Q: Consider the I-131 nucleus that is used to treat thyroid. Mass of I-131 is 130.78302 amu. 1) what is...
A: To answer questions regarding I-131 nucleus.
Q: pH Titration Curve for titrating 29.0 mL of Aqueous Lithium hydroxide with 0.788 mol/L Hydrobromic A...
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for...
Q: Determine the value of delta Smix for a gaseous mix that contains 0.25 bar of 1-butene and 0.18 bar ...
A: Given:- Determine the value of ∆Smix for a gaseous mix that contains 0.25 bar of 1-butene and 0....
Q: A volume of 500.0 mL of 0.160 M NAOH is added to 545 ml. of 0.250 M weak acid (K, = 8.52 x 10-5). Wh...
A:
Q: 1, The pke value of cacium luoride is 10.41. Colculate the solubilly in g/ at 25 Cin a pure water b ...
A: Given: pKspCaF2 = 10.41 To determine solubility in g/L at 25oC in a) pure water b) 0.0200 M Calcium ...
Q: How many mL of water must be added to 300.0 mL of 0.750 M HCl to dilute the solution to 0.25 M?
A: At initial , volume(V1) = 300mL Molarity (M1) = 0.750M of HCl We have to make a dilute HCl solution...
Q: In the transamination of an alpha amino acid plus alpha-ketoglutarate, what are the two products of ...
A: In transamination reaction, transfer of an amino group (-NH2) takes place from the amino acid to the...
Q: The change in entropy describes if the final state is more expansive than the initial state. If it ...
A: Entropy is degree of randomness or disorderness. As if we dissolve borax in then final state is exp...
Q: The value of ΔGo for the reaction, N2(g) + 3 H2(g) ⇌ 2 NH3(g) is -32.90 kJ at 298 K. Calculate the ...
A:
Q: Which of compounds l-V corresponds to the infrared spectrum (attached). Provide a clear rationale fo...
A: Infrared spectroscopy give information about what type of functional group.
Q: pre-equivalence end stage equivalence post-equivalence
A: Titrant is the substance which is added in analyte to major the pH . If we add more titrant. Then th...
Q: (h) How do you prepare the following alkyl halide using the Gabriel synthesis reaction? Provide the ...
A: Gabriel synthesis is one the method used to prepare primary amine here we are required to show the m...
Q: To 0.360 L of 0.150 M NH 3 H is added 0.140 LL of 0.110 M MgCl 2.
A: first calculate the moles of every compound used n(NH3)= (0.150 mol/L )(0 .360 L) = 0.054 moles NH3 ...
Q: 1. A solution is prepared by dissolving 12 grams of turmeric powder in 350 grams of water. Calculate...
A: 1) Given, Mass of turmeric powder = 12 g Mass of water = 350 g Mass percent of each component of the...
Q: Data -. Endgroup analysis by titration: .8 mols of 1,6-hexanediamine were reacted vith 9.8 mols of a...
A:
Q: In an experiment to determine the amount of aspirin in a tablet, one of the bottles of NaOH used was...
A: Here we required to predict the effect of concentration NaOH in determination of the concentration o...
Q: Paragraph Styles 3. 4. 2. Determine the weight needed to create the concentration 475 ml of the foll...
A: Since you have asked multiple question, we will solve the first question for you. If you want any sp...
Q: For the reaction 2A + B + 2C D + 2E, the rate law is: rate =k[A]2[B][C] . Which of the following sta...
A: The Rate of a reaction is proportional to the initial concentration of reactants raised to some powe...
Q: Please identify the each stament below whether it is true or false, and leave an explanation. Thank ...
A: Given:- Identify whether it is true or false, and leave an explanation:- 1. All the gases in the w...
Q: Consider the reaction: C(s) + H2O + heat (double arrow) CO(g) + H2, what will happen when you remove...
A: Le Chatelier's principle states that when concentration, temperature or pressure of the system is ch...
Q: pH (calculated and theoretical) of prepared buffer solutions Show all work with units. The answers...
A:
Q: Consider the reaction below. Which of the following would increase the partial pressure of B at equi...
A: Answer: This question is based on Le-chatalier's principle, according to which on changing any param...
Q: 1.0mL of 6.0M HCl is added to 100mL of a buffer that contains equal moles of acetic acid (CH3COOH, K...
A: Initial pH of the buffer containing equal moles of acetic acid and sodium acetate can be given as: p...
Q: What is the coefficient, b, in the reaction below, if Kc = 2.94e-5 and Kp = 9.54e-9 at 402.98°C? ...
A:
Q: Enter your answer in the provided box. For the reaction N2(2) + 3H,(3)5 2NH3(g) Kp is 0.000522 at 59...
A: N2(g) + 3H2 <.....> 2NH3(g) T = 591°C = 591°C + 273 = 864K
Q: Rubidium-87 decays by B production to strontium-87 with a half life of 4.7 x 1010 years. What is the...
A:
Q: Question 8 Indicate whether each sample image demonstrates emulsification or not. 100um 1. Emulsifie...
A: Emulsification is the process of mixing liquids to form an emulsion. It consists of two immiscible l...
Q: Be sure to answer all parts. At 430°C, the equilibrium constant (Kp) for the reaction 2NO(g) + 0;g) ...
A: Recall the given reaction, 2 NO (g) + O2 (g) <-----> 2 NO2 (g), the equilibrium constant ...
Q: consider the reaction: H2(g) + NO(g) arrow forward N2O(g). What is the order of reaction with respec...
A: Detail solution is given below
Q: for h3po3, ka1 = 2.26 x 10-2 and ka2 = 4.79 x 10-7. calculate the ph of a 0.321 molar solution of h3...
A: Given, for h3po3, ka1 = 2.26 x 10-2 and ka2 = 4.79 x 10-7. The ph of a 0.321 molar solution of h3po...
Q: Consider the reaction: N2 (g) + 3 Br2 (g) =2 NBr3 (g) At equilibrium, the concentrations of N2 and B...
A:
Q: QUESTION: The majority of our daily tasks are governed by various thermochemical concepts. What do y...
A:
Q: rate
A:
Q: the injector is kept at higher temperature than the column so that it will move quickly. If the samp...
A: A multiple choice question based on analytical separations that is to be accomplished.
Q: zinc and copper are used in the measurement of food. Are there other topics possible
A: A question based on measurement that is to be accomplished.
Q: I am trying to determine absorbance values to construct a calibration curve. I am given that, 3,6,9,...
A: A question based on analytical process that is to be accomplished.
Q: Consider the I-131 nucleus that is used to treat thyroid. Mass of I-131 is 130.78302 amu. 1) If the ...
A: We have to used equation of first order rate constant. As we know nuclear reaction follows first ord...
Q: For the rate law Rate = k[A][B]3/2, the order with respect to A is ______, the order with respect to...
A:
Q: A 0.60 M solution of a weak acid, HA, has a pH of 3.41. What is the percentage ionization of the aci...
A: ★ Answer : percentage of ionization of the acid is 0.064%
Q: Write the IUPAC name of но, H,
A: The following steps are required for IUPAC naming- 1-> first of all find the longest carbon chain...
Q: Express Ksp in terms of molar solubility, x, for the salts with the given formulas. AB(s) Ksp = AB,(...
A: Since you have posted a question with multiple sub-parts, as per our company guidelines we are suppo...
Q: 12) If gloves are not worn when handling the cuvette in a spectrophotometry xperiment, how would thi...
A: To explain the effect of not wearing the gloves in a spectrophotometry experiment on 1) Absorbance r...
Q: Which of the following is not a polyprotic acid or base? HCl H3PO4 Ca(OH)2 H2SO3...
A: Given, HCl H3PO4 Ca(OH)2 H2SO3
Q: MULTIPLE CHOICE Question 8 How do phosphate-containing fertilizers and detergents lead to low-oxygen...
A: Oxygen is required for survival of living organisms. If proper amount of oxygen is not available the...
Q: the injector is kept at higher temperature than the column so that it will move quickly. If the samp...
A: In the given problem we have to check whether the given two statements are correct or not in referen...
Q: MULTIPLE CHOICE Question 11 Why are POPS so dangerous to organisms? They acidify water, so they can ...
A: POP's are so dangerous for organism because these are persistent, resisting normal processes that br...
Q: Experiment 2: Investigating the Properties of Buffer Solutions Test Data: pH of Cola Solution ...
A: We are given titration data for the addition of cola(pH = 2.5, acid) and borax solution(pH = 9.7, ba...
Q: The solubility of CO2(g) in water at 25 °C and under a pressure of 1.00 atm is 1.45 g/L. If the air ...
A:
Q: Ionic Compounds are highly soluble in water. a) Identify which compound below is water soluble, expl...
A: Polar molecules are the molecules having net dipole moment and non polar molecules have zero dipole ...
Q: A solution is 1.5×10-2 M in Ba?+ and 1.8×10-2 M in Ca²+. Consider that Ksp (BaSO4) ==1.07 × 10¬10 Ks...
A: Given : [Ba+2] = 1.5 x 10-2 M [Ca+2] = 1.8 x 10-2 M Ksp(CaSO4) = 7.1 x 10-5 Ksp (BaSO4) = 1.07 x ...
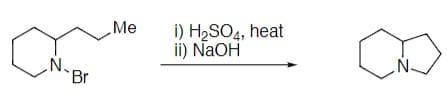
Step by step
Solved in 2 steps with 2 images




