L-ornithine 2 carbamoylphosphate 3 L-citrulline 4 argininosuccinate 5 fumarato 6 L-arginine 7 urea L-Asp L-aspartate CPS-1 carbamoyl phosphate synthetase OTC Omnithine transcarbamoylase ASS argininosuccinate synthetase ASL argininosuccinate lyase ARG1 arginase 1 OH NH₂ H₂N NH CHI ASL CH₂ Coo CH₂ CH₂ coo Which of the reactions is requires energy input? (Answer by choosing the enzyme used)? 2ATPHCONH₂ 2 ADP. PCPS-1 H₂N ARG1 H₂O отс fessels - -8-5-9-8- CH₂ ASS H.M.000 ATP PP, L-Asp AMP
L-ornithine 2 carbamoylphosphate 3 L-citrulline 4 argininosuccinate 5 fumarato 6 L-arginine 7 urea L-Asp L-aspartate CPS-1 carbamoyl phosphate synthetase OTC Omnithine transcarbamoylase ASS argininosuccinate synthetase ASL argininosuccinate lyase ARG1 arginase 1 OH NH₂ H₂N NH CHI ASL CH₂ Coo CH₂ CH₂ coo Which of the reactions is requires energy input? (Answer by choosing the enzyme used)? 2ATPHCONH₂ 2 ADP. PCPS-1 H₂N ARG1 H₂O отс fessels - -8-5-9-8- CH₂ ASS H.M.000 ATP PP, L-Asp AMP
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter20: Electron Transport And Oxidative Phosphorylation
Section: Chapter Questions
Problem 19P
Related questions
Question
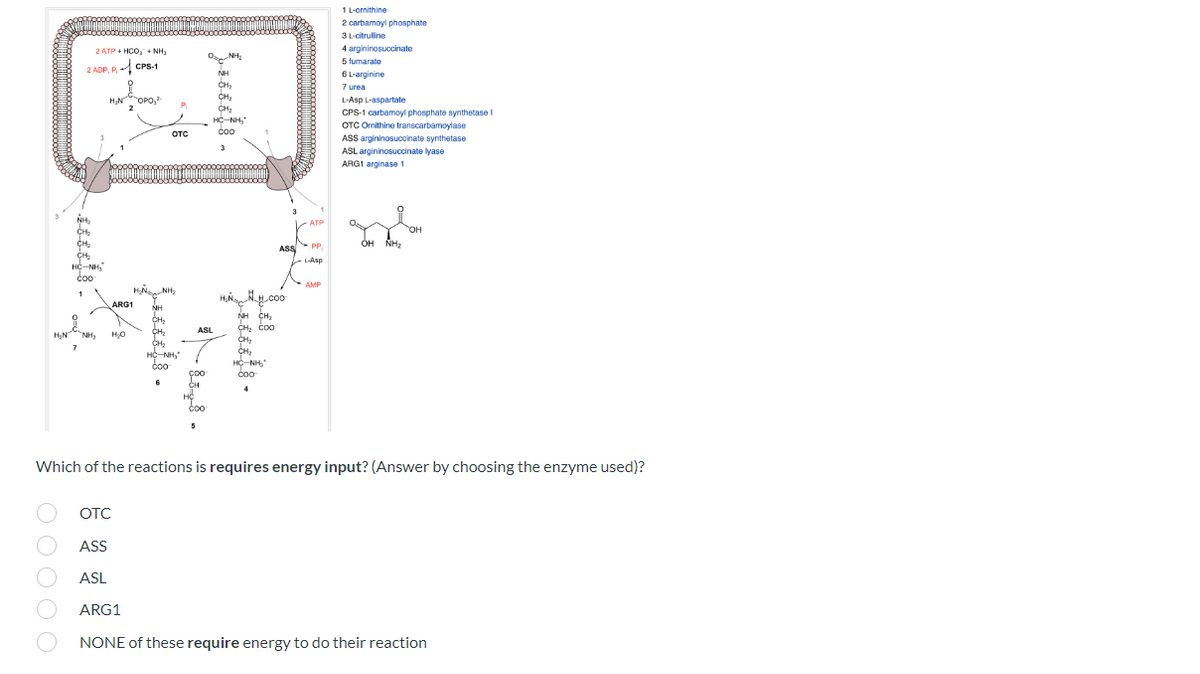
Transcribed Image Text:2 ATP + HCO₂ + NH₂
H₂N
O O O
2 ADP, PCPS-1
H₂N OPO,
2
sessis-+-
*
HỘ NHỎ
NH₂
HNGANH,
NH₂
H₂O
P₁
1
OTC
coo
H₂NHCOO
NH CHI
CH₂ Coo
CH₂
CH₂
HỘ NHỎ
coo
4
Which of the reactions is requires energy input? (Answer by choosing the enzyme used)?
OTC
ASS
ASL
ARG1
NONE of these require energy to do their reaction
ARG1 ΜΗ
CH₂
НО
CH₂
CH₂
HỘ NHỎ
coo
-
6
O-NH₂
NH
CH₂
CH₂
CH₂
HỘ NH
COO
3
ASS
*- 85.8 •
ATP
PP
L-Asp
AMP
1 L-ornithine
2 carbamoyl phosphate
3 L-citrulline
4 argininosuccinate
5 fumarato
6 L-arginine
7 urea
L-Asp L-aspartate
CPS-1 carbamoyl phosphate synthetase I
OTC Ornithine transcarbamoylase
ASS argininosuccinate synthetase
ASL argininosuccinate lyase
ARG1 arginase 1
OH
OH NH₂
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning