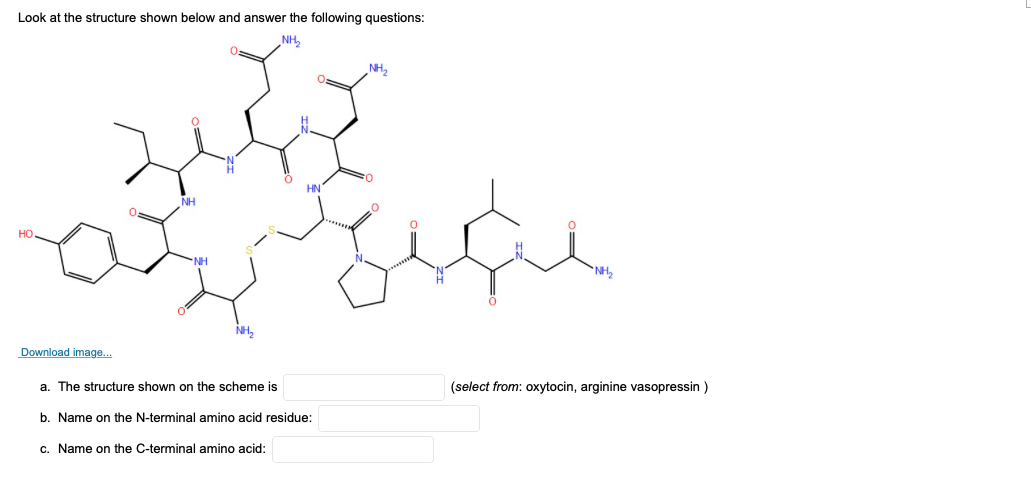
Look at the structure shown below and answer the following questions: HN NH но NH Download image. a. The structure shown on the scheme is |(select from: oxytocin, arginine vasopressin )
Q: Give the force of interaction involved in the protein folding of the protein structure A and C: COO ...
A: Hydrophobic interaction - Interaction between nonpolar group by excluding water molecules
Q: Basal Metabolic Rate (BMR) depend - on All Size of body O Mass of body O
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any sp...
Q: The pH of lemon juice is 2, and the pH of orange juiceis 4. Which of these is more acidic, and by ho...
A: pH refers to power of hydrogen or potential of hydrogen. It indicates the presence of H+ . It is mea...
Q: What is the effect of the NDM gene?
A: NDM (New Delhi Metallo-β-lactamase) gene, found in Enterobacteriaceae, is a genetic element encodin...
Q: What are the important considerations that you have to remember in antibody identification What is t...
A: Introduction: The ABO blood group system is considered to be an important blood group system in huma...
Q: Which of the following equations accurately shows enzyme-catalyzed conversion of substrate to produc...
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation energy...
Q: Which monosaccharide will be produced when sucrose undergoes hydrolysis reaction? O Glucose and arab...
A: Glucose is a monosaccharide and it is a simple sugar that is the most important source ...
Q: What is the difference between obligate and non-obligate chain terminating nucleotides? How were sof...
A: Chain termination is a chemical method which stops the formation of intermediates in a chain propaga...
Q: 16. What would be the expected result in electrolytes if there has been a carry over of anticoagulan...
A: K+ will be elevated up to 7.0mEq/L A healthy plasma/serum K+ level is in the range of 3.5-5.2 mmol/L...
Q: DNA samples were then run in agarose gel electrophoresis against a molecular standard of 100 bp and ...
A: The procedure for isolation of genomic DNA is aimed at obtaining highly pure genomic DNA that can be...
Q: 50 y = 47.5837x + 14.4554 R? = 0.9989 [AMP] = 5 mM y = 37.2228x + 13.0690 R? = 0.9991 [AMP] = 5 mM [...
A: The given plot is the line-weaver Burk plot of the enzyme kinetic data. Line-weaver Burk plot gives ...
Q: Why do the BP and HR change with drops in temperature? Explain the physiologic mechanism behind thi...
A: Blood pressure ( BP) is the pressure exterted by the blood against the walls of blood vessels during...
Q: Which among the following indicates correct the three-letter abbreviation and one-letter abbreviatio...
A: There are twenty naturally occurring amino acids. These amino acids are conventionally represented b...
Q: why chromotography isn't a good fit to detemine mitochondrial RNA polymerase
A: Mitochondrial RNA polymerase (mtRNAP) is vital for biogenesis of mitochondria as well as mitochondri...
Q: Which statement is NOT true of sterols? Question 8 options: Sterols have a structure that incl...
A: Sterols are fat molecules that are commonly found in cells. Sterols are non-polar molecules that are...
Q: A substance that affect the rate of the reaction without being consumed Select one: a. enzymes b. a...
A: The study of chemical reactions catalysed by enzymes is known as enzyme kinetics. Enzymes, which act...
Q: Which of the following is true concerning cellulose? Question 24 options: Can only be broken d...
A: Carbohydrates are composed of carbon, hydrogen, and oxygen in the ratio of 1:2:1. The carbohydrates ...
Q: Applying: Prokaryotic and eukaryotic cells have many differences, but they also share some common fe...
A: Prokaryotes are microscopic single-celled organelles that lack nuclear envelopes. Their cell wall is...
Q: Click on the scheme on a functional group that has positive charge at pH 10.0: H2N. NH2 NH H2N COOH
A: The pKa of the ionizable groups of amino acids, play an important role in determining the net charge...
Q: ATP Synthase is known to catalyze the synthesis of ATP with a ΔG°’ close to zero, and a Keq' close t...
A: The F1/F0 ATP synthase catalyzes the conversion of ADP to ATP coupled with the movement of protons a...
Q: In cation exchange chromatography, positively charged proteins exit the column first. In anion excha...
A: Chromatography is a technique in which components of a mixture are separated. It consist of a mobile...
Q: MULTIPLE CHOICE 1. High density lipoproteins is an example of which kind of protein? A. Regulatory...
A: Proteins are composed of twenty standard amino acids attached together via peptide bonds. These twen...
Q: 1H. please help me in detail. For molecular Mechanism of ATP versus GTP selectivity of adenylate kin...
A: ATP is the major energy carrier in any cell where as GTP is selective for only a few pathways. Adeny...
Q: NEED ANSWER ASAP PLEASE
A: Metabolic pathways are the combination of catabolic and anabolic reactions. Metabolic pathways synth...
Q: The pyruvate dehydrogenase complex catalyzes the oxidative decarboxylation of pyruvate to form acety...
A:
Q: QUESTION 1 Number of reducing end(s) in sucrose (structure shown below) is/are CH2OH H H. он носн, H...
A: Reducing sugars are molecules with a free aldehyde or free keto group. Due to the presence of a free...
Q: Phosphocreatine (G0ʹ = -43.1 kJ/mol) has a higher phosphoryl group transfer potential than ATP (G0...
A: Hi! Since you have posted multiple questions and have not mentioned which to answer, we shall answer...
Q: Consider the following statements: (1) The term sugar is a general designation for both monosacchari...
A: Tge term SUGAR is a general designation for both mono and disaccharide. Glucose (monosaccharide) is ...
Q: Rhodium and other transition metals are generally used in heterogeneous/homogeneouslenzyme catalysis...
A: Enzymes are the biocatalyst which promotes the biochemical reactions in any living organism. The enz...
Q: 2. Sanger sequencing question. a). Why the Sanger sequencing chemistry would not work if too much dd...
A: Sanger Sequencing method was designed by Frederick Sanger in 1977 where he proposed use of electroph...
Q: CH3 CH2-CH,-CH2 CH2-CH,CH2-CH2-CH2 CH2-CH2 ĉ-SCOA
A: Beta oxidation is the process by which long chain acyl CoA are broken down to acetyl CoA. Each round...
Q: An enzyme is present at 100 nM (nanomolar) and has a Vmax value of 25 uM/s (micromolar/second). The ...
A: Enzyme concentration= 100 nM Vmax value= 25 uM/s Km for substrate = 5.2 uM
Q: TRUE OR FALSE Coconut oil will give (positive) result to Molisch test.
A: Molisch test is used to detect presence of carbohydrate in a mixture or sample. This test is based o...
Q: Give examples of proteins classified according to their biological role.
A: Proteins are the long chain of polymers amino acids.Proteins serve as an important structural consti...
Q: Which is responsible for the stabilization of parallel polypeptide chains in a beta-pleated sheet co...
A: Secondary structure refers to the local folded structures that occur inside a polypeptide as a resul...
Q: O His-Tyr Question 23 At low salt concentration, proteins are less soluble. Changing the solubility ...
A: Salting out : Protein precipitation at the high salt concentration
Q: The ammonium sulphate-precipitated phycocyanin (Sample 3) still contains a lot of other proteins. Br...
A: Phycocyanin - Accesory pigment to chlorophyll and is the pigment-protein complex belongs to phycobil...
Q: what type of inhibition is shown and where does the inhibitor bind to the enzyme?
A: Enzymes are usually protein molecules which catalyzes the biochemical reaction by decreasing the act...
Q: Give the systematic names and the first three digits in the E.C. classifications of the enzymes cata...
A: Enzymes are biocatalyst. They increase the speed of reaction by lowering the activation energy. A fo...
Q: Calcitonin reduces Ca+ from the blood
A:
Q: 7. To improve the digestion of food proteins in chronic inflammatory-dystrophic disorders of the pan...
A: Chronic inflammatory dystrophies are a disorder of pancreas also called chronic pancreatitis. In thi...
Q: Which among the following statements is correct? Group of answer choices Both statements are incor...
A: Proteins are the linear heteropolymers of twenty standard amino acids attached together via peptide ...
Q: What is the best interpretation from the results?
A: 1. Negative ninhydrin test shows that the sample does not contain free amino acids. 2. Positive xant...
Q: Define about primary miRNAs (pri-miRNAs) ?
A: MicroRNAs (miRNAs) are small regulatory RNAs derived from stem-loop sections of original transcripts...
Q: The protein illustrated in the attached picture was isolated from a superhuman being. Scientists the...
A: Introduction: Proteins are also known as polypeptides are organic compounds that are made up of amin...
Q: Analyzing: Analyze the following pair of monosaccharides and determine which of the following is an ...
A: Since there are multiple questions and they are not interlinked, as per our company guidelines only ...
Q: 1. Cite five diseases that could be diagnosed with the determination of enzyme concentration. 2. Giv...
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since yo...
Q: Choose from the following pairs. Which are constitutional isomers? Pair A Br
A: constitutional isomers are structural isomers that they differ in structure but have the same formul...
Q: The enzymatic activity of an enzyme with Kg = 2 mM that converts substrate S into product P is measu...
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that o...
Q: Which of the following carbohydrates have two different types of glycosidic linkages present? more t...
A: Glycosidic linkage : It joins 1 carbohydrate molecule to another group/carbohydrate and is present i...
Step by step
Solved in 3 steps with 1 images

- You have a soluble protein that is highly flexible and is only 23 kDa in size. What is the most suitable technique (X-ray crystallography, NMR, cryo-EM) for structure determination of this protein? Explain your reasoning.What could be the implications if there is a misfolding in the protein structure?Illustrate the structure of a peptide with the given sequence below at the given conditions. Serine-Threonine-Glutamic Acid-Lysine-Aspartic Acid-Arginine Form with net charge: -1 Form with net charge: +3 Dominant form at pH 7
- Which of the numbered bonds in this polypeptide fragment are peptide bond(s)? (Select all that apply!)You are working in a bioengineering lab, and you are asked to create a peptide with a helix with length of 3 nm, a beta strand with length of 35 Å, and a linker containing 25 amino acid residues. a. How many amino acid residues are in this peptide? b. What is the mass of the peptide? 2. Why is quinary structure necessary for a full understanding of how drugs work in cells?In the given structure below, 1. What protein was used as the template for modelling the structure of the assigned amino acid sequence? 2. How many alpha-helices and beta-sheets are there?
- Please calculate and plot the charge on a peptide with the sequence of NEYK over the pH range from 1 to 13. Please use the pKa values from the table. You may calculate the charge at 1 pH unit intervals.What if the protein does not have a primary structure, what is its implication? Explain and give example.What is the primary reason the a-helix conformation in polypeptides such a stable form? A. The a-helix structure is stabilized by hydrophobic interactions. B. The a-helix structure is stabilized by intra-helical hydrogen bonds. C. The a-helix structure is stabilized by disulfide bonds. D. The a-helix structure is stabilized by proline residues.
- Consider the following peptide to answer the questions below: A M A K K E S H I F T A I D E What is the total charge of this peptide at pH = 5? What is the N terminal amino acid (full name)? C terminal amino acid? How many fragments would result if this peptide underwent proteolytic cleavage with V-8 protease and what are these fragments (list them)? Would this peptide be a candidate to form an alpha helix? Why or why notDraw the structure of the amino acid(Arginine) at the pH of 8.55Calculate the Rf values for each of the amino acids and enter your data in the table.Based on your calculations, what is the identity of the Unknown amino acid? Rank the amino acids from most hydrophilic to most hydrophobic

