meaker with 1.00x10² mL of an acetic acid butfer with a pH of 5.000 is sitting on a benchtop. The total molarity of acid d conjugate base in this buffer is 0.100 M. A student adds 7.00 mL of a 0.430 M HCl solution to the beaker. How ach will the pH change? The pK, of acetic acid is 4.740. press your answer numerically to two decimal places. Use a minus ( - ) sign if the pH has decreased. View Available Hint(s) ? ApH
meaker with 1.00x10² mL of an acetic acid butfer with a pH of 5.000 is sitting on a benchtop. The total molarity of acid d conjugate base in this buffer is 0.100 M. A student adds 7.00 mL of a 0.430 M HCl solution to the beaker. How ach will the pH change? The pK, of acetic acid is 4.740. press your answer numerically to two decimal places. Use a minus ( - ) sign if the pH has decreased. View Available Hint(s) ? ApH
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter16: Reactions Between Acids And Bases
Section: Chapter Questions
Problem 16.58QE
Related questions
Question
Transcribed Image Text:I Review I Constants I Periodic Table
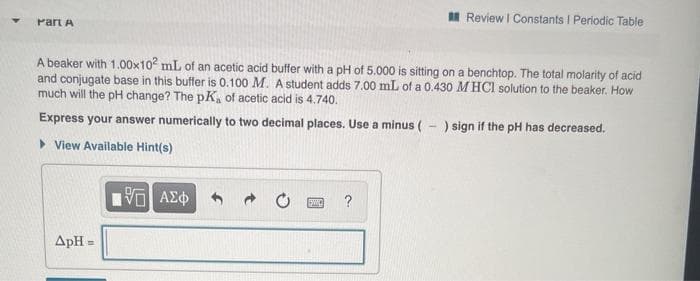
Part A
A beaker with 1.00x102 mL of an acetic acid buffer with a pH of 5.000 is sitting on a benchtop. The total molarity of acid
and conjugate base in this buffer is 0.100 M. A student adds 7.00 mL of a 0.430 M HCI solution to the beaker. How
much will the pH change? The pK, of acetic acid is 4.740.
Express your answer numerically to two decimal places. Use a minus ( - ) sign if the pH has decreased.
> View Available Hint(s)
?
ApH =
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning



Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning



Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole