado averages about 632 mm Hg. How many atmospheres is this?
A:
Q: than S. __H2S is a stronger acid than H2O because S atoms are larger than O atoms. __H2O is a…
A:
Q: What is the solubility of Cr(OH)₃ at a pH of 10.30? (Ksp Cr(OH)₃ is 6.70 × 10⁻³¹)
A: The dissociation of Cr(OH)3 is shown as- Cr(OH)3→Cr3+(aq) + 3OH-(aq) Ksp of Cr(OH)3 = 6.70×10-31
Q: A 2.53 L container at 74.1 °C contains 6.31 g of N2O3(g). The N2O3 gas decomposes completely,…
A:
Q: Dwi the complete electron Configuration for ion formed by the the common monatomic Clement…
A: ♤ Atomic number of potassium, K : 19Group : 1Period : 4It is a monoatomic element since it is made…
Q: Calculate the volume in liters of a 9.5 x 10 mm mercury (1) chloride solution that contains 225.…
A: Molarity is defined as the number of moles present in one liter of the solution. The unit of…
Q: What is the pH of a 3.65 L solution of 4.55 M NH3? How many grams of LiOH would it take in the same…
A: Given data: Volume of NH3 solution = 3.65 L Concentration of ammonia solution = 4.55 M Volume of…
Q: Draw the skeletal line bond structure of (r)-3-fluorobutanoyl chloride
A: Given -> R-3-fluorobutanoyl chloride
Q: ess Consider the insoluble compound silver hydroxide, AgOH. The silver ion also forms a complex with…
A:
Q: Question 6: Draw the organic products formed when each alkyne is treated with two equivalents of…
A: When an alkyne is treated with 2 equivalents of HBr then 2 H and 2 Br adds on the triple bond to…
Q: For each step of the metabolic pathway shown give the name of the most likely enzyme to catalyse the…
A: Phase I metabolism: Oxidation, reduction, hydrolysis Phase II metabolism: sulfation, methylation,…
Q: Calculate the number of phosphorus atoms in a 150.0 g sample of diphosphorus pentoxide (P₂O5). Be…
A:
Q: Consider the following two acids and their ionization constants: НСООН Кa = 1.7 x 10-4 HCN K₂ = 4.9…
A:
Q: Consider the following reaction between oxides of nitrogen: NO₂(g) + N₂O(g) →> 3NO(g) The following…
A: ∆G° = ∆H° - T∆S° If ∆H° = Positive, ∆S° = positive Increase in temperature increases T∆S and ∆G°…
Q: Complete and balance the following redox reaction in basic solution. Be sure to include the proper…
A: To balance the reaction in basic solution: 1) write the equation in two halves 2) check if all…
Q: Use the observations about each chemical reaction in the table below to decide the sign (positive or…
A: please find the below attachment.
Q: If you mixed 375 ml of 5.2 M HCO2H with 375 ml of 5.2 M CH3CO2-, what would be the final pH?
A: Question is that if we mixed same volume of formic acid HCO2H and HCO2– solution with same…
Q: Problem 4: C6H12O₂ ¹H NMR th 3.9 4.0 ntegral ratio: 1.0 1.5 или 3.0 1.4 مالا لیا است 1.3 1.2 2.0…
A:
Q: When methanol (CH3OH) is combusted, such as when in a gasoline blend, the following reaction occurs:…
A: we have to calculate the standard free energy for the reaction
Q: Chlorine monofluoride, ClFClF, and iodine monofluoride, IFIF, are interhalogen compounds compounds…
A: To compare the dipole moment of given compounds, we'll find out the Electronegativity difference…
Q: Draw the mechanism of the synthesis of crystal violet with diethyl carbonate, THF and HCl.
A: Crystal violet is synthesized from diethyl carbonate and 4-bromo-N,N-dimethylaniline. This synthesis…
Q: The following chemical reaction takes place in aqueous solution: CuCl₂(aq) + 2NaOH(aq) →…
A: The given reaction is, CuCl2 (aq) + 2 NaOH (aq) → Cu(OH)2 (s) + 2NaCl (aq) In order to write the net…
Q: You performed an isocratic reversed-phase HPLC separation of phenol, toluene and catechol using a…
A: An isocratic reversed-phase HPLC separation of phenol, toluene and catechol is carried out and…
Q: A chemist carried out an elimination reaction of 1,1-dimethyl-2- (dehydrobromination)…
A:
Q: Predict the major organic product formed when the compound shown below is heated with CH3NH₂.…
A: Aminolysis reaction - ester can convert into amides when it heated with amines.
Q: How does chromatography separate the components of leave samples? 2. Can you use chromatography…
A: In a chromatographic system, the sample is transported through the stationary phase by a liquid or…
Q: 2. What is the major product of this reaction? OH A. B. C. D. OH 8 conc. H₂SO4 140°C
A: We have given the reactant is 1-cyclobutylethanol. We need to find the product of the reaction when…
Q: Using the ionization energy values, explain the usual degrees of oxidation of alkali (+1), alkaline…
A: Ionization energy - it is an energy that required to remove 1 electron from outer shell of a gaseous…
Q: Question: Rank the following groups in order of increasing priority a) -H,-Br,-CH₂CH3,-CH₂CH₂OH b)…
A:
Q: 5. From the information given in the textbook, calculate the standard free energy change at 25.0 °C…
A: First we need to calculate the moles of ammonia produced in this reaction. Then using the moles of…
Q: 5. Calculate: Suppose you had 2.0158 grams of hydrogen (H₂). A. How many moles of hydrogen do you…
A: First we need to write a balanced chemical equation of hydrogen and oxygen to produce water. A.)…
Q: Propose an efficient synthesis for the following transformation: CH3 of-ola CH3 CH3 The…
A: This is a transformation reaction involves multi step . To find out the reagents for this…
Q: Macmillan Learning The pressure of a sample of argon gas was increased from 3.39 atm to 8.23 atm at…
A:
Q: Identify which of the following pairs of condensed structural formulas derived from the same…
A:
Q: Write a balanced equation to represent the complete combustion of each of the following. (Use the…
A: The combustion reaction of hydrocarbon generally produces CO2 and water. but we have to balance the…
Q: 4-B Question) Draw the following by Chemdraw? 1 Acetamides 2 Benzamides 3 Benzenesulfonamides 4…
A: This question belong to IUPAC Nomenclature of organic compounds. I have draw all part with the help…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- In the following cases rearrange the compounds as directed : (Delhi 2010)(i) In an increasing order of basic strength :C6H5NH2, C6H5 N(CH3)2, (C2H5)2NH and CH3NH2(ii) In a decreasing order of basic strength :Aniline, p-nitroaniline and p-toluidine(iii) In an increasing order of pKb values :C2H5NH2, C6H5 NHCH3, (C2H5)2NH and C6H5NH2What is the IUPAC for each compound. Specify E/Z if relevanti need help to choose the required regions necessary to prepare the following compound in the presence of a Catalytuc aqueous acid I really need to get the right answer and cheggs answer might be worng
- In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:For 20.7 the solutions for part c and d dont seem right. Shouldn't reacting with 1 eq of H2 just take away the C=C bond and not the C=O bond. (H2 reduces C=C selectively to form a ketone according to page 738. Also the both the C=C and C=O bond should be reduced if excess H2 is usedDuring an acid base extraction of an organic compound dissolved in dichloromethane with sodium bicarbonate (NaHCO3), you do not recover any of the desired extracted product after completing all the remaining steps. What could the most probable reason(s) be for the observed outcome? -The organic compound you want to extract is also a base; an acid is needed -You did not degassed/vent the separatory funnel -You did not shake the separatory funnel enough to react the base with the organic compound -You did not heat the mixture before the extraction -The organic compound evaporate from separatory funnel during the extraction
- Calculate ΔSsys° (J/K) for the catalytic hydrogenation of acetylene to ethane: C2H2(g) + H2(g) → C2H4(g) Substance S° (J/K·mol) C2H2(g) 200.8 H2(g) 130.58 C2H4(g) 219.4Explain the mechanism-Carboxylation—Reaction of RMgX with CO2Carbonic anhydrase catalyzes the hydration of carbon dioxide: H2O + CO2 -> HCO3- + H+The following data is available: (image) - Calculate the Michaelis Menten constants - In the presence of sulfanilamide the Michaelis Menten constants for the reaction are Vmax: 7.57e-5M/s and Km: 20M. What type of compound is sulfanilamide and what is its mechanism of action?
- I am working on a practice assignment for my organic II course and am having difficulty with a question that asks to identify the reaction sequence used to synthesize isopropylcyclopentane. I would really appreciate the help!15.1 mg of β-carotene resulted in a blood plasma level of 43 nmol L-1 of Vitamin A.100% of the β-carotene is converted to Vitamin A (286 g mol-1), and that absorption of β-carotene occurs predominately in the small intestine in an aqueous volume of 600 mL, estimate the equilibrium constant for the reaction Vitamin A (intestine) ⇌Vitamin A (plasma)Compounds Mass 3-nitrophthalic acid used 200 mg = 0.2g 8% aqueous hydrazine used 0.4mL 3-nitrophthalhydrazide obtained 130 mg = 0.13 g sodium hydrosulfite dihydrate 0.6 g luminol obtained 70 mg = 0.07 g compute yield for nitrophthalhydrazide in the first step (assume nitrophthalic acid is limiting reagent) compute yield for luminol in the second step (using nitrophthalhydrazide as limiting reagent) compute yield for the overall reaction

