If [1-14C] Ribose-5-phosphate is incubated with a mixture of purified transketolase- transaldolase-phosphopentose isomerase- phosphopentose epimerase and glyceraldehyde-3-phosphate Fructose- 6-phosphate will be labeled in the 1 and 3 positions.
If [1-14C] Ribose-5-phosphate is incubated with a mixture of purified transketolase- transaldolase-phosphopentose isomerase- phosphopentose epimerase and glyceraldehyde-3-phosphate Fructose- 6-phosphate will be labeled in the 1 and 3 positions.
Chapter22: Medications Used For Gastrointestinal System Disorders
Section: Chapter Questions
Problem 10RQ
Related questions
Question
Multiple answers are accepted for this question
![Please save all answers. Questions that you save can still be edit
) You have 1 UNSAVED ANSWER -
SAVE ALL ANSWERS
Question 31
Which of the following statements are True?
O Multiple answers: Multiple answers are accepted for this question
Select one or more answers and submit. For keyboard navigation... SHOW MORE V
If [1-14C] Ribose-5-phosphate is incubated with a mixture of purified
transketolase- transaldolase-phosphopentose isomerase-
a
phosphopentose epimerase and glyceraldehyde-3-phosphate Fructose-
6-phosphate will be labeled in the 1 and 3 positions.
Starting with Glucose 6-phosphate labelled in carbon number 1 with
radioactive carbon and taken through the oxidative phase of the
Pentose Phosphate Pathway and the first transaldolase reaction gives
b
rise to a 7-carbon intermediate labeled with radioactive carbon in
carbon number 1.
Avidin is a protein that binds extremely tight to biotin. So avidin
would be a potent inhibitor of Acetyl CoA Carboxylase.
The two electrons derived from the a-Ketoglutarate Dehydrogenase
d
complex come from dihydrolipoic acid.
In Fatty Acid synthesis carbon atoms 15 and 16 of Palmitic acid are
e
donated by Acetyl-CoA.
Although HCO- is required for fatty acid synthesis its carbon atom](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fd5cc39c9-8b2b-4beb-a3cb-5d1bdcb587ab%2F2e9c00af-9b53-477b-836f-2ac10f63fa68%2Fvwi445l_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Please save all answers. Questions that you save can still be edit
) You have 1 UNSAVED ANSWER -
SAVE ALL ANSWERS
Question 31
Which of the following statements are True?
O Multiple answers: Multiple answers are accepted for this question
Select one or more answers and submit. For keyboard navigation... SHOW MORE V
If [1-14C] Ribose-5-phosphate is incubated with a mixture of purified
transketolase- transaldolase-phosphopentose isomerase-
a
phosphopentose epimerase and glyceraldehyde-3-phosphate Fructose-
6-phosphate will be labeled in the 1 and 3 positions.
Starting with Glucose 6-phosphate labelled in carbon number 1 with
radioactive carbon and taken through the oxidative phase of the
Pentose Phosphate Pathway and the first transaldolase reaction gives
b
rise to a 7-carbon intermediate labeled with radioactive carbon in
carbon number 1.
Avidin is a protein that binds extremely tight to biotin. So avidin
would be a potent inhibitor of Acetyl CoA Carboxylase.
The two electrons derived from the a-Ketoglutarate Dehydrogenase
d
complex come from dihydrolipoic acid.
In Fatty Acid synthesis carbon atoms 15 and 16 of Palmitic acid are
e
donated by Acetyl-CoA.
Although HCO- is required for fatty acid synthesis its carbon atom
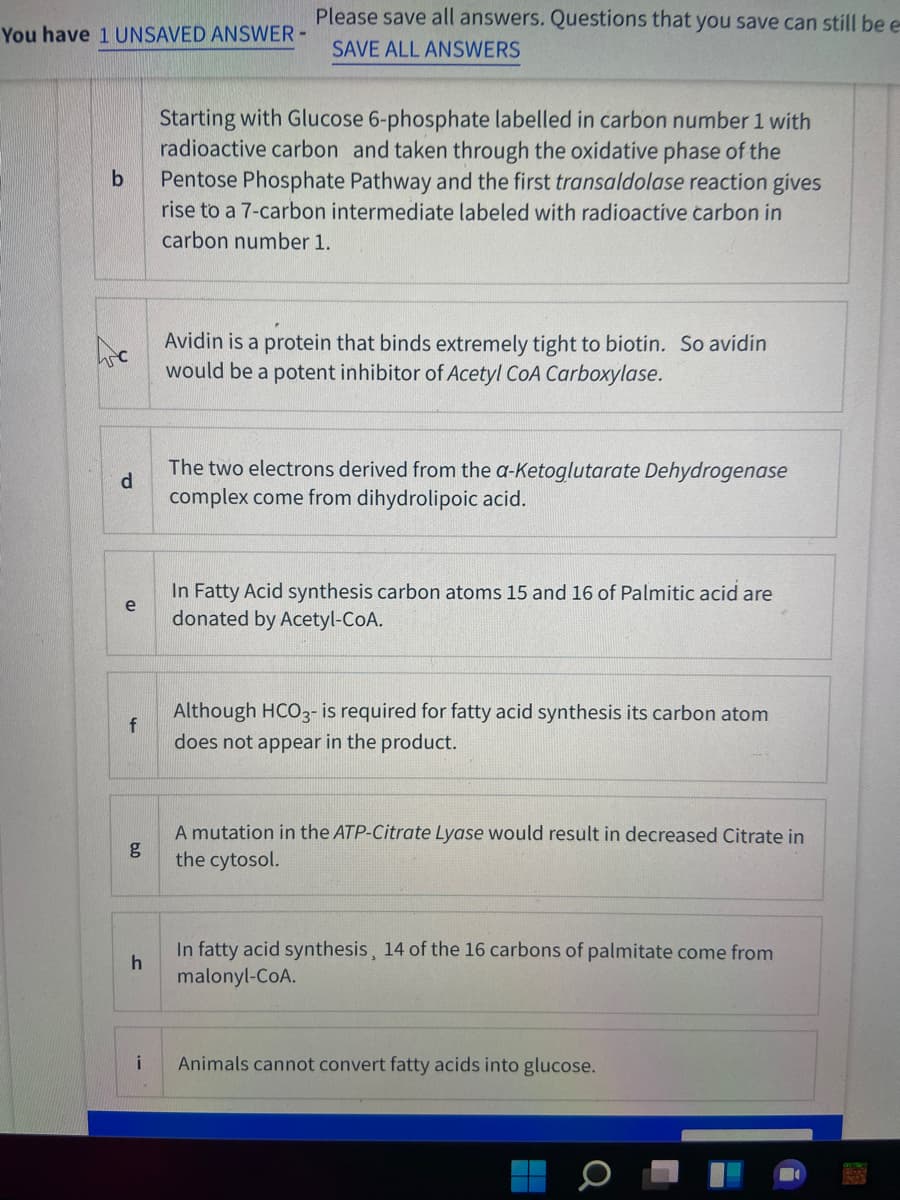
Transcribed Image Text:Please save all answers. Questions that you save can still be e
You have 1 UNSAVED ANSWER-
SAVE ALL ANSWERS
Starting with Glucose 6-phosphate labelled in carbon number 1 with
radioactive carbon and taken through the oxidative phase of the
Pentose Phosphate Pathway and the first transaldolase reaction gives
rise to a 7-carbon intermediate labeled with radioactive carbon in
carbon number 1.
Avidin is a protein that binds extremely tight to biotin. So avidin
would be a potent inhibitor of Acetyl CoA Carboxylase.
The two electrons derived from the a-Ketoglutarate Dehydrogenase
d
complex come from dihydrolipoic acid.
In Fatty Acid synthesis carbon atoms 15 and 16 of Palmitic acid are
e
donated by Acetyl-CoA.
Although HCO3- is required for fatty acid synthesis its carbon atom
does not appear in the product.
A mutation in the ATP-Citrate Lyase would result in decreased Citrate in
the cytosol.
In fatty acid synthesis, 14 of the 16 carbons of palmitate come from
malonyl-CoA.
i
Animals cannot convert fatty acids into glucose.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

