Match each of the following with its correct answer The following reaction is : C (s) + 2 S (0) + 87.9 kJ → CS2 () Choose... Choose... inversely endothermic exothermic Based on Charles law, the volume occupies by any sample of gas at a constant pressure is ......... proportional to the absolute temperature directly Choose... Based on Grahams law , the rate of effusion of the gas is proportional to the square root of the molar mass Choose... Based on Boyle's law, the volume occupies by any sample of gas at a constant temperature is proportional to the pressure, Choose... The following reaction is : CH,OH + 3/2 0, - CO 2(9) + 2H,00 + 726.4 kJ (1) Choose...
Match each of the following with its correct answer The following reaction is : C (s) + 2 S (0) + 87.9 kJ → CS2 () Choose... Choose... inversely endothermic exothermic Based on Charles law, the volume occupies by any sample of gas at a constant pressure is ......... proportional to the absolute temperature directly Choose... Based on Grahams law , the rate of effusion of the gas is proportional to the square root of the molar mass Choose... Based on Boyle's law, the volume occupies by any sample of gas at a constant temperature is proportional to the pressure, Choose... The following reaction is : CH,OH + 3/2 0, - CO 2(9) + 2H,00 + 726.4 kJ (1) Choose...
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter16: Spontaneity, Entropy, And Free Energy
Section: Chapter Questions
Problem 121MP: Consider a sample containing 5.00 moles of a monatomic ideal gas that is taken from state A to state...
Related questions
Question
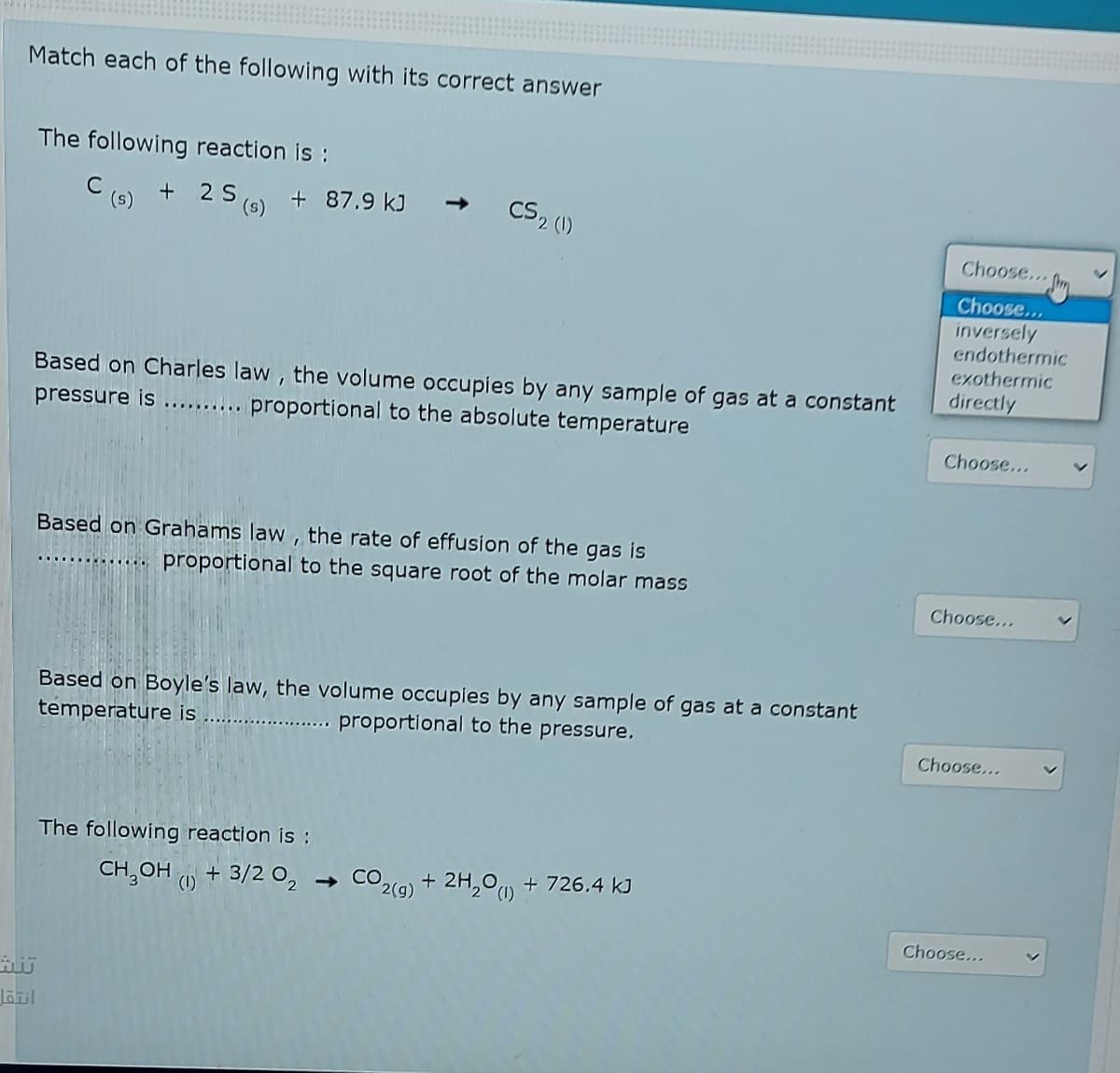
Transcribed Image Text:Match each of the following with its correct answer
The following reaction is :
C
(s)
+
(6) + 87.9 kJ -
CS2 (1)
Choose...
Choose...
inversely
endothermic
exothermic
Based on Charles law, the volume occupies by any sample of gas at a constant
pressure is .......... proportional to the absolute temperature
directly
Choose...
Based on Grahams law , the rate of effusion of the gas is
proportional to the square root of the molar mass
...... O
Choose...
Based on Boyle's law, the volume occupies by any sample of gas at a constant
temperature is
proportional to the pressure.
Choose...
The following reaction is :
CH,OH O + 3/2 O,
- CO2(g) + 2H,°u + 726.4 kJ
(1)
Choose...
Jail
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning