Q: 2. A solid sample of one of the following polymers (PETE, PP, PVDC and PTFE) was found to be…
A: A solid sample of one of the following polymers (PETE, PP, PVDC and PTFE) was found to be insoluble…
Q: Finding Molecular Weights from Experimental Data a. Endgroup analysis by titration: 9.8 mols of…
A:
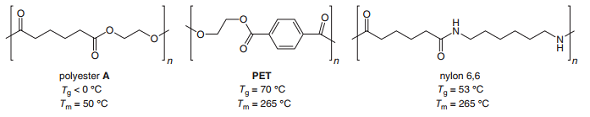
Q: Explain the differences observed in the T, and Tm values for each pair of polymers: (a) polyester A…
A: Given that, a)
Q: An amino acid mixture of phenylalanine, glycine and glutamic acid is to be separated bypaper…
A: As multiple questions are mentioned here, so the solution to the first part is given here. Paper…
Q: What proportion of benzoic acid should be used with an equimolar mixture of adipic acid and…
A:
Q: The polyester can be mixed with additional phthalic anhydride (0.5 mole of phthalic anhydride for…
A: (a)
Q: 2. The a chain of haptoglobin exhibits genetic polymorphism since glu- tamic acid can be replaced by…
A: 2)First, we will see the difference between the SDS and Native polyacrylamide gel electrophoresis…
Q: Q5. Give one condition for the denaturation step employed in blotting
A: As per the guideline, since you have asked multiple questions, we have solved the first question for…
Q: How do strong and weak synthetic ion-exchange resins differ in structure?
A: Ion-exchange resins: Resins with large pours cavity is hold the ions by its ionic property. Cationic…
Q: In the following reaction. What is the purpose of the hexanes in the reaction? - NH2 H2N NH2 NaOH,…
A: Hexanes, NaOH and H2O are used for the synthesis of nylon-6,6.
Q: Write polymerization reaction scheme for Polymerization of styrene with benzoyl peroxide as…
A:
Q: When styrene (vinylbenzene) is commercially polymerized, about 1–3% of 1,4-divinylbenzene is often…
A:
Q: When styrene (vinylbenzene) is commercially polymerized, about 1–3% of 1,4-divinylbenzene is often…
A:
Q: Give introduction to olefins ?
A: Olefin, also called alkene.
Q: One of the earliest commercial plastics was Bakelite®, formed by the reaction of phenol with a…
A: Bakelite is a condensation polymer of phenol and formaldehyde. It is a thermosetting polymer. It is…
Q: An equimolar blend of 1,4-hydroquinone diallylether and ethylene glycol dimercaptoacetate containing…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: .A linear polyester was synthesized from a mixture of diacid and glycol with carboxyl to hydroxyl…
A: In order to find the carboxyl functionality of the polymer, end group analysis can be done. The…
Q: Two samples of polystyrene, samples A and B, were synthesised. A from the addition of n- butyl…
A: Introduction: Polymers: Polymers are the large molecules made up of repeating units. The physical…
Q: Explain why the addition of a small amount of glycerol to the polymerization mixture gives a stiffer…
A: The addition of a small amount of glycerol to the polymerization mixture gives a stiffer urethane…
Q: Why does a solid form at the interface of the two solutions? PROCEDURE Synthesis of Nylon In a…
A: Given that, the synthesis of Nylon-6,10. We have to tell why the solid polymer formed at the…
Q: To meet the ever increasing demand of global population, the demand for textile products and…
A: When a color-producing substance is involved in bonding with a material on which it is applied and…
Q: A solid sample of one of the following polymers (PETE, PP, PVDC and PTFE) was found to be insoluble…
A: PETE, PVDC and PTFE has either halogen atom like chloro, fluoro group or ester group in monomer unit…
Q: Describe the preparation of i. 500 mL of 3.50% (w/v) aqueous glycerol
A:
Q: Calculate the acid number of a polyester with the molecular weight of its repeating unit of 120…
A: Acid number (AN) of the polymer describes the number of free acid that can be neutralized by the…
Q: 17. What is the Ceiling and fluidity temperature ? 18. This polystyrene has a number average…
A: "Since you have asked multiple question, we will solve the first question for you preferably. If you…
Q: The difference in melting points between trans-oleic acid (18:1,D9)(44.5C) and cis-oleic acid…
A: given data is trans-oleic acid (18:1,D9) melting point is (44.50C) cis-oleic acid melting point is…
Q: Which layer (aqueous or organic) will phenylalanine partition into during extraction? water or…
A: Solubility depends on the polarity of the compound and solvents .
Q: 1-What is the mechanical properties of a hardened monofilament nylon? 2- What is the processing…
A: The simplest chemical unit that has key functional groups such as the unsaturation unit is known as…
Q: (CH,), -N- nylon 6 the O H H (CH,), -C-N-(CH,),2-N- nylon 6,12 In What is the mole fraction of…
A: The number average molecular weight of a polymer can be written as the product of number average…
Q: The polyester can be mixed with additional phthalic anhydride (0.5 mole of phthalic anhydride for…
A: Given reaction is,
Q: For end-group analysis, 0.8632 g of a carboxyl terminated polybutadiene (CTPB) sample dissolved in…
A: The amount of potassium hydroxide, KOH, used in the titration is; mKOH=VKOH×Eq.wtKOH×NKOH1000 mL…
Q: What is the role of benzoyl peroxide in addition polymerisation of alkenes? Explain its mode of…
A: Benzoyl peroxide acts as initiator in polymerization of alkenes by giving chain reactions. The…
Q: Give a single benefit for each polymerisation from carrying them out under a nitrogen atmosphere?…
A: Introduction: The polydispersity index (PDI) is dependent on the two parameters, i.e (I) number…
Q: What are the fundamental differences between a good solvent and a theta solvent, and how do they…
A: Good solvent And theta Solvent differs in interaction in polymer chains
Q: Propose a suitable process flow sheet in accordance with the required equipment (such as smelter,…
A: Solution: We know nylon 6 is prepared from caprolactam by ring opening reaction. Therefore, it is…
Q: What monomers would you use to produce analogous polyester fibers?
A: Nylon is a synthetic polymer composed of repeating units linked by amide links. Whereas, polyesters…
Q: How can I calculate polycarboxylate(liquid) density?
A: To explain: The procedure for calculating the density of polycarboxylate liquid.
Q: Explain the differences observed in the Tg and Tm values for each pair of polymers: (a) polyester A…
A: (a) The glass transition temperatureTg of Polyester A is less than 0°C and for PET it is 70°C. It…
Q: If linear siloxane polymers are required, dimethyldichlorosilane with a high purity is necessary,…
A: A polymer is a substance or material made up of very large molecules, known as macromolecules, that…
Q: Explain the Synthesis of adipic acid and 1,6-diaminohexane for nylon 6,6 synthesis ?
A:
Q: Define Polylactic acid dissolvining in chloroform?
A: Polylactic acid (PLA) is a thermoplastic polymer having monomer formula is (C3H4O2)n or…
Q: How many moles of monofunctional A must be added to a mixture of one mole of B2 and one mole of A3…
A:
Q: A sample of polystyrene is composed of a series of fractions of different sized molecules.…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: You want to determine the molar mass of the polymer shown below by end group analysis using…
A: In this determine molecular mass of polymer with the help of given data and then determine value of…
Q: What information about polymers is obtained by using a magnet during NMR spectroscopy
A: The role of the magnet is to polarize the nuclei to produce a net magnetization within the sample.…
Q: 6-Calculate molecular weight, weight average molecular weight, and polydispersity index for the…
A: Number average molecular weight of the polymer / molecular weight of the polymer (Mn) =∑1iNiMi∑1iNi…
Q: Explain why the preparation of Nylon-6,10 occurs under milder conditions when decanediocdiacid…
A: Nylon 6,10 is a semicrystalline polyamide. It contains 16 carbon atoms in their structure. It is…
Q: Why does the nylon form at the interface of sebacoyl chloride and hexamethylene diamine?
A: We have to discuss the reason as to why nylon is formed at the interface of sebacoyl chloride and…
Explain the differences observed in the Tg and Tm values for each pair of
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 5 images

- Hexanoic acid was added to an immiscible biphasic solvent sysem, water and CCl4 at 20.0OC and the equilibrium concentrations of hexanoic acid were determined to be 3.66 g/L in H2O and 67.0 g/L in CCl4. Caluclate the distrubution coeffiecent (D1) of hexanoic acid in CCl4 with respect to water.At constant pressure and 25°C, what is ΔrH° for the following reaction2C2H6(g) + 7O2(g) → 4CO2(g) + H2O(l)if the complete consumption of 14.5 g of C2H6 liberates 752.3 kJ of heat energy?I am not sure how they got 10 ns^-1, this is occurring at SATP conditions. .
- Phenol reacts with three equivalents of bromine in CCl4 (in the dark) to give a product of formula C6H3OBr3. When thisproduct is added to bromine water, a yellow solid of molecular formula C6H2OBr4 precipitates out of the solution. The IRspectrum of the yellow precipitate shows a strong absorption (much like that of a quinone) around 1680 cm-1. Proposestructures for the two productsCalculate the amount of phycocyanin in Sample 1 in mg where A620=0.211 and A650=0.086, taking into account the dilution factor as per question 6 (100ul), and the total volume of extract as per question 4 (140ml) . Note your answer to 2 decimal placesIs the product pure? how do you know? here is the results: Starting mass of benzyl 1.0883g Collected mass of product 0.8957g Product melting point 135˚C Mixture melting point (product mixed with benzoin) 114˚C Mixture melting point (product mixed with meso-hydrobenzoin) 135˚C
- Suppose you have a soil that is made up of 10% organic matter with CEC = 200 cmol/kg, 40% kaolinitewith CEC = 10 cmol/kg, and 50% vermiculite with CEC = 100 cmol/kg.1- Calculate the overall CEC of the soil by taking a weighted average of the three soil components.2- What percentage of the overall CEC is contributed by the organic matter? How does that numbercompare to the 10% of the soil itself that is organic?The Ksp of Al(OH)3 (MM: 78) is 1.90 xx 10^(-33). a) What is the solubility of Al(OH)3 in g/L?D. Preparation of 3% (vol/vol) Alcohol Solution1. Compute for the volume of denatured ethanol needed to prepare 25 mL of 3% (vol/vol) solution. Note that the concentration of the denatured alcohol stock solution is 98%.2. Explain in detail how the solution will be prepared in the laboratory.

