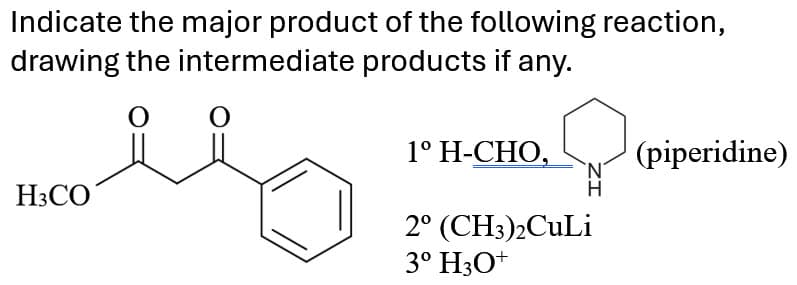
Indicate the major product of the following reaction, drawing the intermediate products if any. H3CO 1° H-CHO, (piperidine) N 2° (CH3)2CuLi 3º H3O+
Q: None
A:
Q: None
A: Step name of the compound is "2-bromo-7-methyloctan-3-one" Steps followed while naming are :- 1.…
Q: Question 6 Scenario: The 2nd year Food Science & Technology experienced EB during pressing of juice…
A: Answer the given question.
Q: A student reads a barometer in the laboratory and finds the prevailing atmospheric pressure to be…
A:
Q: B. Written questions 1. Draw the Newman projection of 4 (four) molecules below with your eyes in the…
A: Step 1: Identify the Bond for RotationLocate the carbon-carbon (C-C) bond you want to visualize the…
Q: please solve this step by step with explaination
A: EXPLANATION It is close since a bigger hydration entropy shows the molecule is easily soluble in…
Q: 13 Draw the complete arrow pushing mechanism for the following reaction. Indicate the Lewis acid and…
A: References Smith, M. B. (2020). March's advanced organic chemistry: reactions, mechanisms, and…
Q: Considering which of the OH groups should be more accessible to MsCl, suggest a mechanism for the…
A: The objective of the question is to propose a mechanism for the rearrangement of cyclobutane to…
Q: Step by step chemistry process of spf and how it is made Chemicals used in spf Bad and good…
A: Step 1:The Manufacturing Steps:1. Mixing the Base: Sunscreen usually comes in lotions or creams.…
Q: How long will it take for the concentration of A to decrease from 0.500 M to 0.200 M in the…
A: Step 1:Step 2:
Q: Conjugate acid base pKa 2) Consider the reaction B: () + H3O+ →>> BH(+) + H2O. For the following…
A:
Q: None
A: Step 1:(a)Let me explain the net ionic equation for the reaction between chromium(III) chloride and…
Q: 2. Draw the missing structure(s) in each of the following reactions. The missing structure(s) can be…
A: Step 1: Step 2: Step 3: Step 4:
Q: A student dissolves 12.2 g of lithium chloride (LiC1) in 200. g of water in a well-insulated open…
A: Step 1: Step 2: Step 3: Step 4:
Q: Please classify and explain the following as either aromatic (A) or nonaromatic (NA)
A: Step 1:To answer this question, we must be familiar with the 4 laws of aromaticity. 1. It must be…
Q: Please give the major product of any of the given letter a, b, c or d. Explain the mechanism
A: 1) a)In the given Image, the overall reaction represents the Williamson ether synthesis, where an…
Q: Dinitrogen tetraoxide, a colorless gas, exists in equilibrium with nitrogen dioxide, a reddish brown…
A: Step 1: (1) First reaction:N2 (g) + 2O2 (g) ⇌ N2O4 (g) K1 = [Product]/[Reactant]K1 =…
Q: None
A: The two compounds in the image are constitutional (structural) isomers. So the answer is option…
Q: For the fusion reaction shown, calculate the change in energy of the reaction in units of joules per…
A: Step 1: The reaction: 12H+12H→23He+01nThe atomic mass of 12H=2.01410uThe atomic mass of…
Q: Describe how enzymes increase the rates of chemical reactions
A: The passage describes how enzymes act as accelerators for chemical reactions in living things.…
Q: dont provide handwriting solution...
A: The objective of this question is to determine the amount of the reagent in excess that remains…
Q: How much energy must be supplied to break a single aluminum-27 nucleus into separated protons and…
A: The objective of this question is to calculate the energy required to break a single aluminum-27…
Q: Debye and Hückel were able to show that in dilute solutions the activity coefficient i of an ionic…
A: Step 1: Step 2: Step 3: Step 4:
Q: a) Calculate the theoretical percent water for each value of n--divide the sum of the atomic masses…
A: The complete table with their sum of the atomic mass of water, hydrate, and percent of hydrate is…
Q: Draw the structure(s) of the major organic product(s) of the following reaction after aqueous…
A: Given that the compound is 1. Acid chlorides (also known as acyl chlorides) can react with…
Q: None
A: Answer: O-C≡OClF5Hybridisationspsp3d2Electronic geometryLinearOctahedralMolecular…
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the alkoxide side product.…
A: LiAlH4 is a reducing agent. Here LiAlH4 is a source of H+ which attacks carbonyl carbon and then…
Q: Draw a triacylglycerol with 12 carbon chains that are monounsaturated (you can choose the site of…
A: Step 1: Any unsaturated fatty acid's carbon-carbon double bonds can be converted to carbon-carbon…
Q: Convert each pressure measurement to atm. 1275 mm Hg
A: The objective of the question is to convert the given pressure measurement from millimeters of…
Q: Use this information to answer each question: A student constructs an electrochemical cell with a…
A:
Q: 1. Predict the product of the following reaction and show the mechanism. 1) KOiPr, iPrOH 2) PhCH₂Br…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 3 PHASE DIAGRAMS. Sketch the temperature-composition (“T-x") phase diagram for acetone, (CH3)2 CO),…
A: To sketch the temperature-composition (T-x) phase diagram for the acetone-chloroform mixture, we…
Q: Draw the structure of the major organic product of the reaction. CI (CH3)2₂CuLi ether, -78° C Marvin…
A: Gilman's reagent, typically composed of an organocopper compound such as lithium organocopper…
Q: + H-Br Energy I 3. H-Br addition to 3-methylbut-1-ene forms 2-bromo-2-methylbutane as the major…
A: Step 1: Step 2: Step 3: Step 4:
Q: what is the product? n (+ CuLi 1. CULI 2. H₂O 3. 2 NH Ν H+
A:
Q: Which of the following molecules does not possess a complete octet ofelectrons around its central…
A: In determining which molecule has its central atom not possessing octet of electrons, we determine…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts.
A: Molecule with an Aldehyde Group (CHO): The image depicts a molecule with a three-carbon chain ending…
Q: Determine the pH of each solution. a) 0.16 M NH4Cl (Kb(NH3)=1.76×10^−5) b) 0.16 M NaC2H3O2…
A: Step 1 To determine the pH of each solution, we need to understand that NH4Cl and NaC2H3O2 are salts…
Q: 5. What is the product of the following reaction? [2 points] CHCH3 OH H2SO4 ? heat CH3 Ш. CH=CH2 I.…
A:
Q: Consider the relation between the sequences of DNA, mRNA, tRNA, and the resulting polypeptide. Part:…
A: Step 1: DNA Informational Strand to DNA Template StrandThe DNA template strand is complementary to…
Q: None
A: EXPLAINED ABOVE IN DETAILED
Q: 4) An undergraduate researcher, James, was given the task of removing a t-butyl ether from the…
A: The proposed reaction mechanism explains why James obtained the ortho product (C) instead of the…
Q: Which statement is false about the resonance structure? Water molecule has one Lewis structure…
A: False statement: Resonance structures of the same molecule have different total of electrons. True…
Q: What are the uses of NMR in new drug discoveries? Please answer at your own easy words. Please don't…
A: Nuclear Magnetic Resonance (NMR) is a powerful technique used in drug discovery for several key…
Q: dont provide handwriting soliution ......
A: Step 1: given line segment with endpoint (-4, -1) and (0, -2) We have…
Q: Please answer the following chem question
A: Step 1:Step 2:Step 3:
Q: Calculate the solubility (in M) of O2 in the blood of a scuba diver at a depth of 100 feet, where…
A: We can calculate the solubility using the the formula for Henry's Law:Sg=kPgWhere:Sg is the…
Q: Fill in the missing product
A: I can help you with the missing product in the chemical reaction.The aromatic ring in the reactant…
Q: Question 18 Predict the expected product for each reaction and name the correct starting material to…
A: Step 1: Description of the question A question based on reactions of alkenes. An epoxide is given…
Q: 2) What is the product of the following reaction? -N=C=N- ΌΗ
A: This reaction between a carboxylic acid with a carbodiimide is one of the pathways to convert a…
Step by step
Solved in 2 steps with 1 images

- What is the rate law implied by the mechanism given below? CH3COCH3(aq) + H+(aq) ←→ CH3C(OH)CH3+(aq) (fast, reversible) CH3C(OH)CH3+(aq) → CH3C(OH)=CH2(aq) + H+(aq) (slow) CH3C(OH)=CH2(aq) + Br2(aq) → CH3C(OH)CH2Br+(aq) + Br-(aq) (fast) CH3C(OH)CH2Br+(aq) → CH3COCH2Br(aq) + H+(aq) (fast) A. Rate = k[CH3COCH3][H+] B. Rate = k[CH3COCH3] C. Rate = k[CH3COCH3][Br2] D. Rate = k[CH3COCH3]2 E. Rate = k[CH3COCH3][Br2]/[H+] (Answer is A, looking for explanation why!).Match the reactant with the product of its reaction with HClO4. Leave blank if there is no reaction. Group of answer choices K2S KHCO3 HNO3 K2SO3When mixing aniline and acetic acid together, which direction will be favored at equilibrium? C6H5NH2 (aq) + CH3COOH (aq) <--> C6H5NH3+ (aq) + CH3COO-1 (aq)
- Answer choices for each are the following: unimolecular reaction Br- acetone Cl- substitution reaction elimination reaction CH3OH CH3O- F-Present the concerted pathway of the reaction below. (2E)-hex-2-ene + H2 and PtRank the species in each group in order of increasing nucleophilicity. a. CH3CH2S−CH3CH2O−, CH3CO2− in CH3OH b. CH3NH2, CH3SH, CHOH in acetone c.−OH, F−, Cl− in acetone d. HS−, F−, Cl− in CH3OH
- Are the following nucleophiles, electrophiles, they be both a nucleophile or electrophile, or neither? pyridine alkenes Hg+ (ex. Hg(OAc)2 CH3ONa PBr3In this equilibrium reaction, what is the favored direction? forward, resverse, or equlibrim? CH3CH2CH(OH)CH2CH3 + Na-1CH2CH2CH3 <==>Draw the products of the following reactions. Use curved arrows to show where the pair of electrons starts and where it ends up. a. ZnCl2 + CH3OH b. FeBr3 + Br c. AlCl3 + Cl−



