for E, values, calculate AG" for oxidation of malate by malate dehydrogenase.
Q: 182. Pyruvate dehydrogenase is inhibited allosterically by- a) AMP c) NADH b) Pyruvate d) Insulin…
A: Pyruvate dehydrogenase is regulated by :- 1) Allosteric regulation - components of PDH complex -…
Q: Briefly describe the dual role of lipoic acid in the pyruvate dehydrogenase complex
A: Lipoic acid is essentially used in aerobic processes. It is important biological compound for the…
Q: Describe the common characteristic among the reactions catalyzed by pyruvate dehydrogenase (PDH),…
A: Pyruvate dehydrogenase (PDH), isocitrate dehydrogenase (ID), and alpha-ketoglutarate dehydrogenase…
Q: Glutamate dehydrogenase functions to produce ammonia for the urea cycle. Use the standard reduction…
A: Glutamate dehydrogenase is the enzyme that is responsible for the production of ammonia. Ammonia…
Q: The clinical symptoms of two forms of galactosemia—deficiency of galactokinase or of UDPglucose:…
A: Galactosemia is a rare gentic metabolic disorder that affects an individual's ability to metabolize…
Q: Explain how the conversion of alanine to pyruvic acid (pyruvate) can be identified as an oxidation…
A: Oxidation is the gain of oxygen, loss of electrons, and loss of hydrogen from an atom. The oxidation…
Q: The standard reduction potential for ubiquione (A or coenzyme Q) is .045 V, and the standard…
A: Reduction potential determines the tendency of a chemical substance to acquire electrons and thereby…
Q: In the estimation of glucose with benedict's reagent, what is resposnible for the white precipitate…
A:
Q: Hypercholesterolemic individuals taking statins are sometimes advised to take supplements of…
A: The high cholesterol level in the blood of the body is referred to as hypercholesterolemia. It is an…
Q: Describe the reaction catalyzed by each enzyme: (a) aspartate transaminase; (b) glyceraldehyde…
A: Introduction: a) Transamination is the transfer of an amino group from one amino acid to an α-keto…
Q: full oxidation of 2 moles of glucos cose produce 36ATP O
A: Overall ATP production for glucose oxidation : C6H12O6 + 6CO2 + ADP +Pi -----> 6CO2 + 6H2O +ATP
Q: write a balanced equation for the overall reaction catalyzed by each of the three enzymes in the…
A: The pyruvate dehyrdrogenase complex is complex of 3 enzymes that plays a role in the metabolsim by…
Q: Ribose-5-phosphate is produced by oxidative decarboxylation of 6-phosphogluconate catalyzed by the…
A: Above questions is from PENTOSE PHOSPHATE PATHWAY.
Q: For myristic acid, C 13H 27CO 2H: (a) How many molecules of acetyl CoA are formed from complete…
A: β-oxidation : β-oxidation of fatty acids involves four series of reactions. In this reaction the…
Q: Which of the following statements are FALSE? a A compound is an inhibitor of…
A: Glyceraldehyde 3-phosphate dehydrogenase is an enzyme that catalyzes the important intermediate…
Q: Are any of the intermediates in the β-oxidation pathway chiral? Explain.
A: Chiral: it is defined as a molecule with lack of internal plane of symmetry and has four different…
Q: In this experiment, thearubigins were studied for their ability to inhibit the activity of amylase.…
A: Here i discuss about the questions regarding the experiment to check the amylase activity.
Q: Predict which one of the five steps of the α-ketoglutarate dehydrogenase complex reaction is…
A: Alpha-ketoglutarate dehydrogenase is a highly regulated enzyme, which could determine the metabolic…
Q: Considering the evidence that led Krebs to propose a cyclic pathway for oxidation of pyruvate,…
A: Introduction: Metabolically plants and animals are different in several metabolic stages. Plants…
Q: 2. Isocitrate dehydrogenase is an enzyme of 2 subunits with two active sites, catalyzing the…
A: Metabolism includes biosynthesis/ reduction (an anabolic process) and oxidation (catabolic…
Q: Five coenzymes are required by a-ketoglutarate dehydrogenase, the enzyme in the citric acid cycle…
A: The citric acid cycle is also called as TCA cycle is a series of chemical reactions used by all…
Q: . In the experiment shown below, fibroblasts obtained from a normal subject (closed symbols) or from…
A: The LDL (low-density lipoprotein) level regulation depends on the HMG CO A reductase.
Q: A sample of glycogen from a patient with liver disease is incubated with orthophosphate,…
A: Glycogen is the storage form of glucose. it is a large, branched polymer of glucose residues that…
Q: Outline the mechanism of the conversion of a-ketoglutarate to succinylCoA catalyzed by…
A: The last common mechanism for the aerobic oxidation of fuel molecules is the citric acid cycle. In…
Q: The half-reactions involved in the lactate dehydrogenase (LDH) reaction and their standard reduction…
A: Redox potential is a measure of the tendency of a chemical species to acquire electrons from or lose…
Q: What concentration of Proinebrium is required to achieve 50% inhibition of ethylene glycol…
A: Perineurium can be defined as a protective sheath present around the nerve fascicle and is made p of…
Q: Citrate Synthase exists as a dimer but can catalyze the formation of only 1 citrate from Acetyl-CoA…
A: The formation of citrate from Acetyl- CoA, and Oxaloacetate is catalyzed by the enzyme citrate…
Q: Consider the complete oxidation of one mole of the fatty acyl CoA of myristic acid (14:0). Determine…
A: Beta oxidation is the oxidation of fatty acid at beta carbon. It comprises of three stages:…
Q: Pernicious anemia. Purine biosynthesis is impaired by vitamin B12 B12 deficiency. Why? How might…
A: Pernicious anemia is a type of anemia. This is a condition in which the blood has a lower than…
Q: In own words, give 5 or more reasons why most of the clinical features of the diseases…
A: Mitochondria are an essential component of eukaryotic cells, and their failure has been linked to a…
Q: Thiolase is homologous in structure to the condensing enzyme. On the basis of this observation,…
A: Thiolases are enzymes that are involved in the mevalonate pathway. They convert two units of…
Q: What are the three Krebs Cycle inborn errors mentioned in the paper? Why are they called…
A: Inborn error of metabolism is a type of genetic inheritance condition where it blocks the metabolic…
Q: a) Write an equation for the reaction catalyzed by fumarate reductase. b) NADH produced by the…
A:
Q: Suggest a name for an enzyme that catalyzes each of t a.) Oxidation of nitrite b.) Decarboxylation…
A: An enzyme is a substance that acts as a catalyst in living organisms, regulating the rate at which…
Q: [methyl-14C]Pyruvate was administered to isolated liver cells in the presence of sufficient malonate…
A: When the radioactive pyruvate at methyl carbon (carbon number 3) would be administered to the liver…
Q: Describe the roles of dUTPase, thymidylate synthase, and dihydrofolate reductase in the synthesis of…
A: The de novo dTMP synthesis pathway catalyzes conversion of deoxyuridylate (dUMP) to dTMP.
Q: Considering the evidence that led Krebs to propose a cyclic pathway for oxidation of pyruvate,…
A: The Citric acid cycle, or the Tri-carboxylic acid cycle is commonly known as the Kreb's cycle, which…
Q: Explain the role of copper sulfate, CuSO4 as a component of the Benedict’s reagent for the…
A: Benedict solution contains sodium citrate, sodium carbonate, and the pentahydrate of copper(II)…
Q: Malonate competes with succinate in the succinate dehydrogenase reaction. Explain why increasing the…
A: Enzymatic inhibitors decrease enzyme activity. Where the enzyme inhibition is of 3 types…
Q: Why most of the clinical features of the diseases Krebs Cycle inborn errors ( a- ketoglutarate…
A: Krebs cycle : This pathway plays an important role in the living cell metabolism also known as…
Q: , which indicates AG for each glycolytic reac- tion under intracellular conditions. Assume that…
A: Glycolytic : It is the metabolic pathway that converts glucose C6H12O6 into pyruvate, CH3COCOO-, and…
Q: Amount (in μmol) 120 100 80 60 40 20 0 0 50 100 150 200 Time (s) 250 300 350
A: Salivary amylase is the Enzyme that acts on starch to break them into glucose units. Starch---->…
Q: When the identical subunits of chicken liver fatty acid synthase are dissociated in vitro, all of…
A: The fatty acid synthase is a multi-enzyme protein. It catalyzes the synthesis of fatty acids. In…
Q: In affinity chromatography experiment, Cibarcon-blue sepharose is used to purify LDH (lactate…
A: Affinity Chromatography is a Chromatographic techniques which is employed to separate the proteins…
Q: Consider the complete oxidation of one mole of arachidic acid (20:0). ___________ a. How many…
A: Beta-oxidation is the process that is responsible for the breakdown of fatty acids. Beta-oxidation…
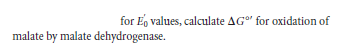
The reaction is
NAD+ + Malate ---> NADH + oxaloacetate + H+
Step by step
Solved in 2 steps

- Extending the Mechanism of Methylmalonyl-CoA Mutase to Similar Reactions Based on the mechanism for the methylmalonyl-CoA mutase (see problem 14), write reasonable mechanisms for the following reactions shown.Examine the ActiveModel for alcohol dehydrogenase and describe the structure and function of the catalytic zinc center.Radiolabeling with 14C-Glutamate Describe the labeling pattern that would result from the introduction into the TCA cycle of glutamate labeled at Cy with 14C.
- Referring to for E′0 values, calculate ΔG°′ for oxidation of malate by malate dehydrogenase.Outline the mechanism of the conversion of a-ketoglutarate to succinylCoA catalyzed by α-ketoglutarate dehydrogenase complex. Include all products, coenzymes, and reactions in your discussion.Alpha-ketoglutarate dehydrogenase exhibits feedback inhibition by
- Why Alpha-Ketoglutarate dehydrogenase (a-KGDH) called deficiency? What are the probable causes? Explain.For myristic acid, C 13H 27CO 2H: (a) How many molecules of acetyl CoA are formed from complete β-oxidation? (b) How many cycles of β-oxidation are needed for complete oxidation?Hypercholesterolemic individuals taking statins are sometimes advised to take supplements of coenzyme Q. Explain.
- For the following aspartase reaction in the presence of the inhibitor hydroxymethylaspartate, determine Km and whether the inhibition is competitive or noncompetitive. You have to plot thegraph on the graph paper and also by using excel.[S] V, No Inhibitor V, Inhibitor Present(molarity) (arbitrary units) (same arbitrary units) 1 x 10-4 0.026 0.0105 x 10-4 0.092 0.0401.5 x 10-3 0.136 0.0862.5 x 10-3 0.150 0.1205 x 10-3 0.165 0.142The half-reactions involved in the lactate dehydrogenase (LDH) reaction and their standard reduction potentials are (see attached)). Calculate ΔG at pH 7.0 for the LDH-catalyzed reduction of pyruvate under the following conditions: (a) [lactate]/[pyruvate] = 1 and [NAD+]/[NADH] = 1. (b) [lactate]/[pyruvate] = 160 and [NAD+]/[NADH] = 160. (c) [lactate]/[pyruvate] = 1000 and [NAD+]/[NADH] = 1000. (d) Discuss the eff ect of the concentration ratios in Parts a–c on the direction of the reaction.In the complete b-oxidation of stearic acid (18:0) How many moles of acetyl CoA are produced? How many times would the fatty acid pass through the fatty acid spiral for complete oxidation?

