Q: 5. Write the reaction mechanism of the following transformations: a) H,N pH= 5 b) но H*
A:
Q: Given the following elementary reaction what is the molecularity? A + 2B → AB2 A) unimolecular B)…
A: Given reaction, A + 2B -----> AB2 In these reaction, 1 atom of A and 2 atom of B.
Q: Compare the reaction order for this reaction and determine the mechanism. The reaction shown below…
A: Here the question is given two different of mechanism we have to identify which follow first order…
Q: Identify the molecularity of the elementary reaction below: 2NO(g) + Br,(g) → 2NOBr(g) A)…
A:
Q: H + KOH + KI + H. H
A:
Q: Based on the reaction profile (below) for the reaction 2A + B+ C G, what is the overall rate law for…
A:
Q: 7IO3–(aq) + 9H2O + 7H+(aq) ⇆ I2(aq) + 5H5IO6(aq) What is the Kc expression for the reaction?
A: Given :- 7IO3–(aq) + 9H2O + 7H+(aq) ⇆ I2(aq) + 5H5IO6(aq) To be determined :- Kc expression of…
Q: Complete all the chemical equation below. Give the reagent, reactant, product and condition use.…
A: Alkenes are more reactive compared to the alkane due to presence of it's weak pi bond.
Q: Identify the molecularity of the elementary reaction below: 2HI(g)→ H₂(g) + I₂(g)
A: Molecularity is number of molecules react to give product
Q: Consider the following mechanism: 1. ClO-(aq) + H2O(l) ⇌ HClO(aq) + OH-(aq) FAST 2. I-(aq) +…
A: Molecularity: Molecularity of any reaction is the number of molecules that colloid in order to react…
Q: The following reaction 2A → B + C is second order, following the rate law Rate = k[A]². If the…
A:
Q: -1.-1 The rate constant for a second order reaction is 3.33×10² L mol's. The oncentration of the…
A: Order of a reaction is defined as sum of power of concentration of reactant For second order…
Q: Give the mechanism of the reactions: (d) Ме Me Me т-СРВА Me Me OBn Me OBn (e) i. NCS ii. m-CPBA i.…
A: Given reactions:
Q: EtO, Eto NABH, N. ELOH NH 6 5a Ph Ph
A: The above reaction proceeds through a two step mechanism. At first step the bolu hydride group…
Q: OH 1. 2 mol PhMgBr OCH, 2. H;O*
A: In organic chemistry, the reactions can be shown step by step. The electron transfer from a…
Q: 4. A mechanism for a reaction is as follows (all are in gaseous state): CAHSOH2+ H2O CH9OH + H30…
A:
Q: I10- 80 50 B 40 30 A+A 20 10 D+E Reaction coordinate → a. What is the overall reaction? b. Identify…
A: Please note: As per our company guidelines we are supposed to answer only three parts of one…
Q: If you wrote the rate law expression for the reaction A +B C, based on the following data, what…
A: Given reaction is A + B --> C For this reaction rate law expression is rate…
Q: The reaction A+B⟶C+D rate=k[A][B]2 has an initial rate of 0.0140 M/s. What will the initial…
A: Reaction equationA+ B ----> C+DRate = k [A][B] 2Initial rate = 0.0140 M/sWhat will the initial…
Q: 10) Write the mechanism of the following reaction? 1) MgBr :HO: CI: + 2 MgBrX 2) H30®
A:
Q: Consider the following mechanism: Just C (1) ClO−(aq) + H2O(l) ⇌ HClO(aq) + OH−(aq)…
A: The rate of the reaction is referred to the change in the molar concentration in the distinct…
Q: 2A-B+C Two trials of the above reaction are run with the same initial concentration of A The first…
A: Given : We have to calculate rate constant for the second half reaction.
Q: Place the mechanism steps for this reaction in the correct order: H-Br :Bri Br-H t H-Br H-: >
A: The given reaction is, The reaction is cleavage of ether with strong acid. The initial step is…
Q: What is the rate law for the elementary termolecular reaction 3A→ products ? 1. O Rate - kA O Rate…
A: The rate law of a chemical reaction is an equation which corelates the rate of reaction with the…
Q: 35. be deduced from the rate equation of the reaction, CH;COCH, + I2 → CH;COCH21 + H* +F given by…
A: The order of a reaction is defined as the sum of of the powers to which the concentration terms are…
Q: What type of mechanism is the given reaction most likely to follow? A 1. B 2. C D Br excess HBr ОН +…
A: the correct option is as follows:
Q: A mechanism involving the following elementary reactions has been proposed: 2 X (g) → X2 (g) (fast)…
A: The slowest is mainly utilized for the prediction of any certain reaction's rate law. The compound…
Q: Calculate the order of the following reaction: rate = k[A]* %3D Trial [A] Initial Rate (M/s) 1 |0.56…
A: The answer to the following question is-
Q: ki [2] Given the mechanism: NO(g) + O2(g) SNO;(g) [fast, reversible] k-1 (1) k2 NO:(g) + NO(g) →…
A: Please note: As per our company guidelines we are supposed to answer only three parts of one…
Q: Br 1. Mg, Et,0 3. H3O* 2.
A:
Q: Given the reaction mechanism below, identify the catalyst. А+ B -> С+D C + A-> B + E Overall: 2A ->D…
A:
Q: (a) For a reaction A + B → P, the rate is given by Rate = k[A] [B]2(i) How is the rate of reaction…
A: Given: A reaction A + B → P Rate = k[A] [B]2 According to the…
Q: The reaction E + 3B →2C has the following mechanism: 1. E + A + B → D + C 2. 2B + D → C + A What…
A: Intermediate is formed during the reaction and destroy before ending of reaction and never appeared…
Q: 2. Give the mechanism and products for the reaction below. 1. `MgBr 2. + 3. Н, Н.О
A: RMgX i.e. Grignard reagent that is a good source of R- that carry out nucleophilic addition to alkyl…
Q: Consider this two-step mechanism for a reaction. NO2 (g) + Cl2 (9) →CINO2 (g) + Cl (g) Slow NO2 (9)…
A: We know a faster reaction have lowest activation energy and a slower reaction should have highest…
Q: Identify the molecularity of the elementary reaction below: 2HI(g) H2(g) + I2(g) A) unimolecular B)…
A:
Q: (4) Account for the relative rates of the following two reactions: [Fe(CN).J • [*Fe(CN).J*…
A:
Q: The following reaction 2A → B + C is second order, following the rate law Rate = k[A]². If the…
A: Second order reaction means, the rate of the reaction is proportional to the second power of the…
Q: 15. Which of the following steps appearing in a mechanism would be third order overall? SOCh O So: +…
A: “Since there are multiple questions and it is not mentioned that which one has to be solved so I am…
Q: Using the reaction – energy diagram (at the right side), what happens to the activation energy when…
A: Activation energy: When the reactant molecules convert into the products, they required the minimum…
Q: For the following reaction mechanism for the reaction A+2B →D ki A+B k-1 k2 В + с — write the…
A: Expression for d[A]/dt d[A]dt = -k1AB + k-1C
Q: 4. The following is a possible mechanism for a reaction involving hydrogen peroxide in aqueous…
A:
Q: Write out a possible reaction mechanism t the following reactions: CI CI 2 HCI CI CA Br 1 Br2 Br t.
A: In the given reactions there is an addition of chlorine or bromine to it, in the first reaction 2…
Q: Given the following mechanism: NO(9) • Fag) NO,F:(g) NO Fg) NO,F(g) + F(g) Fig) NO,(g) → NO F(9)…
A: Let us discuss these questions.
Q: What is the mechanism of the reaction?
A: The replacement of any atom or group of atoms by an electrophile is termed as electrophilic…
Q: 11. A researcher wants to synthesize some pentaaminefluoromanganese(l) starting with…
A: This substitution reaction undergo through dissociative mechanism. The mechanism of the…
Q: CO,Me OSIME, CO,Me Br A Br
A: Concept ; (3,3) sigmatropic reaction or rearrangement. In this reaction the sigma bond between the…
Q: Using the reaction – energy diagram (at the right side), what happens to the activation energy when…
A:
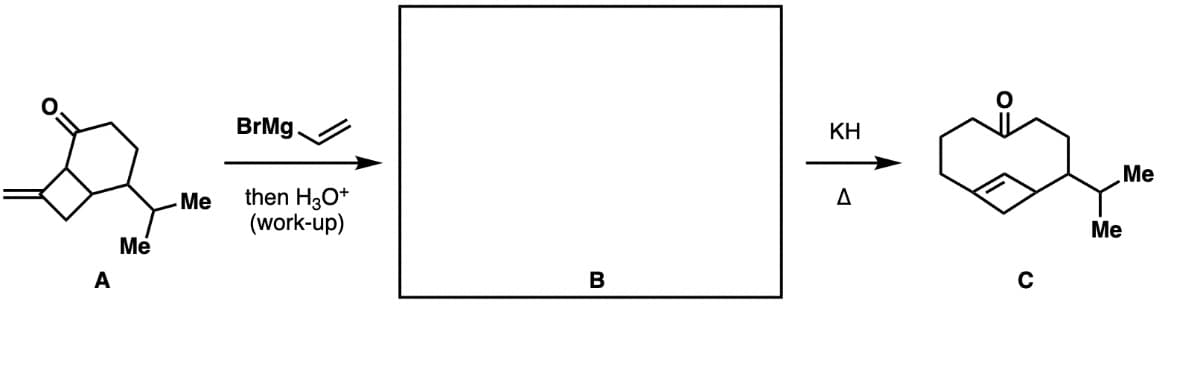
Step by step
Solved in 3 steps with 2 images


