MODEL 10.2. Tagyngene Perim Carlady Lia N aid Pari NADPI ATP And Inda. ATP Caul NAD Cea NADI derpni ATP ATT ATT NADMI NADI Pre lnd Enerpp proa Tatywid N lu Figure 10.2.1 Catabolism (left) vs. Anabolism (right) (Taken from page 518 of Biochemistry by Garrett and Grisham 4th Edition) The w ing bi tyl
MODEL 10.2. Tagyngene Perim Carlady Lia N aid Pari NADPI ATP And Inda. ATP Caul NAD Cea NADI derpni ATP ATT ATT NADMI NADI Pre lnd Enerpp proa Tatywid N lu Figure 10.2.1 Catabolism (left) vs. Anabolism (right) (Taken from page 518 of Biochemistry by Garrett and Grisham 4th Edition) The w ing bi tyl
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter23: Fatty Acid Catabolism
Section: Chapter Questions
Problem 20P: Understanding a Ubiquitous Series of Metabolic Reactions Study Figure 23.9. Where else in metabolism...
Related questions
Question
100%
Answer for the number 2 question. Thank you. No need for long explanation.
Transcribed Image Text:MODEL 10.2.
Tagyngene
Perim
Carlady
Lia
N aid
Pari
NADPI
ATP
And
Inda.
ATP
Caul
NAD Cea NADI
derpni
ATP
ATT
ATT
NADMI
NADI
Pre lnd
Enerpp proa
Tatywid
N lu
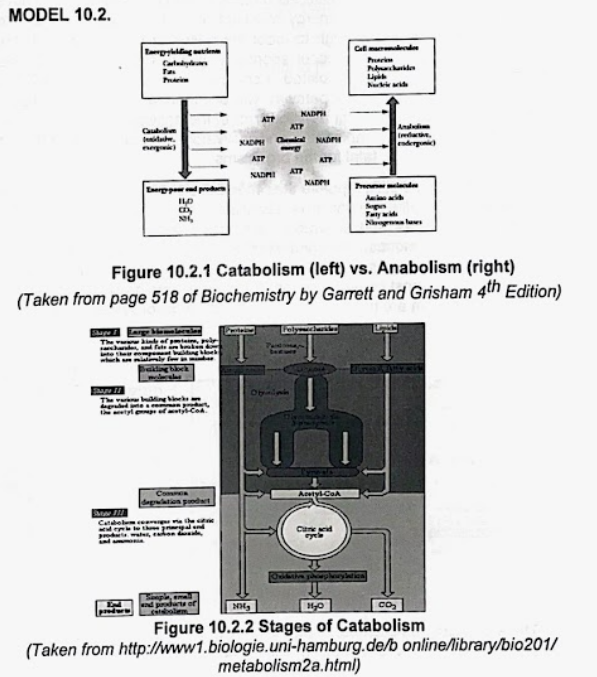
Figure 10.2.1 Catabolism (left) vs. Anabolism (right)
(Taken from page 518 of Biochemistry by Garrett and Grisham 4th Edition)
The v ing
Comn
AcetylCoA
degation pndat
Cadel e
th pgalnd
p , han dle
Cend
a j ry
Co
Figure 10.2.2 Stages of Catabolism
(Taken from http://www1.biologie.uni-hamburg.de/b online/library/bio201/
metabolism2a.html)

Transcribed Image Text:ACTIVITY 10.2
Refer to Figures 10.2.1 and 10.2.2 of Model 10.2 and answer
the question that follows
1. What is metabolism?
2. What are the two types of metabolism?
3. Which metabolic reaction requires an input of energy?
4. Which metabolic reaction releases energy?
5. Which reaction yields a more complex molecule and is
reductive?
6. Which reaction yields a simpler molecule and is oxidative?
7. What are the products of catabolism? Which part of the cell does
this pathway take place?
8. What are the products of anabolism? Which part of the cell does
this pathway take place?
9. Which reaction is divergent? Why?
10. Which reaction is convergent? Why?
11. How can you relate waterfalls to a mole of glucose?
12. What are the steps in catabolism?
13. What is the common pathway of the degraded complex
molecules?
213 Copyright 2019. All Rights Reserved.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning