lon X has an equilibrium potential of Ex = -50 mV. At the resting membrane potential (-70mV), which direction is the electrical force acting on X? O Outward O Inward O There is no electrical force on X At the resting membrane potential (-70mV), what is the net direction of movement of X* across the membrane? Assume ion channels for X are open. O Direction cannot be determined from this information O Inward O Outward
lon X has an equilibrium potential of Ex = -50 mV. At the resting membrane potential (-70mV), which direction is the electrical force acting on X? O Outward O Inward O There is no electrical force on X At the resting membrane potential (-70mV), what is the net direction of movement of X* across the membrane? Assume ion channels for X are open. O Direction cannot be determined from this information O Inward O Outward
Human Physiology: From Cells to Systems (MindTap Course List)
9th Edition
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Lauralee Sherwood
Chapter4: Principles Of Neural And Hormonal Communication
Section: Chapter Questions
Problem 17RE
Related questions
Question
Asap plz all parts
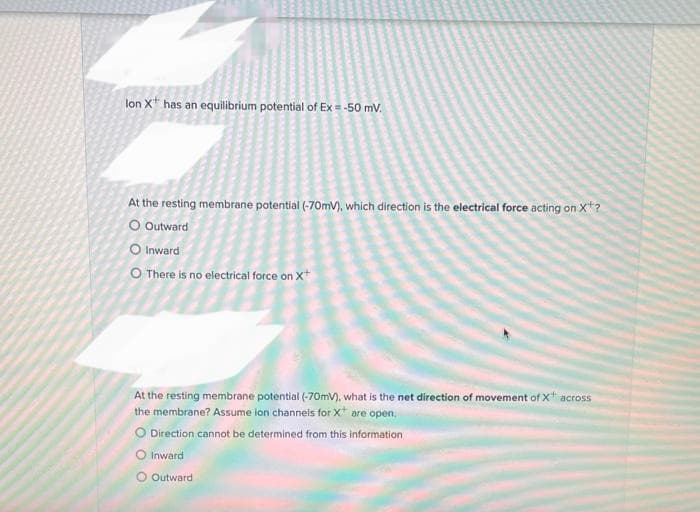
Transcribed Image Text:lon X has an equilibrium potential of Ex = -50 mV.
At the resting membrane potential (-70mV), which direction is the electrical force acting on X*?
O Outward
O Inward
O There is no electrical force on X+
At the resting membrane potential (-70mV), what is the net direction of movement of X* across
the membrane? Assume ion channels for X are open.
O Direction cannot be determined from this information
O Inward
O Outward
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College